
���и������ӣ���ָ�������²��ܴ����������( )
��ij��ɫ����������Һ�У�Cl����Na����MnO ��SO
��SO
��ˮ���������c(H��)��10��12mol��L��1����Һ�У�K����Na����Cl����NO
��ʹ��ɫʯ����Һ������Һ�У�Fe2����Mg2����NO ��Cl��
��Cl��
�ܼ������NaOH��Һ��ɵõ�������Һ��K����Ba2����HCO ��Cl��
��Cl��
��c(H��)<c(OH��)����Һ�У�SO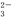 ��S2����K����Na��
��S2����K����Na��
�������Ը��������Һ�У�Na����NH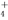 ��I����ClO��
��I����ClO��
����ʹpH��ֽ����ɫ����Һ�У�Na����AlO2����K����CO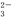
A���٢ۢܢ� B���ڢۢݢ� C���ڢݢ� D���ڢܢݢ�

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ��ӱ�ʡ�߶���9.21�ܿ���ѧ���������棩 ���ͣ�ѡ����
����˵������ȷ����( )
A��8NH3(g)+6NO2(g)=7N2(g)+12H2O(g)����H<0 ����÷�Ӧһ�����Է�����
B����ȡ�����е�Ԫ��ʱ��Ϊ��I-��ȫ����ΪI2����HNO3������������H2O2Ч����
C��ԭ��طŵ�����У���������һ�����ᣬ����������һ������
D������Ksp( MgCO3)< Ksp[Mg(OH)2],���ˮ����MgCO3����ת��ΪMg(OH)2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017���Ĵ�ʡ���½���ѧ����У�����ϵ�һ��������ѧ���������棩 ���ͣ�ѡ����
����ָ����Ӧ�����ӷ���ʽ��ȷ����
A��Ca(ClO)2��Һ��ͨ������SO2��Ca2++2ClO��+SO2+H2O=CaSO4��+H++Cl��+ HClO
B����FeCl2��Һ��������NaClO��Һ��6Fe2++3ClO��+3H2O=2Fe(OH)3��+4Fe3++3Cl��
C��NH4HCO3��Һ��������NaOH��Һ��ϣ�HCO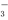 ��OH��===CO
��OH��===CO ��H2O
��H2O
D����Fe(NO3)3��Һ�м���������HI��Һ��2Fe3��+2I�� = 2Fe2��+I2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ɽ��ʡ�Ͳ��и�����ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ������
�桢�����ѡ����䲻����ѧ�γ����Ľ���Ԫ�أ����ڹ�ҵ������������Ҫ���á�
��1����������(CeO2)��ƽ�������ʾ����������ҪӦ�á�CeO2��ϡ�����H2O2�������¿�����Ce3+��CeO2�ڸ÷�Ӧ����___________����
��2����Ȼ��Cr��Ҫ��+3�ۺ�+6�ۣ�Cr2O7-�����ڡ�+6�۵�Cr������ϸ����ͻ�䣬�������������ƽ��仹ԭΪ+3�۵ĸ���д�����ӷ���ʽ��__________________
��3���ѣ�Ti������Ϊ����ʮһ���͵Ľ���������ҵ����550��ʱ���������Ȼ��ѷ�Ӧ���Ƶ��ѣ��÷�Ӧ�Ļ�ѧ����ʽ�� ��
��4��NiSO4��xH2O��һ����ɫ������ˮ�ľ��壬�㷺���ڶ�������صȣ����ɵ�Ʒ���(�����⣬������ͭ��п������Ԫ��)Ϊԭ�ϻ�á������������£�

 ������Һ���м���FeS��Ϊ�˳�ȥCu2+��Zn2+�����ʣ���ȥCu2+�����ӷ���ʽΪ__________��
������Һ���м���FeS��Ϊ�˳�ȥCu2+��Zn2+�����ʣ���ȥCu2+�����ӷ���ʽΪ__________��
�ڶ���Һ���ȼ�H2O2�ٵ�pH����pH��Ŀ���� ��
����Һ�����ʵ���Ҫ�ɷ���NiSO4����Na2CO3���˺��ټ�����ϡ�����ܽ�������NiSO4��������������Ŀ����____________��
��Ϊ�ⶨNiSO4��xH2O������x��ֵ����ȡ26��3 g�����������ȫʧȥ�ᾧˮ��ʣ�����15��5 g������x��ֵ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ɽ��ʡ�Ͳ��и�����ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�������ȣ�ClO2����һ�ֻ���ɫ������ˮ�����壬����������ˮ������ʵ����ͨ�����¹����Ʊ��������ȡ�
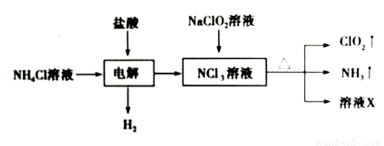
����˵����ȷ����
A�����ʱ������Ӧ�����ӷ���ʽΪ��NH4++3Cl�� 2H2��+NCl3
2H2��+NCl3
B����ҺX�д������ڵ���������Cl����OH��
C�����ñ���ʳ��ˮ��ȥClO2�е�NH3
D������������ȥ����ˮ�в�����NaClO2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ɽ��ʡ�Ͳ��и�����ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵����ȷ����
A������ȼ�ϵ�ع���ʱ��1mol H2��������ʧȥ2mol����
B��0��1mol/LNa2CO3��Һ���Ⱥ���Һ��CO32-����ĿС��6.02��1022��
C�����³�ѹ�£�22��4LCl2�к��еķ�����Ϊ6.02��1023��
D�������£�ϡ��0��1mol/LCH3COOH��Һ����Һ�ĵ�����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꼪��ʡ��һ�ϵ�һ���¿���ѧ�Ծ��������棩 ���ͣ������
��1�����飨CH4����Ħ������Ϊ �����ԭ������Ϊ ��
��2��8gCH4��Լ���� �����ӣ��ڱ�״������ռ�����ԼΪ L��
��3���������ʵ�����ȵļ���Ͱ�����NH3����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꼪��ʡ��һ�ϵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
������������ȷ����
A�����ʵ����������ʵ����� B�����ʵ����������ʵ�����
C�����ʵ�������Ħ�� D�����ʵ����ĵ�λ��Ħ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017�����ʡ������ѧ��9���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��9g���۸�һ���������������ĩ����γ����ȼ������� ���ȷ�Ӧ֮�����ù����к���������Ϊ18g������������ĩ�����ǣ� ��
���ȷ�Ӧ֮�����ù����к���������Ϊ18g������������ĩ�����ǣ� ��
��Fe2O3��MnO2 ��MnO2��V2O5 ��Cr2O3��V2O5 ��Fe3O4��FeO
A���٢� B���٢� C���ڢ� D���ڢ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com