
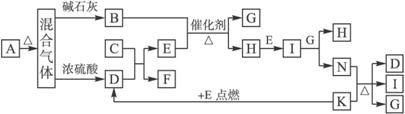
��8�֡���ͼ�е�B��K�ֱ�����йط�Ӧ��һ�ַ�Ӧ������������A��C��F��K�ǹ��塣��̬����A���Ⱥ����ɵ�����������ͨ����ʯ�ң�ֻʣ������B����ͨ��Ũ������ֻʣ������D�������ʼ��ת����ϵ����ͼ��ʾ��
��ش��������⣺
(1)д��C�ĵ���ʽ��_______________��
(2)д��ʵ������ȡB�Ļ�ѧ����ʽ��_____________________________________________��
(3)д��N��K��Ӧ�����ӷ�Ӧ����ʽ��___________________________________________��
(4)���������ͨ����ʯ�ҵõ�������B��ͨ��Ũ����õ�������D������֮����7��11�������A��_______________��
(1)![]()
(2)Ca(OH)2+2NH4Cl![]() CaCl2+2H2O+2NH3��
CaCl2+2H2O+2NH3��
(3)C+4H++4![]()
![]() 4NO2 ��+CO2 ��+2H2O
4NO2 ��+CO2 ��+2H2O
(4)NH4HCO3��(NH4)2CO3�Ļ����
�����������ͻ�ƿھ�����Ŀ�Ŀ�ʼ���֣���Ϊ��һ���ְѷ�Ӧ�����Ͳ��ַ�Ӧ�ﶼ���г���Ϊ�ƶ������ṩ�˳������֪��������������ͨ����ʯ�Һ�ܿ��ܵõ�NH3(B)��ͨ��Ũ����ɵ�CO2(D)��CO2��Na2O2��Ӧ��O2(E)�������������ڴ��������¼��ȷ�Ӧ��NO(H)��H2O(G)��NO��O2��Ӧ����NO2(I)��NO2��H2O����HNO3(N)��CO2��Mg����ȼ������MgO��C(K)��C��HNO3����NO2(I)��CO2(D)��H2O(G)������A��NH4HCO3��(NH4)2CO3��
(4)n(NH3)��n(CO2)=![]() ��
��![]() =2��1
=2��1
���Թ���A��(NH4)2CO3��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��8�֡���ͼ�Dz��ֶ�����Ԫ�صĵ��ʼ��仯�����ת����ϵͼ(�йط�Ӧ���������ɵ�ˮ����ȥ)����֪��A��B��C��DΪ���ַǽ������ʣ�����B��C��D�ڳ��³�ѹ�������壻��Ӧ�٢��ǻ��������е���Ҫ��Ӧ��������E���γ��������Ⱦ��֮һ��������K�dz��õĵ��ʣ�������L����Ư���ԣ�����Cl2��NaOH��Ӧ���Ƶã�J������Ԫ����ɣ�����Է�������Ϊ32����Ҫ����գ�

(1)AԪ�������ڱ��е�λ����______________��
(2)J�Ļ�ѧʽΪ______________��
(3)д��L��Һ�뻯����E��Ӧ�����ӷ���ʽ��________________________________��
(4)��Fˮ��Һ�������ϵõ�һ����ɫ����Һ���ڴ���Һ��ֻ�����������ӣ�����������Ũ�ȴӴ�С˳������ǿ��ܵ�һ�ֹ�ϵ��ʾ������
| ��Һ����� | ����Ũ�ȴ�С˳�� | |
| 1 | ���� | |
| 2 | ���� | |
| 3 | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��13�֣�����ѧ����ѡ���л���ѧ������
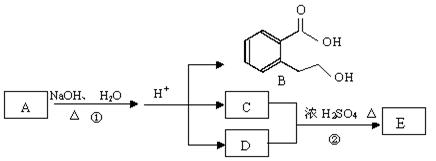
��ͼ�� A��B��C��D��E��Ϊ�л��������֪��C�ܸ�NaHCO3������Ӧ���úͣĵ���Է���������ȣ���EΪ��֧���Ļ����
������ͼ�ش����⣺
��1����֪E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ������E�ķ���ʽΪ ��
��2��������B���ܷ����ķ�Ӧ�� ������ĸ��ţ���
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧd��������Ӧ e��ˮ�ⷴӦ f���û���Ӧ
��3����Ӧ�ڵĻ�ѧ����ʽ�� ��
��4��C�����еĹ����������� ��A�Ľṹ��ʽ�� ��
��5��ͬʱ������������������B��ͬ���칹�����Ŀ�� ����
���м��ȡ�������ṹ �����ڷǷ������� ���� FeCl3 ��Һ������ɫ��Ӧ��д����������һ��ͬ���칹��Ľṹ��ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013������ʡ�����еڶ�ʮ��ѧ�����߿��캽���ԣ�������ѧ�Ծ����������� ���ͣ������
����ѧ����ѡ���л���ѧ������
��ͼ�� A��B��C��D��E��Ϊ�л��������֪��C�ܸ�NaHCO3������Ӧ���úͣĵ���Է���������ȣ���EΪ��֧���Ļ����
������ͼ�ش����⣺
��1����֪E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ������E�ķ���ʽΪ ��
��2��������B���ܷ����ķ�Ӧ�� ������ĸ��ţ���
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧ d��������Ӧ e��ˮ�ⷴӦ f���û���Ӧ
��3����Ӧ�ڵĻ�ѧ����ʽ�� ��
��4��C�����еĹ����������� ��A�Ľṹ��ʽ�� ��
��5��ͬʱ������������������B��ͬ���칹�����Ŀ�� ����
���м��ȡ�������ṹ �����ڷǷ������� ���� FeCl3��Һ������ɫ��Ӧ��д����������һ��ͬ���칹��Ľṹ��ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ������ʡ�����и����߿��캽���ԣ�������ѧ�Ծ��������棩 ���ͣ������
����ѧ����ѡ���л���ѧ������
��ͼ�� A��B��C��D��E��Ϊ�л��������֪��C�ܸ�NaHCO3������Ӧ���úͣĵ���Է���������ȣ���EΪ��֧���Ļ����
������ͼ�ش����⣺
��1����֪E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ������E�ķ���ʽΪ ��
��2��������B���ܷ����ķ�Ӧ�� ������ĸ��ţ���
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧ d��������Ӧ e��ˮ�ⷴӦ f���û���Ӧ
��3����Ӧ�ڵĻ�ѧ����ʽ�� ��
��4��C�����еĹ����������� ��A�Ľṹ��ʽ�� ��
��5��ͬʱ������������������B��ͬ���칹�����Ŀ�� ����
���м��ȡ�������ṹ �����ڷǷ������� ���� FeCl3��Һ������ɫ��Ӧ��д����������һ��ͬ���칹��Ľṹ��ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com