
��1����Ҫ����д���пո��谢���ӵ�����ΪNA��
��39 g Na2O2�е�Na+_________ NA��
��0.5mol H2O�е�������Ϊ_________NA��
��1.204��1024��ˮ���ӵ�����Ϊ_________ g��
�ܱ�״���£�10g CO��CO2�������������6.72L����˻�������У�CO��CO2�����ʵ���֮���� ��
��9.2g����������NOx�к���Nԭ����Ϊ0.2mol����x��ֵΪ ��
��2�����ڷ�����ᴿ���ʵķ����У�
A����(����) B��ȡ C���� D�ؽᾧ E��Һ
������ᴿ���и������� ����ѡ����������ʵķ����ı�����ڙM���ϡ�
����ѡ����������ʵķ����ı�����ڙM���ϡ�
�ٳ�ȥCa(OH)2��Һ��������CaCO3�� ��
�ڷ������Ȼ�̼��ˮ�Ļ���� ��
�۷������ͺͲ��� ��
���ᴿ�Ȼ��ƺ�����ػ����Һ�е������ ��
��3���ҳ����и��������������������ʵ����ͬ��һ�����ʣ��û�ѧʽ��д�ں���ĺ����ϡ�
�� CaO��Na2O��CO2��CuO ��
�� H2��N2��P��Cu ��
�� S��Fe��Cu��Zn ��
�� HCl��H2O��H2SO4��HNO3 ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�갲��ʡ����У�߶�����ĩ��ѧ���������棩 ���ͣ�ѡ����
�������Һ�ĵ絼��Խ��������Խǿ����0.100mol��L��1��NaOH��Һ�ֱ�ζ�10.00mL Ũ�Ⱦ�Ϊ0.100mol��L��1�������CH3COOH��Һ�����ô�������õζ���������Һ�絼����ͼ��ʾ������˵������ȷ����( )
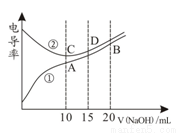
A�����ߢٴ����ζ�CH3COOH��Һ������
B������ͬ�¶��£�A��B��C ������Һ��ˮ����� c(H+)��B��A=C
C��D����Һ�У�c(Cl��)=2c(OH��)��2c(H+)
D��A����Һ�У�c(CH3COO��)��c(OH��)��c(H+) = 0.05mol��L��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�����и߶���ѧ����ĩ��ѧ�Ծ��������棩 ���ͣ��ƶ���
A��B��D��E���ڡ�G��M����Ԫ��λ��Ԫ�����ڱ�ǰ�����ڣ�ԭ��������������Ԫ�����ڱ���ԭ�Ӱ뾶��С����A��Bԭ�������������δ�ɶԵ��ӣ�������DE2Ϊ����ɫ���壬�ڵĵ�������ˮ��Ӧ��ֻ����������G��ǰ�������е�һ��������С��Ԫ�أ�M�ǵ�������Ԫ�أ������ֻ��һ�����ӣ����������Ӿ�������
��ش��������⣺
��1��Ԫ��B��D��E�ĵ�һ�������ɴ�С��˳��Ϊ________����Ԫ�ط��ű�ʾ����
��2����̬Gԭ�ӵļ۵����Ų�ʽ�� ��M��Ԫ�����ڱ��е�λ���� ��
��3��Ԫ��D�ͣ���ɷ��ӣΣڣ��ռ乹��Ϊ________���÷���Ϊ ���ӣ���Ի�Ǽ��ԣ������У�ԭ�ӵ��ӻ���ʽΪ________��
��4��D������������Ӧ��ˮ�����������DA3�����������ӻ������ҳ����£����ס�������Һ��pH������5������ˮ�������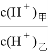 = ������Һ���������ӵ����ʵ���Ũ���ɴ�С��˳���� ��
= ������Һ���������ӵ����ʵ���Ũ���ɴ�С��˳���� ��
��5��Ԫ��Xλ�ڵ������ڣ����̬ԭ�ӵ��ڲ���ȫ���������ӣ�������������Ϊ2��Ԫ��Y��̬ԭ�ӵ�3p�������4�����ӡ�
��X��Y���γɻ�������X�Ļ��ϼ۵�������������Y�ﵽ8���ӵ��ȶ��ṹ��û�����Ļ�ѧʽΪ____________��
��E���⻯�H2E�����Ҵ��е��ܽ�ȴ���H2Y����ԭ���� ��
��X���Ȼ����백ˮ��Ӧ���γ������[X(NH3)4]Cl2��1mol��������к��Ц� ������ĿΪ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�����и߶���ѧ����ĩ��ѧ�Ծ��������棩 ���ͣ�ѡ����
��ij������һҺ�����ʱ��ƽ��ʱ���ΪV1ml�����Ӷ���ΪV2ml�����Ӷ���ΪV3ml����V3��V1��V2������ʹ�õ����������� �� ��
A. ����ƿ B. ��Ͳ C. ��ʽ�ζ��� D. ������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�����и߶���ѧ����ĩ��ѧ�Ծ��������棩 ���ͣ�ѡ����
����������ȷ���ǣ� ��
A�����߷�Ӧ���¶ȣ���ʹ��Ӧ��Ļ��Ѹ�ٽ���
B���Ҵ������Ͷ��ǿ�������Դ��Ӧ�����ƹ㡰�Ҵ����͡�
C���õ��ˮ�ķ�����ȡ�����������Ի�����Դ���������
D���ƹ�ʹ��̫���ܡ����ܵ���Դ�������ڻ�������ЧӦ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�㰲�и�һ��ѧ����ĩ��ѧ�Ծ��������棩 ���ͣ�ѡ����
��106 g��Na2CO3��84 gNaHCO3�Ⱦ����ǿ�Ⱥ��ٷֱ������������ᷴӦ������˵������ ����
����
A���ų�CO2������һ���� B��ǰ�����ɵ�NaCl�Ǻ��ߵĶ���
C����������һ���� D����������������ǰ�ߵ�һ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�㰲�и�һ��ѧ����ĩ��ѧ�Ծ��������棩 ���ͣ�ѡ����
����������Ҫ������������Щ���ؾ�����
�ٷ��ӵ�ֱ�� �ڷ��ӵ���Է�������
�۷��Ӽ��ƽ������ ����������ʵ���
A���٢� B���ڢ� C���٢� D���ۢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017�켪��ʡ�������������Ļ�ѧ���������棩 ���ͣ�ʵ����
�������ף���ѧʽ��POCl3���������뵼����Ӽ������άԭ�ϡ��Ȼ�ˮ�ⷨ�������������������£�
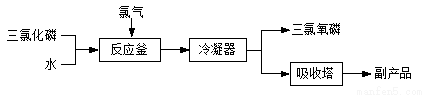
��1���Ȼ�ˮ�ⷨ�����������Ļ�ѧ����ʽΪ___________________________��
��2���Ȼ�ˮ�ⷨ������������ʱ����������ף���ҪΪH3PO3��H3PO4�ȣ���ˮ����֪H3PO3��һ�ֵ���ɫ���壬��������ˮ�Ķ�Ԫ���ᡣ
������20 mL H3PO3��Һ��ͬŨ�ȵ�NaOH��Һ40mLǡ����ȫ��Ӧ�����ɵ���Ϊ___________________������Ρ�����ʽ�Ρ�����
��H3PO3�У�PԪ�صĻ��ϼ�Ϊ_______________________��H3PO3���Խ���Һ�е�Ag+��ԭ���Ӷ����ڻ�ѧ������д�������ӷ���ʽ_________________________��
�۴�����ˮʱ���������м�������Ư�ۣ��ټ�����ʯ�ҵ���pH����Ԫ��ת��Ϊ����Ƴ��������գ�����Ư�۵�������______________________________________��
����������ķ�ˮ��c(PO43��)=4��10��7mol��L��1����Һ��c(Ca2+)=________mol��L��1������֪Ksp[Ca3(PO4)2]=2��10��29��
��3��ͨ��������·����Բⶨ�������ײ�Ʒ��ClԪ�غ�����ʵ�鲽�����£�
��ȡa g��Ʒ����ƿ�У���������NaOH��Һ������ȫˮ����ϡ���������ԡ�
������ƿ�м���0.1000 mol��L��1��AgNO3��Һ40.00 mL��ʹCl����ȫ������
�������м���2 mL������������ҡ����ʹ�������汻�л��︲�ǡ�
��������ָʾ������c mol��L��1 NH4SCN��Һ�ζ�����Ag+���յ㣬�������������
��֪��Ksp(AgCl)=3.2��10��10��Ksp(AgSCN)=2��10��12
�ٵζ�ѡ�õ�ָʾ����__________��ѡ����ĸ����
a��FeCl2b��NH4Fe(SO4)2 c������ d������
��ʵ���������δ�����������������������ClԪ�غ�������_______���ƫ����ƫС�����䡱����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ӱ�ʡ��������ĩ��ѧ�Ծ��������棩 ���ͣ�������
��MnO2��FeSO4��Һ�������ַ�Ӧ����ˣ�����Һ������60����ټ���Na2CO3��Һ�����տ��Ƶü�ʽ̼����[aMnCO3��bMn(OH)2��cH2O]��
��1���÷���м�����ᷴӦ�Ʊ�FeSO4��Һʱ��������м�������ֵ�Ըߣ�ԭ���� ����Ӧǰ�轫����м����Na2CO3��Һ���ݣ���Ŀ���� ��
��2��Ϊ�ⶨ��ʽ̼������ɣ�ȡ7.390 g��Ʒ�������ᣬ����CO2 224.0 mL(��״��)�������500 mL��Һ��ȷ��ȡ10.00 mL����Һ����0.0500 mol��L-1 EDTA(��ѧʽNa2H2Y)����Һ�ζ����е�Mn2+(ԭ��ΪMn2+ +H2Y2��=M nY2��+2H+)�����յ�ʱ����EDTA����Һ28.00 mL��ͨ������ȷ������Ʒ�Ļ�ѧʽ��
nY2��+2H+)�����յ�ʱ����EDTA����Һ28.00 mL��ͨ������ȷ������Ʒ�Ļ�ѧʽ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com