
 ijѧ����0.1000mol/L KOH��Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
ijѧ����0.1000mol/L KOH��Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����| �ζ����� | ������Һ���/mL | ����Һ�����/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 20 | 1.02 | 21.03 |
| 2 | 20 | 2.00 | 25.00 |
| 3 | 20 | 0.60 | 20.6 |
| c(��)��V(��) |
| V(��) |
| c(��)��V(��) |
| V(��) |
| 21.03-1.02+20.6-0.6 |
| 2 |
| c(��)��V(��) |
| V(��) |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��12�֣�ijѧ����0.1000 mol��L��1������������Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
A��ȡ20.00 mL����������Һע��ྻ����ƿ�У�������2��3�η�̪��Һ
B���ñ�����������Һ��ϴ�ζ���2��3��
C����ʢ�б�����������Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ������������Һע���ʽ�ζ�������0���̶�����2��3mL��
E������Һ������0����0���̶����£����¶�����Ϊ3.10mL
F������ƿ���ڵζ������棬�ñ�NaOH��Һ�ζ����յ㲢���µζ��ܵĶ�����
�ش��������⣺
��1����ȷ�IJ���˳���ǣ�B�� ��F������ţ���
��2������B���������Ŀ���� ��
��3������A�������֮ǰ�������ô�����Һ��ϴ��ƿ����Եζ������Ӱ��
�� ���ƫ��ƫС������Ӱ�족����
��4���жϵ���ζ��յ��ʵ�������� ��
��5�����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ mL��
��6�����ݶ���������ô���Һ��Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��12�֣�ijѧ����0.1000 mol��L��1������������Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
A��ȡ20.00 mL����������Һע��ྻ����ƿ�У�������2��3�η�̪��Һ
B���ñ�����������Һ��ϴ�ζ���2��3��
C����ʢ�б�����������Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ������������Һע���ʽ�ζ�������0���̶�����2��3mL��
E������Һ������0����0���̶����£����¶�����Ϊ3.10mL
F������ƿ���ڵζ������棬�ñ�NaOH��Һ�ζ����յ㲢���µζ��ܵĶ�����
�ش��������⣺
��1����ȷ�IJ���˳���ǣ�B�� ��F������ţ���
��2������B���������Ŀ���� ��
��3������A�������֮ǰ�������ô�����Һ��ϴ��ƿ����Եζ������Ӱ��
�� ���ƫ��ƫС������Ӱ�족����
��4���жϵ���ζ��յ��ʵ�������� ��
��5�����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ mL��
��6�����ݶ���������ô���Һ��Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꽭��ʡ��Ǩ�������غ�����ѧ�߶���ѧ�����п���ѧ������������ ���ͣ�ʵ����
ijѧ����0.1000 mol��L��1������������Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
A��ȡ20.00 mL����������Һע��ྻ����ƿ�У�������2��3�η�̪��Һ
B���ñ�����������Һ��ϴ�ζ���2��3��
C����ʢ�б�����������Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ������������Һע���ʽ�ζ�������0���̶�����2��3mL��
E������Һ������0����0���̶����£����¶�����Ϊ3.10mL
F������ƿ���ڵζ��ܵ����棬�ñ�NaOH��Һ�ζ����յ㲢���µζ��ܵĶ������ش��������⣺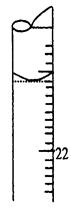
��1����ȷ�IJ���˳���ǣ�B�� ��F������ţ���
��2������B���������Ŀ���� ��
��3������A�������֮ǰ�������ô�����Һ��ϴ��ƿ����Եζ������Ӱ��
�� ���ƫ��ƫС������Ӱ�족����
��4���жϵ���ζ��յ��ʵ�������� ��
��5�����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ mL��
��6�����ݶ���������ô���Һ��Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�콭��ʡ��Ǩ�и߶���ѧ�����п���ѧ���������棩 ���ͣ�ʵ����
ijѧ����0.1000 mol��L��1������������Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
A��ȡ20.00 mL����������Һע��ྻ����ƿ�У�������2��3�η�̪��Һ
B���ñ�����������Һ��ϴ�ζ���2��3��
C����ʢ�б�����������Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ������������Һע���ʽ�ζ�������0���̶�����2��3mL��
E������Һ������0����0���̶����£����¶�����Ϊ3.10mL
F������ƿ���ڵζ��ܵ����棬�ñ�NaOH��Һ�ζ����յ㲢���µζ��ܵĶ������ش��������⣺
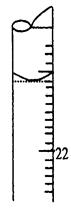
��1����ȷ�IJ���˳���ǣ�B�� ��F������ţ���
��2������B���������Ŀ���� ��
��3������A�������֮ǰ�������ô�����Һ��ϴ��ƿ����Եζ������Ӱ��
�� ���ƫ��ƫС������Ӱ�족����
��4���жϵ���ζ��յ��ʵ�������� ��
��5�����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ mL��
��6�����ݶ���������ô���Һ��Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013���㽭ʡ�����и߶����п��Ի�ѧ�Ծ��������棩 ���ͣ������
��12�֣�ijѧ����0.1000 mol��L��1������������Һ�ζ�δ֪Ũ�ȵ�������Һ��������ɷֽ�Ϊ���¼�����
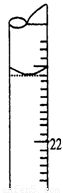
A��ȡ20.00 mL����������Һע��ྻ����ƿ�У�������2��3�η�̪��Һ
B���ñ�����������Һ��ϴ�ζ���2��3��
C����ʢ�б�����������Һ�ļ�ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ
D��ȡ������������Һע���ʽ�ζ�������0���̶�����2��3mL��
E������Һ������0����0���̶����£����¶�����Ϊ3.10mL
F������ƿ���ڵζ������棬�ñ�NaOH��Һ�ζ����յ㲢���µζ��ܵĶ�����
�ش��������⣺
��1����ȷ�IJ���˳���ǣ�B�� ��F������ţ���
��2������B���������Ŀ���� ��
��3������A�������֮ǰ�������ô�����Һ��ϴ��ƿ����Եζ������Ӱ��
�� ���ƫ��ƫС������Ӱ�족����
��4���жϵ���ζ��յ��ʵ�������� ��
��5�����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ mL��
��6�����ݶ���������ô���Һ��Ũ��Ϊ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com