��������1����ͭ����Ҫ�ɷ���Cu
2S��ұ��ͭ�������Ը��������Һ��Ӧ����ͭ���ӣ���������ӣ�����������ӱ���ԭΪ�����ӣ����ԭ���غ�͵���غ���ƽ��д����
��2��������Һ�����������ʵ�����������������ʵ�����������������ʵ�������Ϸ�Ӧ3Cu+8H
++2NO
3-=3Cu
2++2NO��+4H
2O�������Ӷ�����ϵ��Ԫ���غ���������
��3��������ͼ����д��Ӧ�����е��Ȼ�ѧ����ʽ����ϸ����������������Ȼ�ѧ����ʽ��
�����ݻ�ѧ����ʽ���պ��������䣬��Ӧ�����������ɵĶ�����̼��ˮ��������ͬ����Ͻ���������=
| ������Ľ������� |
| ������������ |
��100%������õ���
��4����������ͭ��������������������������ͭ��д��ѧ����ʽ��
��������ͭ�ܽ��С����п������ͭ��Һ������п��������ת����Ӧ������ͭ��
���
�⣺��1����ͭ����Ҫ�ɷ���Cu
2S��ұ��ͭ�������Ը��������Һ��Ӧ����ͭ���ӣ���������ӣ�����������ӱ���ԭΪ�����ӣ���Ӧ�����ӷ���ʽΪ��Cu
2S+2MnO
4-+8H
+=2Cu
2++SO
42-+2Mn
2++4H
2O��
�ʴ�Ϊ��Cu
2S+2MnO
4-+8H
+=2Cu
2++SO
42-+2Mn
2++4H
2O��
��2��ij����c��HNO
3��=2mol?L
-1��c��H
2SO
4��=4mol?L
-1�ķ�����Һ100mL�����������������������n��H
+��=0.1L��2mol/L+0.1L��4mol/L��2=1mol��n��NO
3-��=0.1L��2mol/L=0.2mol��n��SO
42-��=0.1L��4mol/L=0.4mol��3Cu+8H
++2NO
3-=3Cu
2++2NO��+4H
2O�����ݶ�����ϵ��֪���������ȫ����Ӧ����������0.8mol��ͭ0.3mol��������������ͭ�������Ϊ0.3mol��������Ʊ�����ͭ���壨CuSO
4?5H
2O��������=0.3mol��250g/mol=75g��
�ʴ�Ϊ��75g��
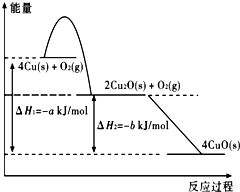
��3��������ͼ�������д�Ȼ�ѧ����ʽ����2Cu��s��+
O
2��g��=2CuO��s����H=-
KJ/mol����2Cu
2O��s��+O
2��g��=4CuO��s����H=-bKJ/mol��
���ݸ�˹���ɼ��㣨�١�2-�ڣ���
�õ�2Cu��s��+
O
2��g��=Cu
2O��s����H=-��
��kJ?mol
-1��
�ʴ�Ϊ��2Cu��s��+
O
2��g��=Cu
2O��s����H=-��
��kJ?mol
-1��
�ں���ͭ�̵�ͭƬ�����費���������ʣ��ڿ�������������ȫ��Ӧ�����ⶨ����Ӧǰ������������ͬ��˵����Ӧ���������������ɵĶ�����̼��ˮ������������ͬ����õ�����ͭ����ͭ���ʵ���Ϊx��ͭ�����ʵ���Ϊy��
2Cu+O
22CuO��
x 0.5x
Cu
2��OH��
2CO
32CuO+CO
2+H
2O
y y y
0.5x��32=18y+44y
x��y=31��8
����Ԫ���غ���㣬����������=
| ������Ľ������� |
| ������������ |
��100%=
��100%=
��100%=34%��
�ʴ�Ϊ��34%��
��4������ͭ��������������������������ͭ����Ӧ�Ļ�ѧ����ʽΪ��CuS+2O
2 CuSO
4 ��
�ʴ�Ϊ��CuS+2O
2 CuSO
4 ��
������������������ͭ��Һ������п����һ�������£��ܽ��С�Ŀ������ת��Ϊ�ܽ�ȸ�С�Ŀ��������ͬ�����£�����K
SP��CuS����K
SP��ZnS�����ʷ�Ӧ���Է���������ͭ�ܽ���С����п����������ת����Ӧ����Ӧ�����ӷ���ʽ��Cu
2+��aq��+ZnS��s��=CuS��s��+Zn
2+��aq����
�ʴ�Ϊ��Cu
2+��aq��+ZnS��s��=CuS��s��+Zn
2+��aq����һ�������£��ܽ��С�Ŀ������ת��Ϊ�ܽ�ȸ�С�Ŀ��������ͬ�����£�����K
SP��CuS����K
SP��ZnS�����ʷ�Ӧ���Է�������

 ͭ���仯�����ڹ�ҵ��ũҵ���Ƽ����ճ��������й㷺Ӧ�ã�
ͭ���仯�����ڹ�ҵ��ũҵ���Ƽ����ճ��������й㷺Ӧ�ã�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�