
������һ��ǿ������������������������ȡ�
(1)O3��KI��Һ��Ӧ���ɵ����ֵ�����________��________(�����ʽ)��
(2)O3��ˮ���ֽ⣬һ�������£�O3��Ũ�ȼ���һ�������ʱ��(t)���±���ʾ����֪��O3����ʼŨ��Ϊ0.0216 mol/L��
��pH�����ܼ���O3�ֽ⣬������O3�ֽ�������õ���________��
����30�桢pH��4.0�����£�O3�ķֽ�����Ϊ________mol/(L��min)��
�۾ݱ��еĵݱ���ɣ��Ʋ�O3�����������·ֽ��������������˳��Ϊ________(����ĸ����)��
a��40�桢pH��3.0������ b��10�桢pH��4.0
c��30�桢pH��7.0
(3)O3���ɳ���������(ԭ����ͼ)���ϡ�����Ƶá�
��ͼ������Ϊ________(�A����B��)����缫��ӦʽΪ_______________________��
����C��ͨ��O2����A���ĵ缫��ӦʽΪ________��
����C����ͨ��O2��D��E���ֱ��ռ���x L��y L����(��״��)����E���ռ���������O3��ռ���������Ϊ________(����O3�ķֽ�)��
 ��ѧ�����ϵ�д�
��ѧ�����ϵ�д� �·Ƿ��̸����100��ϵ�д�
�·Ƿ��̸����100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��֪I����Fe2+��SO2��Cl����H2O2���л�ԭ�ԣ�������������Һ�л�ԭ�Ե�ǿ��˳��ΪCl��< Fe2+< H2O2< I��< SO2�������з�Ӧ���ܷ�������
| A��2Fe3++SO2+2H2O=2Fe2++SO42��+4H+ |
| B��Cl2+SO2+2H2O=H2SO4+2HCl |
| C��H2O2+2H++ SO42��=SO2��+O2��+2H2O |
| D��2I2+H2O2=2H2O+O2��+4 I�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������16�֣������ƾ���(Na2MoO4��2H2O)����������ȴˮϵͳ�Ľ�����ʴ�������⾫����Ҫ�ɷ���MoS2��������PbS�ȣ��Ʊ������ƾ���IJ����������£� ��1�����յĹ����в��õ��ǡ��������ա��Ĵ�ʩ����ô�ʩ���ŵ��ǣ���_______________
��1�����յĹ����в��õ��ǡ��������ա��Ĵ�ʩ����ô�ʩ���ŵ��ǣ���_______________
��____________________________
��2��д������ʱ����MoO3�Ļ�ѧ����ʽΪ��______________________________________
��3��д�����������Ӧ�����ӷ���ʽ���� ��������������������
��4���ؽᾧ�õ���ĸҺ�������´��ؽᾧʱ�ظ�ʹ�ã����ﵽһ����������뾻��������ԭ������ ����������������������
��5����ͼ��̼����3�ֲ�ͬ�����еĸ�ʴ����ʵ������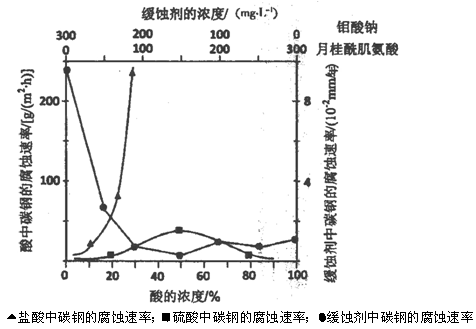
��̼��������������и�ʴ���������Ũ�ȱ仯�����Բ��죬��ԭ��������� ����________________��
�ڿ����������ζ�̼�ֵĻ�ʴԭ�����ڸ��������γ�FeMoO4��Fe2O3����Ĥ��
�ܱ�ʽѭ����ȴˮϵͳ�е�̼�ֹܵ���ʴ���������������������NaNO2����NaNO2��������������_________________________________________��
�������ͼ������ơ��¹��ἡ������Ũ��Ϊ300mg��L��1����ʴЧ�����ʱ�����Ƶ����ʵ���Ũ��Ϊ������ ����
��6��﮺Ͷ������γɵĶ��ε�ص��ܷ�ӦΪ��xLi + nMoS2 Lix(MoS2)n�����طŵ�ʱ��������Ӧʽ�ǣ�___________________________________��
Lix(MoS2)n�����طŵ�ʱ��������Ӧʽ�ǣ�___________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ڣ�Te��Ϊ��A��Ԫ�أ��ǵ�����¼����²��ϵ���Ҫ�ɷ�֮һ����ҵ�Ͽɴӵ�⾫��ͭ������������ȡ�ڡ�
��1����ͭ�к���Cu������Zn��Ag��Au��TeO2�������������⾫��������������Ҫ����TeO2�������������ʼ������������⾫����ͭʱ�������缫��ӦʽΪ ��
��2��TeO2���������������ˮ��������ǿ���ǿ�����������������ȡ�ڵ�һ�ֹ����������£�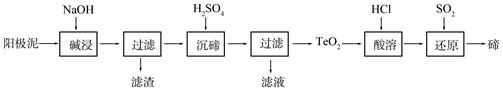
�١������ʱTeO2������Ӧ�Ļ�ѧ����ʽΪ ��
�ڡ����ڡ�ʱ������Һ��pHΪ4.5-5.0������TeO2���������H2SO4��������Һ��ȹ��������ڵij�������ȫ��ԭ���� ����ֹ�ֲ���ȹ���IJ��������� ��
�ۡ����ܡ���SO2ͨ��TeCl4��Һ�н��С���ԭ���õ��ڣ��÷�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�뽫5�����ʣ�N2O��FeCl2��Fe(NO3)3��HNO3��FeCl3�ֱ����������Ӧ�ĺ����ϣ����һ��δ��ƽ�Ļ�ѧ����ʽ��
��1�� + �� + + +H2O��������ƽ��
��2����Ӧ�����У�N2O��FeCl2�����ʵ���֮��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪�������ԣ�KMnO4��HNO3��Biλ�����ڱ��Т�A�壬��3�۽��ȶ���Bi2O3Ϊ���������Bi3������ҺΪ��ɫ��ȡһ���������ữ��Mn(NO3)2��Һ���ν�������ʵ�飬�����¼���£�
�������м���������NaBiO3����Һ��Ϊ�Ϻ�ɫ���ڼ����μ�����H2O2���Ϻ�ɫ��ȥ���������ݲ��������ټ���������PbO2���壬�����ܽ⣬��Һ�ֱ�Ϊ�Ϻ�ɫ��
�ش��������⣺
(1)д��ʵ��ٷ�Ӧ�����ӷ���ʽ�� ��
(2)KMnO4��H2O2��PbO2��������ǿ������˳��Ϊ ��
(3)��Ӧ�۵õ�����Һ��ͨ��SO2���壬������������ ��
(4)��ʵ��ڷų���336 mL����(��״��)����Ӧ�ٱ�������Mn(NO3)2Ϊ mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������ʡ���������ƵƵ������Ϊ��ֹ�ڴ���֮���߲����У�������Ҫ�����ĸ�����������Ư���ȡ�
(1)����������Ŀǰ�����Ϲ��ϵĵ��Ĵ���Ч�����Ĺ�����������������KClO3��SO2��H2SO4�����·�Ӧ�Ƶá���д����Ӧ�����ӷ���ʽ�� ��
(2)��̼������һ������̬Ư������ѧʽ�ɱ�ʾΪNa2CO3��3H2O2��������Na2CO3��H2O2��˫�����ʡ���̼�������������ʾ��ᷢ����ѧ��Ӧ��ʧЧ�����й�̼����ֻ������������Ӧ���� ��
| A��MnO2 | B��KMnO4��Һ | C��ϡ���� | D��Na2SO3��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��Ӧ�з�Ӧ�����������У�AsH3��H2SO4��KBrO3��K2SO4��H3AsO4��H2O��һ��δ֪����X��
(1)��֪KBrO3�ڷ�Ӧ�еõ����ӣ���÷�Ӧ�Ļ�ԭ���� ��
(2)��֪0.2 mol KBrO3�ڷ�Ӧ�еõ�1 mol��������X����X�Ļ�ѧʽΪ ��
(3)����������Ӧ����֪
a�������ԣ�KBrO3��H3AsO4
b�������ԣ�H3AsO4��KBrO3
c����ԭ�ԣ�AsH3��X
d����ԭ�ԣ�X��AsH3
(4)���������ͻ�ԭ���Ļ�ѧʽ������ƽ��ļ������������з����У����������ת�Ƶķ������Ŀ��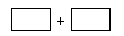
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ұ���봦�����漰������ԭ��Ӧ��
��1������������ұ����Ӧ����ʱ���õ�ⷨ����________��
a��Fe2O3 b��NaCl c��Cu2S d��Al2O3
��2����ͭ��(Cu2S)�ɷ�����Ӧ��2Cu2S��2H2SO4��5O2=4CuSO4��2H2O���÷�Ӧ�Ļ�ԭ����________����1 mol O2������Ӧʱ����ԭ����ʧ���ӵ����ʵ���Ϊ________mol����CuSO4��Һ�м���þ��ʱ���������ɣ���������________��
��3����ͼΪ��⾫������ʾ��ͼ��________(�a����b��)��Ϊ�������ʵĴ�������b������������ɫ��������������ɸ�����ĵ缫��ӦʽΪ__________________________��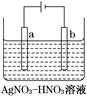
��4��Ϊ������������ĺڰ�(Ag2S)�����������������������ʳ��ˮ�в������Ӵ���Ag2Sת��ΪAg��ʳ��ˮ��������______________________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com