
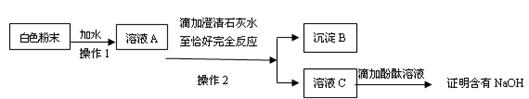
 Ȧ�����ձ�����ֽ���������⣬���貹��IJ��������� _______��
Ȧ�����ձ�����ֽ���������⣬���貹��IJ��������� _______�� ʵ�飬ʵ�������С���һ�¡��ɴ�ȷ�����������ȷ��
ʵ�飬ʵ�������С���һ�¡��ɴ�ȷ�����������ȷ�� ��С�ոĽ��ķ�����_______ ��
��С�ոĽ��ķ�����_______ ��
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ѡ�� | ���缫 | ����� | ������Ӧ | ������Ӧ |
| A | ���� | NaOH | Al-3e-��Al3+ | 2H2O+2e-��2OH-+H2�� |
| B | ���� | ϡ���� | 2Al-6e-=2Al3+ | 6H++6e-��3H2�� |
| C | ���� | Ũ���� | Cu-2e--��Cu2+ | 2NO3-+4H+-4e-��2NO2��+2H2O |
| D | ���� | ϡ���� | Cu-2e-��Cu2+ | 2NO3-+8H+��2NO��+4H2O+6e- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ��һ�ִ�����Ⱦ�ij��ȤС����̽��
��һ�ִ�����Ⱦ�ij��ȤС����̽�� �����ʼ���ɫʵ��ķ�����������·�����
�����ʼ���ɫʵ��ķ�����������·�����


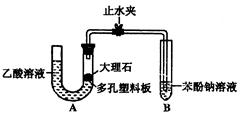
 I����֤
I����֤ �������ԣ�Cװ���п�ѡ�Լ� (�����)��
�������ԣ�Cװ���п�ѡ�Լ� (�����)��| A��Ba(HCO3)2��Һ | B�������� | C����ˮ | D��Ʒ����Һ |
 �Ļ�ԭ�ԣ�Cװ���п����Լ� (������)��
�Ļ�ԭ�ԣ�Cװ���п����Լ� (������)�� �Ļ�ѧ��Ӧ����ʽΪ��___________________________________________________��
�Ļ�ѧ��Ӧ����ʽΪ��___________________________________________________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
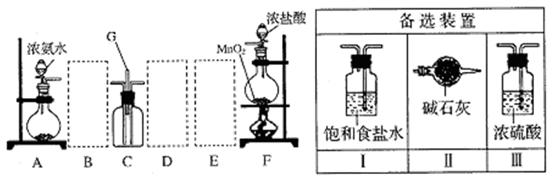
 ��#��#��#Դ��ͼ�е�E��F��������װ�ã������ⶨO2�������
��#��#��#Դ��ͼ�е�E��F��������װ�ã������ⶨO2��������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ����� | ���� | ���� |
| ����һ�� | ������ɫ���� | |
| ������� | | ������������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ��������� | ʵ�鲽�� | ʵ������ | ʵ����� |
| ����2��Сֽ���е����� �ܷ������������� | ȡ����Сֽ���й�������ձ��У���������ˮ���������ڡ� | | ������ ����� |
| ����3���Ҳ�����ʺ�����ʿ�����̼��ƣ��������֤�ҵIJ��룿 | | | �ø������Ʒ����̼��� |
 .
.�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com