
��18�֣���Ҫ��ش���������
��1���ñ�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ�÷�̪��ָʾ�������в����лᵼ��ʵ����ƫ�͵��� (����� )
�ټ�ʽ�ζ���������ˮϴ����û���ñ�Һ��ϴ
������ʽ�ζ��ܼӴ���Һʱ����������ˮϴ����ĵζ���δ�ô���Һ��ϴ
����ƿ������ˮϴ����û���ô���Һ��ϴ
�ܵζ�ǰ�ζ��ܼ�������ݣ��ζ���������ʧ
���յ����ʱ���ӣ���������������ȷ
��2��ij����С��ͬѧ����ͼ��װ�ý���ʵ�飬�Դ��������⣺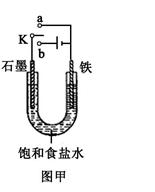
������ʼʱ����K��a���ӣ����������绯ѧ��ʴ�е� ��ʴ��
������ʼʱ����K��b���ӣ����ܷ�Ӧ�����ӷ���ʽΪ ��
��3����֪��Ǧ�����ܵĻ�ѧ����ʽΪ��Pb+PbO2 +2H2SO4 2PbSO4+2H2O
2PbSO4+2H2O
��Ǧ�����ڷŵ�ʱ������ӦΪ ��
��Ǧ�����ڳ��ʱ������ӦΪ ��
�����Ǧ�����ڷŵ�ʱ��·����2mol����ת��ʱ������H2SO4 mol��
��4��������,���ȡ0.1mol/L HA��Һ��0.1mol/L NaOH��Һ��������(���Ի�Ϻ���Һ����ı仯),��û����Һ��pH=8��������Һ��������ʽ�ľ�ȷ����������������֣���
c(OH��)��c(HA)�� ___________ mol/L��
��5����Cl����Al3����HSO4����K����HS�����������У�ֻ��ˮ�ⲻ�ܵ���������� ��ֻ�ܵ��벻��ˮ��������� �����ܵ�������ˮ��������� ��д����ˮ�����ӵ�ˮ�����ӷ���ʽ �� ��
��6����֪25��ʱ��Mg(OH)2���ܶȻ�����Ksp = 5.6��10��12�����ij��Һ��pH = 13������¶�����Һ�е�c(Mg2+) = ____________________��
��1���ڢݣ�2�֣�������2����������2�֣� ��2Cl-+2H2O H2��+C12��+2OH-��2�֣�
H2��+C12��+2OH-��2�֣�
��3��Pb��SO42����2e��=PbSO4��2�֣�PbSO4��2H2O��2e��=PbO2��SO42����4H����2�֣���2��1�֣�
��4��10��8������1�֣���5��Al3��,��1�֣���HSO4����1�֣� HS����1�֣�
Al3+ +3 H2O Al(OH)3 +3H+��1�֣� HS�� + H2O
Al(OH)3 +3H+��1�֣� HS�� + H2O H2S + OH-��1�֣�
H2S + OH-��1�֣�
��6�� 5.6��10-10mol��L��1��1�֣�
����


 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��B��C��D�����ɶ�����Ԫ����ɵij������ʣ�����A��B��C����ͬһ��Ԫ�أ���һ���������ת����ϵ��ͼ��ʾ�����ֲ�������ȥ����
A��B��C��D�����ɶ�����Ԫ����ɵij������ʣ�����A��B��C����ͬһ��Ԫ�أ���һ���������ת����ϵ��ͼ��ʾ�����ֲ�������ȥ����| ���� |
| �� |
| ���� |
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 H++CN-��H2O
H++CN-��H2O H++OH-��CN-+H2O
H++OH-��CN-+H2O HCN+OH-
HCN+OH- H++CN-��H2O
H++CN-��H2O H++OH-��CN-+H2O
H++OH-��CN-+H2O HCN+OH-
HCN+OH-�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
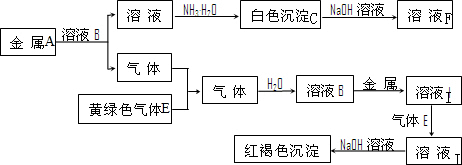
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com