
| ±ąŗÅ | ŹµŃé²Ł×÷ | ŹµŃéĻÖĻóÓė½įĀŪ |
| ¢Ł | ȔɣĮæ·ŪÄ©·ÅČėŹŌ¹ÜÖŠ£¬×¢Čė1mol/LµÄĮņĖį | ·ŪÄ©Öš½„Čܽā£¬ČÜŅŗ³Ź»ĘĀĢÉ« |
| ¢Ś | ½«”°¢Ł”±ÖŠĖłµĆČÜŅŗ·Ö³ÉĮ½·Ż£»ĻņĘäÖŠŅ»·ŻµĪ¼Ó¼øµĪKSCNČÜŅŗ£¬Õńµ“ | ČōČÜŅŗ±äĪŖ ŃŖŗģÉ« ŃŖŗģÉ« £¬ĖµĆ÷ÓŠFe2O3“ęŌŚ |
| ¢Ū | ŌŚĮķŅ»·ŻÖŠ¼ÓČėÉŁĮæKMnO4ČÜŅŗ | ČōČÜŅŗ ×ĻÉ«ĶŹČ„ ×ĻÉ«ĶŹČ„ £¬ĖµĆ÷ÓŠFeO“ęŌŚ |
| ÅØĮņĖį |
| ”÷ |
| a+2b |
| a+3b |
| 4 |
| 5 |
| a+2b |
| a+3b |
| 4 |
| 5 |


 ŗ£µķ»ĘøŌĆūŹ¦µ¼ŗ½ĻµĮŠ“š°ø
ŗ£µķ»ĘøŌĆūŹ¦µ¼ŗ½ĻµĮŠ“š°ø ĘÕĶØøßÖŠĶ¬²½Į·Ļ°²įĻµĮŠ“š°ø
ĘÕĶØøßÖŠĶ¬²½Į·Ļ°²įĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
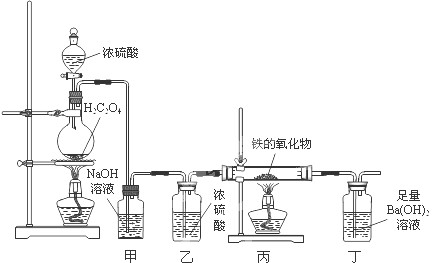
ijĢśµÄŃõ»ÆĪļ·ŪÄ©æÉÄÜŗ¬ÓŠFeO”¢Fe2O3ÖŠµÄŅ»ÖÖ»ņĮ½ÖÖ”£Ä³ŠĖȤŠ”×éĶ¬Ń§ĄūÓĆŅŌĻĀ×°ÖĆ²ā¶ØĢśµÄŃõ»ÆĪļµÄ×é³É£¬Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£ØŅŃÖŖ £©
£©
£Ø1£©×°ÖĆ¼×µÄ×÷ÓĆŹĒ ”£
£Ø2£©×°Öƶ”ÖŠµÄŹµŃéĻÖĻóŹĒ ”£
£Ø3£©Čō±ūÖŠĢśµÄŃõ»ÆĪļµÄ»ÆѧŹ½ĪŖFexOy£¬Ōņ±ūÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£Čō±ūÖŠĢśµÄŃõ»ÆĪļÖŹĮæĪŖ3.92g£¬ĒŅĶźČ«·“Ó¦£¬¶”ÖŠÉś³É³ĮµķµÄÖŹĮæĪŖ13.79g£¬ŹŌČ·¶ØøĆŃõ»ÆĪļÖŠx : y= £¬øĆ¹ĢĢå·ŪÄ©µÄ×é³ÉĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğ½ĖÕŹ”Ńļ֯֊ѧø߶žÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌĢā£Ø±ŲŠŽ£© ĢāŠĶ£ŗĢīæÕĢā
ijĢśµÄŃõ»ÆĪļ·ŪÄ©æÉÄÜŗ¬ÓŠFeO”¢Fe2O3ÖŠµÄŅ»ÖÖ»ņĮ½ÖÖ”£Ä³ŠĖȤŠ”×éĶ¬Ń§ĄūÓĆŅŌĻĀ×°ÖĆ²ā¶ØĢśµÄŃõ»ÆĪļµÄ×é³É£¬Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£ØŅŃÖŖ £©
£©
£Ø1£©×°ÖĆ¼×µÄ×÷ÓĆŹĒ ”£
£Ø2£©×°Öƶ”ÖŠµÄŹµŃéĻÖĻóŹĒ ”£
£Ø3£©Čō±ūÖŠĢś µÄŃõ»ÆĪļµÄ»ÆѧŹ½ĪŖFexOy£¬Ōņ±ūÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£Čō±ūÖŠĢśµÄŃõ»ÆĪļÖŹĮæĪŖ3.92g£¬ĒŅĶźČ«·“Ó¦£¬¶”ÖŠÉś³É³ĮµķµÄÖŹĮæĪŖ13.79g£¬ŹŌČ·¶ØøĆŃõ»ÆĪļÖŠx : y= £¬øĆ¹ĢĢå·ŪÄ©µÄ×é³ÉĪŖ ”£
µÄŃõ»ÆĪļµÄ»ÆѧŹ½ĪŖFexOy£¬Ōņ±ūÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£Čō±ūÖŠĢśµÄŃõ»ÆĪļÖŹĮæĪŖ3.92g£¬ĒŅĶźČ«·“Ó¦£¬¶”ÖŠÉś³É³ĮµķµÄÖŹĮæĪŖ13.79g£¬ŹŌČ·¶ØøĆŃõ»ÆĪļÖŠx : y= £¬øĆ¹ĢĢå·ŪÄ©µÄ×é³ÉĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģ½ĖÕŹ”ø߶žÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌĢā£Ø±ŲŠŽ£© ĢāŠĶ£ŗĢīæÕĢā
ijĢśµÄŃõ»ÆĪļ·ŪÄ©æÉÄÜŗ¬ÓŠFeO”¢Fe2O3ÖŠµÄŅ»ÖÖ»ņĮ½ÖÖ”£Ä³ŠĖȤŠ”×éĶ¬Ń§ĄūÓĆŅŌĻĀ×°ÖĆ²ā¶ØĢśµÄŃõ»ÆĪļµÄ×é³É£¬Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£ØŅŃÖŖ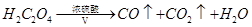 £©
£©
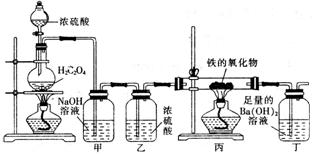
£Ø1£©×°ÖĆ¼×µÄ×÷ÓĆŹĒ ”£
£Ø2£©×°Öƶ”ÖŠµÄŹµŃéĻÖĻóŹĒ ”£
£Ø3£©Čō±ūÖŠĢśµÄŃõ»ÆĪļµÄ»ÆѧŹ½ĪŖFexOy£¬Ōņ±ūÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£Čō±ūÖŠĢśµÄŃõ»ÆĪļÖŹĮæĪŖ3.92g£¬ĒŅĶźČ«·“Ó¦£¬¶”ÖŠÉś³É³ĮµķµÄÖŹĮæĪŖ13.79g£¬ŹŌČ·¶ØøĆŃõ»ÆĪļÖŠx : y= £¬øĆ¹ĢĢå·ŪÄ©µÄ×é³ÉĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
| ±ąŗÅ | ŹµŃé²Ł×÷ | ŹµŃéĻÖĻóÓė½įĀŪ |
| ¢Ł | ȔɣĮæ·ŪÄ©·ÅČėŹŌ¹ÜÖŠ£¬×¢Čė1mol/LµÄĮņĖį | ·ŪÄ©Öš½„Čܽā£¬ČÜŅŗ³Ź»ĘĀĢÉ« |
| ¢Ś | ½«”°¢Ł”±ÖŠĖłµĆČÜŅŗ·Ö³ÉĮ½·Ż£»ĻņĘäÖŠŅ»·ŻµĪ¼Ó¼øµĪKSCNČÜŅŗ£¬Õńµ“ | ČōČÜŅŗ±äĪŖ______£¬ĖµĆ÷ÓŠFe2O3“ęŌŚ |
| ¢Ū | ŌŚĮķŅ»·ŻÖŠ¼ÓČėÉŁĮæKMnO4ČÜŅŗ | ČōČÜŅŗ______£¬ĖµĆ÷ÓŠFeO“ęŌŚ |
 CO”ü+CO2”ü+H2O£¬²¢ÄāÓĆĻĀĮŠ×°ÖĆ½ųŠŠ¶ØĮæĢ½¾æ£®
CO”ü+CO2”ü+H2O£¬²¢ÄāÓĆĻĀĮŠ×°ÖĆ½ųŠŠ¶ØĮæĢ½¾æ£®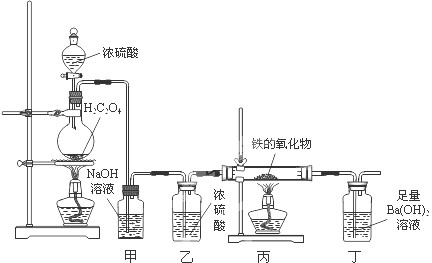
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com