
���� ��1�����������е���Ϊ+4�ۣ����л�ԭ�ԣ������е��������������ԣ�
��2�������ľ���Ϊ��NH4��2SO3��NH4HSO3�����е�һ�ִ����ɽᾧˮ�������ᰱ�е���Ϊ+4�ۣ��ױ������е�����������
��3��KMnO4��Һ��ϡH2SO4 ������������������ɫ��KMnO4��Һ��ɫ���������������������ԭ��Ӧ��������������ɫ��
��� �⣺��1����β��ͨ�백ˮ�У�β���е�SO2�백ˮ��Ӧ����ˮ����ʱ��SO2+NH3•H2O=NH4HSO3����ˮ����ʱ��SO2+2NH3•H2O=��NH4��2SO3+H2O�����ɵ�������Ļ��ϼ�Ϊ+4�ۣ������������������ߵ�+6�ۣ�
�ʴ�Ϊ��2��NH4��2SO3+O2�T2��NH4��2SO4��2NH4HSO3+O2�T2NH4HSO4��
��2������β���백ˮ��Ӧ���õ��ĸ�Ũ����Һ�У���һ���������백ˮ��̼����泥���ˮ�������������NH4HSO3��������̼����立�Ӧ����Ϊ����������Ա�̼��ǿ���ʸýᾧˮ����Ϊ��NH4��2SO3�����ɽᾧˮ����NH4��2SO3��ʽ��Ϊ116����֪�ýᾧˮ�������Է�������Ϊ134��134-116=18���������к���1���ᾧˮ����ѧʽΪ��NH4��2SO3•H2O��
�ʴ�Ϊ����NH4��2SO3•H2O��
�ڶԱ����Ӿ��л�ԭ�ԣ������е��������������ԣ������ᰱ��+4�۵�����л�ԭ�ԣ�
�ʴ�Ϊ����ֹ������隣�������
��3��KMnO4��Һ��ϡH2SO4 ���������Ӧ�ķ���ʽΪ��5SO2+2MnO4-+2H2O=2Mn2++4H++5SO42-���������������������ԭ�ķ���ʽΪ��SO2+I2+2H2O=H2SO4+2HI���������꣬������û�еⵥ�ʲ�������ɫ��
�ʴ�Ϊ��BC��
���� ���⿼����������ԭ��Ӧ�����ӷ���ʽ����д�����ʵļ��顢Ԫ�ػ���������ʣ���Ŀ�Ѷ��еȣ������ڿ���ѧ���ķ��������ͶԻ���֪ʶ���ۺ�Ӧ��������ע��ӻ��ϼ۵ĽǶȷ���������ԭ��Ӧ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
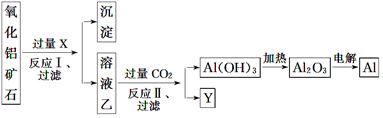
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| ѡ�� | ʵ����� | ���� | ���� |
| A | ��ϡ�����м�����������۳�ַ�Ӧ����KSCN��Һ | ��Һ��ΪѪ��ɫ | HNO3���������ԣ��ܽ�Fe������Fe3+ |
| B | ��ʢ��ij��Һ���Թ��еμ�NaOH��Һ����ʪ��ĺ�ɫʯ����ֽ�����Թܿ� | ��ֽ��ɫ�����Ա仯 | ԭ��Һ����NH4+ |
| C | �������Һ�м���ϡ���ᣬ���ȣ���ȴ���������Cu��OH��2���ټ��� | δ����ɫ���� | ����δ����ˮ�� |
| D | ���з�̪��Na2CO3��Һ�м�������BaCl2���� | ��Һ��ɫ��dz | ֤��Na2CO3��Һ�д���ˮ��ƽ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��״���£�3.36LC2H4��C3H6�Ļ�������к���̼̼˫������ĿΪ0.15NA | |
| B�� | 0.1mol•L-1��NH4��2SO4��Һ��0.2mol•L-1NH4Cl��Һ�е�NH4+��Ŀ��ͬ | |
| C�� | H2��CO�������8.96L������O2�г��ȼ������O2������Ϊ0.2NA | |
| D�� | ��0.1molNH4HSO4����Һ�У���������Ŀ�Դ���0.21NA |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ƶ���
| ������ | Na+ | Al3+ | Fe3+ | Cu2+ | Ba2+ |
| ������ | OH- | Cl- | CO32- | NO3- | SO42- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
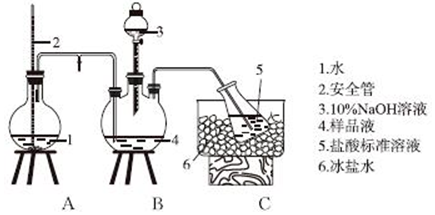
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Cl-һ������ | |
| B�� | CO32-��SO42-һ�����ڣ��Ҷ��ߵĸ�����Ϊ1��2 | |
| C�� | Zn2+��Ca2+һ�������� | |
| D�� | 100mL��Һ��K+��������С��2.34g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �۵㣺���ʯ���ɱ� | B�� | ���Ӱ뾶��O2-��Na+ | ||
| C�� | ���ԣ�KOH��Al��OH��3 | D�� | �ȶ��ԣ�SiH4��H2S |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com