
10�֣�ij��ѧ��ȤС���ijƷ��������Ħ�����ɷּ��京����������̽����
������ϣ�������Ħ������̼��ơ�����������ɣ������������ɷ���������ʱ���������ɡ�
��.Ħ���������������Ķ��Լ���
ȡ����������Ʒ����ˮ�ɷֽ��衢���ˡ�
��1���������м������NaOH��Һ�����ˡ�����������NaOH��Һ��Ӧ�����ӷ���ʽ��__________________________ ___ ______��
��2������1��������Һ����ͨ�����������̼���ټ������ϡ���ᡣ�۲쵽��������______________________ ___ ______��
��.������Ʒ��̼��ƵĶ����ⶨ
������ͼ��ʾװ�ã�ͼ�мг�������ȥ������ʵ�飬��ַ�Ӧ�ⶨC�����ɵ�BaCO3������������ȷ��̼��Ƶ�����������
����ʵ����̻ش��������⣺
��3��ʵ����������������ͨ������������ó��˿ɽ���B��C�еķ�Ӧ���⣬���У�____________________ _____________��
��4��C�з�Ӧ����BaCO3�Ļ�ѧ����ʽ��_______________________ _________��
��5��ʵ����ȷ��ȡ8.00g��Ʒ���ݣ��������βⶨ�����BaCO3ƽ������Ϊ3.94g������Ʒ��̼��Ƶ���������Ϊ_________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�갲��ʡ������ѧ�ڵ�һ���¿���ѧ�Ծ� ���ͣ�ʵ����
(10��)ij��ѧ��ȤС�����ʯ����ˮ��Ӧ���������ȷ�Ӧ������ʵ��̽�����ڳ������ִ����Թܱڸо������⣬����������м��������Է�����
���������¶ȼ���ʢ������ʯ�ҵ�С�Թ�����Ƥ��������һ�𣬷�����ˮ��С�ձ��У��ý�ͷ�ι���С�Թ��л�������ˮ�������������� ��˵����Ӧ���ȡ�������װ����֧�ż��������������ȥ��
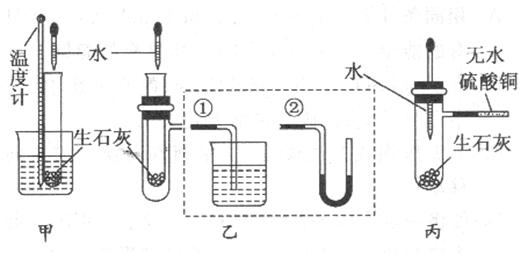
�ҷ�������ʢ������ʯ�ҵ�С�Թܲ����֧�ܵ��Թ��У�֧�ܽӢٻ�ڣ��ý�ͷ�ι���С�Թ��л�������ˮ��������������(�Ӣ�) ��(�Ӣ�) ��˵����Ӧ���ȡ�
���������ý�ͷ�ι���ʢ������ʯ���Ҵ�֧�ܵ��Թ��еμ�ˮ��֧�ܽӵĵ�����ʢ������ˮ����ͭ��ĩ�������������� ��˵����Ӧ���ȣ���ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��0110 ģ���� ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡģ���� ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(10��)ij��ѧ��ȤС�����ʯ����ˮ��Ӧ���������ȷ�Ӧ������ʵ��̽�����ڳ������ִ����Թܱڸо������⣬����������м��������Է�����
���������¶ȼ���ʢ������ʯ�ҵ�С�Թ�����Ƥ��������һ�𣬷�����ˮ��С�ձ��У��ý�ͷ�ι���С�Թ��л�������ˮ�������������� ��˵����Ӧ���ȡ�������װ����֧�ż��������������ȥ��
�ҷ�������ʢ������ʯ�ҵ�С�Թܲ����֧�ܵ��Թ��У�֧�ܽӢٻ�ڣ��ý�ͷ�ι���С�Թ��л�������ˮ��������������(�Ӣ�) ��(�Ӣ�) ��˵����Ӧ���ȡ�
���������ý�ͷ�ι���ʢ������ʯ���Ҵ�֧�ܵ��Թ��еμ�ˮ��֧�ܽӵĵ�����ʢ������ˮ����ͭ��ĩ�������������� ��˵����Ӧ���ȣ���ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�찲��ʡͩ��ʮ�и�����ѧ�ڵ�һ���¿���ѧ�Ծ� ���ͣ�ʵ����
(10��)ij��ѧ��ȤС�����ʯ����ˮ��Ӧ���������ȷ�Ӧ������ʵ��̽�����ڳ������ִ����Թܱڸо������⣬����������м��������Է�����
���������¶ȼ���ʢ������ʯ�ҵ�С�Թ�����Ƥ��������һ�𣬷�����ˮ��С�ձ��У��ý�ͷ�ι���С�Թ��л�������ˮ���������� ���� ��˵����Ӧ���ȡ�������װ����֧�ż��������������ȥ��
���� ��˵����Ӧ���ȡ�������װ����֧�ż��������������ȥ��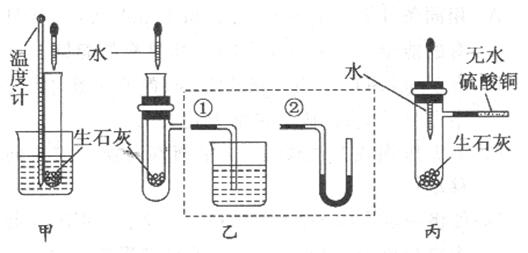
�ҷ�������ʢ������ʯ�ҵ�С�Թܲ����֧�ܵ��Թ��У�֧�ܽӢٻ�ڣ��ý�ͷ�ι���С�Թ��л�������ˮ��������������(�Ӣ�) ��(�Ӣ�) ��˵����Ӧ���ȡ�
���������ý�ͷ�ι���ʢ������ʯ���Ҵ�֧�ܵ��Թ��еμ�ˮ��֧�ܽӵĵ�����ʢ������ˮ����ͭ��ĩ�������������� ��˵����Ӧ���ȣ���ԭ����  ��
��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com