
��ͼ��Ԫ�����ڱ��Ŀ��
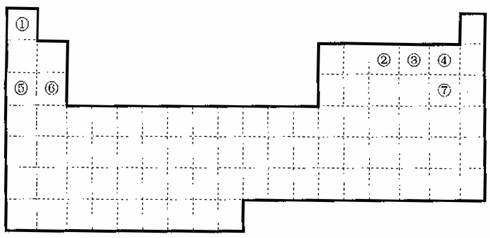
��1����������Ԫ�����ڱ��л�������Ԫ����ǽ���Ԫ�صķֽ��ߡ�
��2������Ԫ�����ڱ��ش��������⣺
A�����ڱ��е�Ԫ�آݺ�Ԫ�آ�����������ˮ�������ǿ��˳����
���û�ѧʽ��ʾ����
B�������б���Ԫ�آܺ�Ԫ�آߵ��⻯����ۡ��е�ߵ�˳����
���û�ѧʽ��ʾ����
C���٨D��Ԫ�ص�ij�����г����»�ѧ�����ȶ���ͨ�����Ա���������
���û�ѧʽ��ʾ����
D��������Ԫ�����ڱ���ȫ���ǽ���Ԫ�ص������� ��ȫ���Ƿǽ���Ԫ�ص������� ����д��ĸa��b��c��d����
a��IA�� b����A�� c����A�� d��VIIA��
 ��3����֪��Ԫ��λ�ڵ������ڣ�����ԭ�Ӱ뾶Ϊͬ���ڽ���Ԫ���е�ԭ�Ӱ뾶��С�ģ���д������������NaOH��Һ��Ӧ�����ӷ���ʽ ����һ��������1g�ٵĵ����������۵ĵ�������ȫȼ������Һ̬����ʱ���ų�������ΪakJ����д���������±�ʾ�ٵĵ���ȼ�յ��Ȼ�ѧ����ʽ ��
��3����֪��Ԫ��λ�ڵ������ڣ�����ԭ�Ӱ뾶Ϊͬ���ڽ���Ԫ���е�ԭ�Ӱ뾶��С�ģ���д������������NaOH��Һ��Ӧ�����ӷ���ʽ ����һ��������1g�ٵĵ����������۵ĵ�������ȫȼ������Һ̬����ʱ���ų�������ΪakJ����д���������±�ʾ�ٵĵ���ȼ�յ��Ȼ�ѧ����ʽ ��
�١�������Ԫ�صĵ����ѱ�Ӧ��������ɴ���ȼ�ϵ���У���ͼ��ʾ�������缫���ɶ����̼���ɣ�ͨ������ֵ����ɿ�϶�ݳ����ڵ缫����ŵ硣
��ش�b�ǵ�ص� ����a�缫�ϵĵ缫��Ӧʽ�� ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
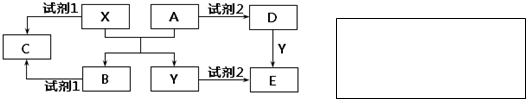
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�������ʮ�����ص�ѧУ�����߿���ѧģ���Ծ���һ���������棩 ���ͣ������

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com