
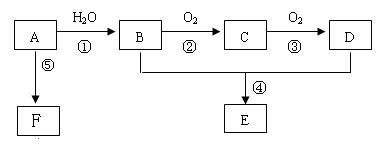
 �� ��
�� �� ��
��  _________
_________ ����ط������������
����ط������������  __�������ƣ���
__�������ƣ��� OH
OH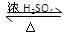 CH3COOCH2CH3 + H2O ��2�֣� ������Ӧ��ȡ����Ӧ��1�֣� ��5������Na2CO3��Һ ��
CH3COOCH2CH3 + H2O ��2�֣� ������Ӧ��ȡ����Ӧ��1�֣� ��5������Na2CO3��Һ �� Һ ��ÿ�ո�1�֣�
Һ ��ÿ�ո�1�֣�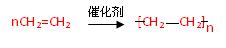 (2��) �Ӿ۷�Ӧ��1�֣�
(2��) �Ӿ۷�Ӧ��1�֣�


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��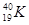 �� ��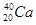 | B�����ʯ��ʯī | C��H2O��D2O | D��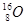 �� ��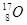 �� ��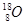 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��CsI | B��NaF | C��LiI | D��KCl |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
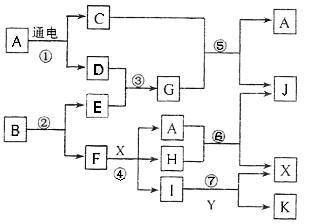
 �밴Ҫ����գ���1��д��A�ĵ���ʽ ��FԪ�������ڱ�λ�� ��2����Ӧ�ݵĻ�ѧ����ʽΪ�� ��
�밴Ҫ����գ���1��д��A�ĵ���ʽ ��FԪ�������ڱ�λ�� ��2����Ӧ�ݵĻ�ѧ����ʽΪ�� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ���û�������CA3��A2D���۵��ɸߵ��͵�˳��Ϊ ���û�ѧʽ��ʾ����
���û�������CA3��A2D���۵��ɸߵ��͵�˳��Ϊ ���û�ѧʽ��ʾ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��������IA�� | B�������ڢ�A�� | C�������ڢ�A�� | D��������IA�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com