
·ÖĪö £Ø1£©¢ŁČÜŅŗÓėAl2O3·“Ó¦ŗóÓŠAl3+Éś³É£¬ĖµĆ÷øĆČÜŅŗĻŌĖįŠŌ£¬ÓėĒāĄė×ÓÄÜ·“Ó¦µÄĄė×Ó²»ÄÜ“óĮæ¹²“ę£»
¢ŚøĆČÜŅŗÓėAl2O3·“Ó¦ŗóÓŠAlO2-Éś³ÉĖµĆ÷øĆČÜŅŗĻŌ¼īŠŌ£¬²»ÓėĒāŃõøłĄė×Ó·“Ó¦µÄĄė×ÓæÉ“óĮæ¹²“ę£»
£Ø2£©CÓėE”¢DÓėE²śÉśĘųĢ壬ĪåÖÖĪļÖŹÖŠ£¬ÄÜÓėĮ½ÖÖĪļÖŹµÄ²śÉśĘųĢåµÄÖ»ÓŠK2CO3£¬ŌņEĪŖK2CO3£¬ŌņC”¢DĪŖHNO3”¢HClÖŠµÄĪļÖŹ£¬¶ųDÓėBÄܲśÉś³Įµķ£¬ŌņDĪŖHCl£¬ÄÜÓėĻõĖįŅų²śÉśAgCl³Įµķ£¬ĖłŅŌCĪŖHNO3£¬BĪŖAgNO3£¬ŌņAĪŖBaCl2£®
½ā“š ½ā£ŗ£Ø1£©¢ŁČÜŅŗÓėAl2O3·“Ó¦ŗóÓŠAl3+Éś³É£¬ĖµĆ÷øĆČÜŅŗĻŌĖįŠŌ£¬Ņ»¶Øŗ¬ÓŠH+£»OH-”¢HCO3-Ņ»¶Ø²»»į“óĮæ“ęŌŚ£¬
¹Ź“š°øĪŖ£ŗH+£»OH-”¢HCO3-£»
¢ŚøĆČÜŅŗÓėAl2O3·“Ó¦ŗóÓŠAlO2-Éś³ÉĖµĆ÷øĆČÜŅŗĻŌ¼īŠŌ£¬H+”¢Mg2+”¢Ag+ÓėOH-²»ÄÜ“óĮæ¹²“ę£¬±Ų¶Øŗ¬ÓŠŅ»ÖÖŃōĄė×Ó£ŗNa+£¬¹ŹČÜŅŗÖŠŅ»¶Øŗ¬ÓŠOH-”¢Na+£¬æÉÄÜŗ¬ÓŠCl-”¢NO3-£¬¹Ź“š°øĪŖ£ŗOH-”¢Na+£»Cl-”¢NO3-£»
£Ø2£©CÓėE”¢DÓėE²śÉśĘųĢ壬ĪåÖÖĪļÖŹÖŠ£¬ÄÜÓėĮ½ÖÖĪļÖŹµÄ²śÉśĘųĢåµÄÖ»ÓŠK2CO3£¬ŌņEĪŖK2CO3£¬ŌņC”¢DĪŖHNO3”¢HClÖŠµÄĪļÖŹ£¬¶ųDÓėBÄܲśÉś³Įµķ£¬ŌņDĪŖHCl£¬ÄÜÓėĻõĖįŅų²śÉśAgCl³Įµķ£¬ĖłŅŌCĪŖHNO3£¬BĪŖAgNO3£¬ŌņAĪŖBaCl2£¬
¹Ź“š°øĪŖ£ŗBaCl2£»AgNO3£»HNO3£»HCl£»K2CO3£®
µćĘĄ ±¾Ģāæ¼²éĄė×ӵĹ²“ę”¢ĪŽ»śĪļµÄĶʶĻ£¬ĪŖøßæ¼³£¼ūĢāŠĶ£¬×¢ÖŲŠÅĻ¢µÄ³éČ”ŗĶŃõ»Æ»¹Ō·“Ó¦µÄ漲飬¶ŌѧɜĖ¼Ī¬ŃĻĆÜŠŌÓŠ½ĻŗƵÄѵĮ·£¬¾ßÓŠ¼įŹµµÄ»ł“”ÖŖŹ¶¼“æɽā“š£¬×¢ŅāŃõ»ÆĀĮµÄĮ½ŠŌ£¬°ŃĪÕĪļÖŹµÄŠŌÖŹ£¬½įŗĻ·“Ó¦ĻÖĻóĶʶĻ£¬ĢāÄæÄѶČÖŠµČ£®


 ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø
ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | 46 | B£® | 40 | C£® | 37.5 | D£® | 17 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
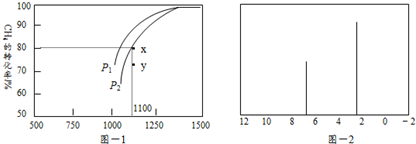
 £®
£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
| ŹµŃé ±ąŗÅ | ŹµŃéÄæµÄ | T/K | Fe/g | c£ØH2SO4£©/mol•L-1 |
| ¢Ł | ĪŖŅŌĻĀŹµŃé×÷²Īæ¼ | 298 | 10 | 1.0 |
| ¢Ś | Ģ½¾æĪĀ¶Č¶Ō·“Ó¦ĖŁĀŹµÄÓ°Ļģ | |||
| ¢Ū | 298 | 10 | 18.4 |
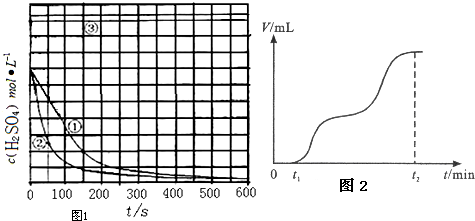
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | æĻ¶Ø²»“ęŌŚµÄĄė×ÓŹĒFe3+”¢Cu2+”¢SO42-”¢CO32- | |
| B£® | æĻ¶Ø“ęŌŚµÄĄė×ÓŹĒNa+”¢K+”¢SO32-”¢Br-”¢Cl- | |
| C£® | ĪŽ·ØČ·¶ØŌČÜŅŗÖŠŹĒ·ń“ęŌŚCl- | |
| D£® | Čō²½Öč ¢ÜøÄÓĆBaCl2ŗĶŃĪĖįµÄ»ģŗĻČÜŅŗ£¬Ōņ¶ŌČÜŅŗÖŠĄė×ÓµÄÅŠ¶ĻĪŽÓ°Ļģ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
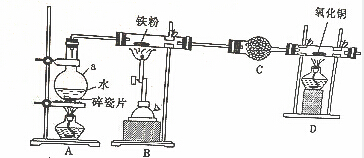
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ÓĆĮæĶ²²āµĆÅÅĖ®·ØŹÕ¼ÆÖʵƵÄĒāĘųĢå»żĪŖ5.28 mL | |
| B£® | ÓĆĮ½Ö»250 mLµÄČŻĮæĘæÅäÖĘ0.1mol/L 500 mLµÄNaOHČÜŅŗ | |
| C£® | ÓĆĶŠÅĢĢģĘ½³ĘµĆ2.50gµØ·Æ£¬ŹÜČČ³ä·ÖŹ§Ė®ŗ󣬹ĢĢåÖŹĮæ¼õĒį0.90g | |
| D£® | ³£ĪĀĻĀ£¬²āµĆ1molN2µÄÖŹĮæĪŖ28g |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā

| A£® | ½šŹōMg²ÉÓĆ¢Ś¶Ń»ż·½Ź½ | |
| B£® | ¢ŁŗĶ¢ŪÖŠŌ×ÓµÄÅäĪ»Źż·Ö±šĪŖ£ŗ8”¢12 | |
| C£® | ¶ŌÓŚ²ÉÓĆ¢Ś¶Ń»ż·½Ź½µÄ½šŹō£¬ŹµŃé²āµĆW gøĆ½šŹōµÄĢå»żĪŖV cm3£¬Ōņ°¢·ü¼ÓµĀĀŽ³£ŹżNAµÄ±ķ“ļŹ½ĪŖ$\frac{MV}{W•£Ø\frac{4}{\sqrt{3}}r£©^{3}}$ | |
| D£® | ¢ÜÖŠæÕ¼äĄūÓĆĀŹµÄ±ķ“ļŹ½ĪŖ£ŗ$\frac{\frac{4}{3}¦Š{r}^{3}”Į4}{£Ø\frac{4}{\sqrt{2}}r£©^{3}}$”Į100% |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com