
| 14mg | ||
|
| 1000mL |
| 20mL |
| 10g��35%-3.36g |
| 10g��35% |


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Fe�����岻��ȱ�ٵ���Ԫ�أ����뺬��������ɲ�������
�� ������ÿ��Ӧ����14mg���ҵ��������о���������ʳ����ȫ��ͨ�����ú�FeSO4��7H2O��Ƭ��������������������ÿ������ú� mg FeSO4��7H2O��Ƭ����
�� �������ơ����г���һ�ֳ����IJ���ҩ�����Fe2���ĺ���Ϊ35.0%����ҩƷ��Fe2���Ỻ�����������ҹ涨��ҩ����Fe2���������ʳ���10���������ٷ��á�Ϊ����ijҩ����۵ġ������ơ��Ƿ�ʧЧ��ȡ10.00g��ҩƷȫ������ϡ���ᣬ���Ƴ�1000mL��Һ��ȡ����20.00mL����0.01000 mol/L KMnO4��Һ�ζ�����ȥKMnO4��Һ24.00mL��ͨ������˵����ҩ���Ƿ��ܷ��ã���MnO4�������������µĻ�ԭ����ΪMn2����ҩƷ�г�FeԪ���������ɷֲ���KMnO4��Ӧ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��㶫ʡ����һ�и���8���¿������ۣ���ѧ���� ���ͣ������
��15�֣������仯����������������й㷺Ӧ�á���ش��������⣺
��1��Fe�����岻��ȱ�ٵ���Ԫ�أ����뺬��������ɲ�������������ÿ��Ӧ����14mg���ҵ��������о���������ʳ����ȫ��ͨ�����ú�FeSO4��7H2O��Ƭ��������������������ÿ������ú� mg FeSO4��7H2O��Ƭ����
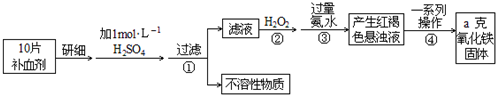
��2��ijͬѧΪ�˼�����е�һƿ����ҩ���ɷ�ΪFeSO4���Ƿ���ʣ��������й����ϣ���֪Fe2+�ܱ����Ը��������Һ������ʹ���������Һ��ɫ���������ѧ��֪ʶ���������ʵ�飺��ҩƬ��ȥ������ϸ���ܽ���ˣ�ȡ��Һ�ֱ������֧�Թ��У���һ֧�Թ��е������Ը��������Һ������һ֧�Թ��е���KSCN��Һ����ͬѧ�۲쵽�������ǣ��������Ը��������Һ����ɫ������KSCN��Һ��죬�ݴ˵ó��Ľ����� ��
��3��������FeS2�������������ұ����������Ҫԭ�ϡ�����һ����ӦΪ
3FeS2��8O2 6SO2��Fe3O4������3 mol FeS2�μӷ�Ӧ����ת�� mol���ӡ�
6SO2��Fe3O4������3 mol FeS2�μӷ�Ӧ����ת�� mol���ӡ�
��4���Ȼ�����Һ������ӡˢ��·ͭ�帯ʴ������Ӧ�����ӷ���ʽΪ ��
��5�����������ƣ�������Ҳ��������ˮ������ʹ��ʱ����������������ʹ���Է�ˮ�е������������ȥ����ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��������2010��������Ĵ��ʼ쿼�ԣ���ѧ������ ���ͣ�������
(8��) Fe�����岻��ȱ�ٵ���Ԫ�أ����뺬��������ɲ�������
�� ������ÿ��Ӧ����14mg���ҵ��������о���������ʳ����ȫ��ͨ�����ú�FeSO4��7H2O��Ƭ��������������������ÿ������ú� mg FeSO4��7H2O��Ƭ����
�� �������ơ����г���һ�ֳ����IJ���ҩ�����Fe2���ĺ���Ϊ35.0%����ҩƷ��Fe2���Ỻ�����������ҹ涨��ҩ����Fe2���������ʳ���10���������ٷ��á�Ϊ����ijҩ����۵ġ������ơ��Ƿ�ʧЧ��ȡ10.00g��ҩƷȫ������ϡ���ᣬ���Ƴ�1000mL��Һ��ȡ����20.00mL����0.01000 mol/L KMnO4��Һ�ζ�����ȥKMnO4��Һ24.00mL��ͨ������˵����ҩ���Ƿ��ܷ��ã���MnO4�������������µĻ�ԭ����ΪMn2����ҩƷ�г�FeԪ���������ɷֲ���KMnO4��Ӧ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com