
����Ŀ��I. ��һ�������£����淴ӦA��B![]() mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯�����
mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯�����
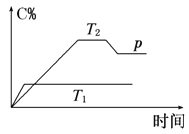
��1���¶�T1________T2(����ڡ������ڡ���С�ڡ�)��
��2������Ӧ��________��Ӧ(����ȡ����ȡ�)��
��3�����A��B��C��Ϊ���壬��m________2(����ڡ������ڡ���С�ڡ�)��
��4�����¶Ⱥ��ݻ�����ʱ������ƽ����ϵ�м���һ������ijϡ�����壬����ϵ��ѹǿ________(�������С�����䡱)��ƽ��_________________�ƶ�(�������Ӧ�������淴Ӧ������)��
II. CO�������ʻ���ѧƷ�Ļ���ԭ�ϣ� 850��ʱ���ں����ܱ�������ͨ��CO��H2O(g)��������CO(g)��H2O(g) ![]() H2(g)��CO2(g)
H2(g)��CO2(g) ![]() H��0���ⶨŨ����ʱ���ϵ���±���
H��0���ⶨŨ����ʱ���ϵ���±���
t/min | c(CO)/ mol��L��1 | c(H2O)/ mol��L��1 |
0 | 0.30 | 0.20 |
2 | \ | 0.10 |
3 | 0.18 | \ |
4 | \ | 0.08 |
�ش���������
��1��t=3minʱ����(��)____________��(��)��ѡ���������������������������
��2��0��2min��CO��ƽ����Ӧ����Ϊ ____________��
��3��������Ӧ�е�COƽ��ת����Ϊ____________��
��4����һ�������£��������з�Ӧ��CO(g)��H2O(g) ![]() H2(g)��CO2(g)�����淴Ӧ��������ʱ��仯������ͼ��ʾ����t1ʱ�ı��ij�ַ�Ӧ����������_____������ţ���
H2(g)��CO2(g)�����淴Ӧ��������ʱ��仯������ͼ��ʾ����t1ʱ�ı��ij�ַ�Ӧ����������_____������ţ���

a������COŨ��
b����ѹ
c������ʱ�������
d��ʹ�ô���
���𰸡����� ���� ���� ���� �� �� ![]() 40% bd
40% bd
��������
I.��1)�¶�Խ�߷�Ӧ����Խ�죬����ƽ���ʱ��Խ�̣�
(2)��ͼ��֪�¶�T1��T2���¶�Խ��C%ԽС���������¶�ƽ�����淴Ӧ�ƶ���
(3)��ͼ��֪��T2�¶�ʱ�ﵽƽ�����������ѹ��C%��С��������ѹǿƽ�����淴Ӧ�ƶ���
(4)���¶Ⱥ��ݻ�����ʱ����ƽ����ϵ�м���һ������ijϡ�����壬��ϵѹǿ������Ӧ������Ũ�Ȳ��䣬ƽ�ⲻ�ƶ���
II.(1)(2)(3)��������ʽ���м��㣬���Եó�3minĩc(H2O)��˵����ʱ�Ѿ��ﵽ��ѧƽ�⣬���û�ѧ��Ӧ���ʵļ��㹫ʽ�������CO�ķ�Ӧ���ʺ�![]() �����������CO��ת���ʣ����(4)���ͼʾ��Ϣ����������ԭ�����н��
�����������CO��ת���ʣ����(4)���ͼʾ��Ϣ����������ԭ�����н��
I.(1)��ͼ��֪���¶�ΪT1�ȵ���ƽ�⣬�¶�Խ�߷�Ӧ����Խ�죬����ƽ���ʱ��Խ�̣���T1��T2���ʴ�Ϊ�����ڣ�
(2)��ͼ��֪�¶�T1��T2���¶�Խ��C%ԽС���������¶�ƽ�����淴Ӧ�ƶ���������ӦΪ���ȷ�Ӧ���ʴ�Ϊ�����ȣ�
(3)��ͼ��֪��T2�¶�ʱ�ﵽƽ�����������ѹ��C%��С��������ѹǿƽ�����淴Ӧ�ƶ�������ѹǿƽ�����������ʵ�����С�ķ����ƶ���˵���÷�Ӧ�淴Ӧ�������������ʵ�����С�ķ���m��2����Ϊ�����ڣ�
(4)���¶Ⱥ��ݻ�����ʱ����ƽ����ϵ�м���һ������ijϡ�����壬����������ʵ���������ϵѹǿ������Ӧ������Ũ�Ȳ��䣬�����淴Ӧ���ʲ��䣬ƽ�ⲻ�ƶ����ʴ�Ϊ��������
II. (1)��������ʽ���㣬�ܹ��ܿ�����3minĩ��c(H2O)=0.08 mol��L��1��4minĩһ����˵��t=3minʱ��Ӧ�Ѿ��ﵽ��ѧƽ�⣬��(��) = ��(��)���ʴ�Ϊ������
(2)���ݷ�Ӧ����ʽ��֪��0��2min CO�����ʵ���Ũ�ȱ仯��0.1 mol��L��1����ƽ����Ӧ����Ϊ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
(3)�ӱ������ݿ�֪���ﵽƽ��ʱ��CO��ƽ��Ũ��Ϊ0.18 mol��L��1����������Ӧ�е�COƽ��ת����Ϊ��![]() ���ʴ�Ϊ��40%
���ʴ�Ϊ��40%
(4)��ͼ�п�֪���ı��������淴Ӧ����ͻȻ�����������ƽ�ⲻ�ƶ���
a������COŨ�ȣ�����Ӧ����ͻȻ�����淴Ӧ����������������䣬������ͼʾ��Ϣ��a�������⣻
b������CO(g)��H2O(g)![]() H2(g)��CO2(g)��Ӧǰ����������ʵ������ֲ��䣬�ʼ�ѹʹ�������淴Ӧ����ͬ�ȷ��ȵ�ͻȻ����ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��b�������⣻
H2(g)��CO2(g)��Ӧǰ����������ʵ������ֲ��䣬�ʼ�ѹʹ�������淴Ӧ����ͬ�ȷ��ȵ�ͻȻ����ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��b�������⣻
c������ʱ�����������ϵ��ѹǿ�����Ƿ�Ӧ�����������Ũ�Ⱦ�δ�ı䣬�������淴Ӧ���ʾ����䣬������ͼʾ��Ϣ��c�������⣻
d��ʹ�ô����ܹ�ͬ�ȷ��ȵ�ͻȻ���������淴Ӧ���ʣ�ƽ�ⲻ�ƶ�������ͼʾ��Ϣ��d�������⣻
�ʴ�Ϊ��bd��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ڸ��������£�����ѡ����ʾ�����ʼ�ת������ʵ�ֵ���
A.H2SiO3![]() SiO2
SiO2 ![]() SiCl4
SiCl4
B.Cu![]() Cu(NO3)2(aq)
Cu(NO3)2(aq) ![]() Cu(NO3)2(s)
Cu(NO3)2(s)
C.ClCH2��CH2Cl![]() HOCH2��CH2OH
HOCH2��CH2OH ![]() HOOC��COOH
HOOC��COOH
D.Al![]() Al2O3
Al2O3![]() NaAlO2(aq)
NaAlO2(aq)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������������Чɱ���¹ڲ�����2019-nCoV����ijѧϰС�������Ҫ���Ʊ�Ũ�Ȳ�С��0.8mol/L�Ĵ�������Һ��װ�ü���ͼ��
����1�����³�ѹ�£�Cl2OΪ�ػ�ɫ���壬�е�Ϊ3.8����42�����ϻ�ֽ�����Cl2��O2��Cl2O������ˮ����ˮ������Ӧ����HClO��
����2���������Ϳ���(�����뷴Ӧ)������� 1��3���ͨ�뺬ˮ8%��̼��������Cl2O������ˮ����Cl2O(����Cl2)�Ƶô�������Һ��
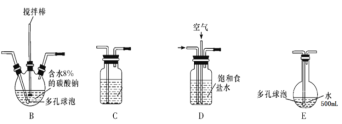
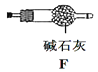
��1���缫YӦ�����ӵ�Դ��___������������������������,װ��C��Ӧʢ��_________����װ�õ�����˳��ΪA��________________________________
��2����Ӧ�����У�װ��B�������ˮ�У���Ŀ����_____________________________����֪װ��B���ﺬ��һ����ʽ�Σ�������Ӧ�Ļ�ѧ����ʽΪ___________________��
��3��ʵ���п����������������ȵķ�����_______________________________
��4��װ��E��ʹ����ɫƽ����ƿ��ԭ���ǣ��û�ѧ����ʽ��ʾ��________________
��5���˷��������������ֱ������ˮ�Ʊ���������Һ���ŵ��ǣ����һ�����ɣ�_____��
��6����װ��B�����ɵ�Cl2O������20%������Eǰ��װ���У����������װ��E��ˮ�У�װ��E����500mL��������ҺŨ��Ϊ0.8mol/L,��������Ҫ��ˮ8%��̼���Ƶ�����Ϊ___g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
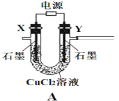
����Ŀ��(1)�����������ž�����ҵ�Ŀ��ٷ�չ�����������������Ȼ���IJ�����Ҳ��֮Ѹ����������ˣ����Ȼ���ת��Ϊ�����ļ�����Ϊ��ѧ�о����ȵ㡣�ش��������⣺Deaconֱ���������ɰ����д����̽��У�
CuCl2(s)=CuCl(s)+![]() Cl2(g) ��H1=83 kJ��mol 1
Cl2(g) ��H1=83 kJ��mol 1
CuCl(s)+![]() O2(g)=CuO(s)+
O2(g)=CuO(s)+![]() Cl2(g) ��H2= 20 kJ��mol 1
Cl2(g) ��H2= 20 kJ��mol 1
CuO(s)+2HCl(g)=CuCl2(s)+H2O(g) ��H3= 121 kJ��mol 1
��4HCl(g)+O2(g)=2Cl2(g)+2H2O(g)����H=_________ kJ��mol 1������O2�ĵ���ʽΪ__________
(2)�����HCl��300��ʱ��Ӧ����1mol SiHCl3�����H2 ���ų�225KJ�������÷�Ӧ���Ȼ�ѧ����ʽΪ________________________��SiHCl3�к��еĻ�ѧ������Ϊ__________
(3)��SiCl4�⻯ΪSiHCl3�����ַ�������Ӧ�ķ�Ӧ����Ϊ��
��SiCl4(g)+H2(g)![]() SiHCl3(g)+HCl(g) ��H1��0
SiHCl3(g)+HCl(g) ��H1��0
��3SiCl4(g)+2H2(g)+Si(s)![]() 4SiHCl3(g) ��H2��0
4SiHCl3(g) ��H2��0
��2SiCl4(g)+H2(g)+Si(s)+HCl(g)![]() 3SiHCl3(g) ��H3
3SiHCl3(g) ��H3
��Ӧ�۵���H3______(����H1����H2��ʾ)��
(4)����������Ŀǰ�����Ϲ��ϵĵ��Ĵ���Ч�����Ĺ���������,������KClO3��H2SO4��������Na2SO3��Ӧ�Ƶ�.��д����Ӧ�����ӷ���ʽ________________________
(5)�Ȼ�麟��������ӣ��磺�ں���ͭ��ʱ���Ȼ�麟�ȥͭ�����������ͭ�Ա㺸�ӣ��䷴ӦΪ��_______CuO+______NH4Cl![]() ______Cu+______CuCl2+______N2��+______H2O
______Cu+______CuCl2+______N2��+______H2O
����ƽ��������ԭ��Ӧ����ʽ___________________________
�ڴ˷�Ӧ��������0.2mol�����壬����__________mol�ĵ���ת�ƣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����NA��ʾ�����ӵ�������ֵ������˵����ȷ����
A. 0.5mol�ۻ�(As4S4)���ṹ����ͼ������NA��S-S��![]()
B. ��1molNH4NO3��������ϡ��ˮ�У�������Һ�����ԣ�����Һ��NH4+����ĿΪNA
C. ��״���£�33.6L���ȼ����к�����ԭ�ӵ���ĿΪ3NA
D. �����£�16.8gFe������ˮ������ȫ��Ӧ��ת�Ƶĵ�����Ϊ0.6NA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����(N)����(P)����(As)�ȵ�VA��Ԫ�صĻ��������о�������������Ҫ��;����ش��������⣺
��1����̬��ԭ�ӵļ۵����Ų�ͼΪ________________����������Ԫ�ص�ԭ���У���һ�����ܴ�����ԭ�ӵ���________________�֡�
��2������Ԫ���Ȼ���RH3(NH3��PH3��AsH3)��ij��������R�ĺ˵�����ı仯������ͼ��ʾ����Y��ɱ�ʾ���⻯��(RH3)���ʿ�����________________(�����)��
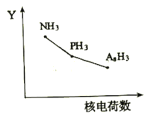
A.�ȶ���B.�е�ʱC.R��H��֮��ļ���D.���Ӽ�������E.��ԭ��
��3�����ⶨ���֣�ij��N2O5������NO2����NO3������������ɣ���������Nԭ�ӵ��ӻ���ʽ��________________�ӻ��������ӵĿռ乹��Ϊ________________��
��4���Ҷ���(H2N��CH2��CH2��NH2)��CuCl2��Һ���γ�������(�ṹ��ͼ)��
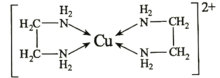
Cu2������λ��Ϊ________________��1mol���������к�����Ϊ________________mol���Ҷ��������װ�[N(CH3)3]�����ڰ������Ҷ��������װ��ķе�ߵö࣬��ԭ����________________��
��5��ͨ����ΪCu3N�����Ӿ��壬�侧���ܿ�ͨ��ͼ1��Born��Haberѭ������õ���ͨ��ͼ1������________________(��ܡ����ܡ�)�����Cuԭ�ӵĵ�һ�����ܣ�Cu3N������Ϊ________________kJ��mol��1��
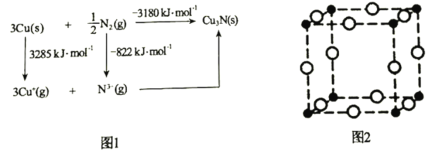
��6��Cu3N������ͼ2��ʾ��Cu���İ뾶Ϊapm��N3���İ뾶Ϊbpm��Cu3N�������ܶ�Ϊ________________g��cm��3(�г�����ʽ���ɣ������ӵ�������ֵ��NA��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����з�Ӧ�����ڼӳɷ�Ӧ����(����)
A. CH4��Cl2![]() CH3Cl��HCl
CH3Cl��HCl
B. CH2===CH2��HCl�D��CH3CH2Cl
C. CH3CH2OH��HBr![]() CH3CH2Br��H2O
CH3CH2Br��H2O
D. 2CH3CH3��7O2![]() 4CO2��6H2O
4CO2��6H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ���¶�����һ��2 L�ĺ����ܱ������з�����Ӧ4A(s)+3B(g)2C(g)+D(g)����2 min��ƽ��״̬����ʱB��Ӧ������0.9 mol������˵����ȷ����
A. ƽ��ʱ��v(A)��v(B)��v(C)��v(D) =4��3��2��1
B. �������ƽ����Է�����������Ϊƽ���־
C. �����������ʹѹǿ����ɼӿ췴Ӧ����
D. C��ƽ����Ӧ����Ϊ0.5 mol/(L��min)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2019�����Ŵ�ѧ��Ұ���ڿ������ںϳ���(��Ҫ�ɷ�CO��H2)��ת�����Ҵ�����ʵʩ�ɿؽ�����������:�ϳ������״����������Ҵ��ķ�ʽ���ɹ�ʵ���Ҵ��ĸ�ѡ���Ժϳɡ�
��1����֪����H2(g)+![]() O2(g)=H2O(g) ��H1=-285.8KJ/mol����CO(g)+
O2(g)=H2O(g) ��H1=-285.8KJ/mol����CO(g)+![]() O2(g)=CO2(g) ��H2=-283KJ/mol����CH3OH(g)+
O2(g)=CO2(g) ��H2=-283KJ/mol����CH3OH(g)+![]() O2(g)= CO2(g)+2 H2O(l) ��H3=-764.6 KJ/mol��
O2(g)= CO2(g)+2 H2O(l) ��H3=-764.6 KJ/mol��
��ҵ�����úϳ����Ʊ��״��Ŀ��淴Ӧ�Ȼ�ѧ����ʽΪ______________________��
��2�����º��������£�������˵���ϳ����Ƽ״��ķ�Ӧ�Ѵ�ƽ��״̬����_______��
A����λʱ��������n mol CO��ͬʱ����2n mol H2
B�� v(H2)��= 2v(CH3OH)��
C��������������ܶȱ��ֲ���
D�������������ƽ��Ħ���������ֲ���
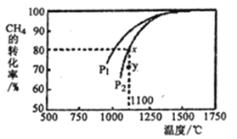
��3��������Ȼ����ȡ�ϳ�����ԭ��Ϊ: CO2(g)+CH4(g)= 2CO(g)+ 2H2(g)�����ܱ�������ͨ�����ʵ���Ũ�Ⱦ�Ϊ0.1 molL-1��CH4��CO2����һ�������·�����Ӧ�����CH4��ƽ��ת�������¶ȼ�ѹǿ�Ĺ�ϵ����ͼ��ʾ����ѹǿP1______P2 (��������������С����)��ѹǿΪP2ʱ����Y�㣺v(��)_____v(��) (��������������С��������������)����Y���Ӧ�¶��µĸ÷�Ӧ��ƽ�ⳣ��K=________ (������������λ��Ч����)��
��4���Զ������ѱ��渲��CuAlO4Ϊ���������Խ�CH4��CO2ֱ��ת�������ᡣ
���ڲ�ͬ�¶��´����Ĵ�Ч���������������������ͼ��ʾ��250-300��Cʱ���¶�������������������ʽ��͵�ԭ����____��
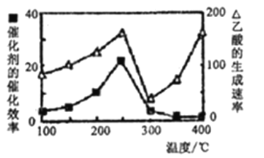
��Ϊ����߸÷�Ӧ��CH4��ת���ʣ����Բ�ȡ�Ĵ�ʩ��_____________________��
��5��һ�������£��״���һ����̼��Ӧ���Ժϳ����ᡣ���������£���a mol/L��CH3COOH��Һ��b mol/ Ba(OH)2��Һ�������ϣ���Ӧƽ��ʱ��2c(Ba2+)=c(CH3COO-)���ú�a��b�Ĵ���ʽ��ʾ���û����Һ�д���ĵ��볣��Ϊ________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com