
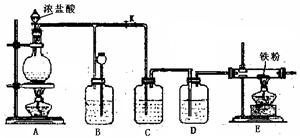
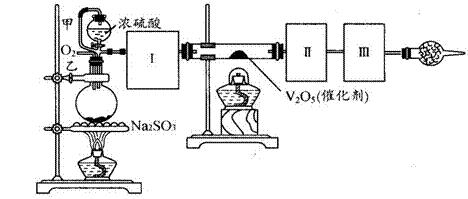
 ��4��E����Ӧ��Ϻر�����K����ȥ�ƾ��ƣ��������ȵ����ã�A����������Cl2��������ʱB�е������� ��B�������� ��
��4��E����Ӧ��Ϻر�����K����ȥ�ƾ��ƣ��������ȵ����ã�A����������Cl2��������ʱB�е������� ��B�������� �� ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
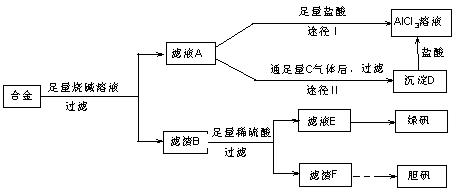
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

| �¶�/�� | 0 | 10 | 20 | 30 | 50 | 80 | 100 |
| �ܽ��(g/100gH20) | 74.4 | 81.9 | 91.8 | 106.8 | 315.1 | 525.8 | 535.7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
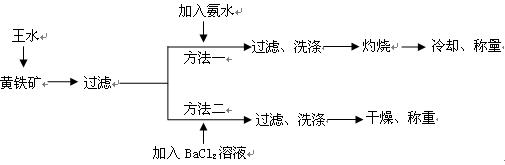
 ��(��=1.42g��cm-3)��3�����Ũ����(��=1.19g��cm-3)��϶��ɵġ�
��(��=1.42g��cm-3)��3�����Ũ����(��=1.19g��cm-3)��϶��ɵġ�| A��BaCl2 | B��NaOH | C��Na2SO4 | D��HCl |
�鿴�𰸺ͽ���>>
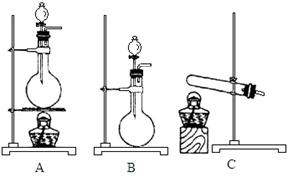
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
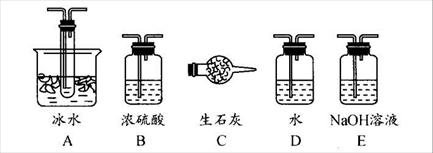
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����ȵIJ�˿��NH3��O2������Ӵ�����˿���ֺ��ȣ�˵������������Ӧ�Ƿ��ȷ�Ӧ |
| B��ij��ˮ��pHС��5.6��˵����ˮ��һ���ܽ���SO2 |
C��ij��Һ�м����Ȼ�����Һ�����ɰ�ɫ������˵��ԭ��Һ��һ������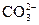 |
| D����þ������Ƭ�õ��������ٲ��ϡNaOH��Һ��þ���ϲ������ݣ�˵��þ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
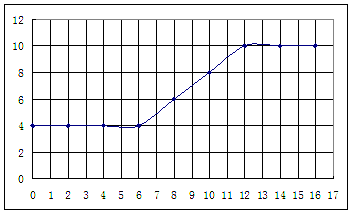
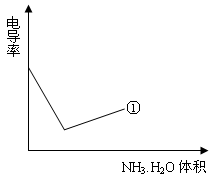
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����ó���ʯ��ˮ����Na2CO3��Һ��NaHCO3��Һ |
| B�����÷�Һ�ķ���������������Ȼ�̼��Һ�з���������� |
| C��Ϊȷ�ⶨ������NaOH��Һ��Ӧ���к��ȣ�������ͼ�����ʵ������ |
| D������ˮ�������顢���Ȼ�̼���Ҵ�������ɫҺ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����������Գ�ȥ�������е�����̼�������� |
| B���ýྻ�IJ�����պȡ��Һ������ʪ���pH��ֽ�ϲⶨ��pH |
| C����2.0mLŨ�Ⱦ�Ϊ0.1mol��L-1��KCl��KI�����Һ�еμ�1~2��0.01mol��L-1 AgNO3��Һ���������ʻ�ɫ��˵��AgCl��Ksp��AgI��Ksp�� |
| D������25.00ml��ʽ�ζ�����ȡ20.00ml ��ˮ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com