
 ������ͼ1װ�òⶨ�к��ȵ�ʵ�鲽�����£�
������ͼ1װ�òⶨ�к��ȵ�ʵ�鲽�����£�| �¶� ʵ����� | ��ʼ�¶�t1�� | ��ֹ�¶�t2�� | �¶Ȳ�ƽ��ֵ��t2-t1���� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 29.5 | |
| 2 | 27.0 | 27.4 | 27.2 | 32.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.2 | |
| 4 | 26.4 | 26.2 | 26.3 | 29.8 | |
���� ��1���������ȼƵĹ��������
��2�����ݸ����к�����ǿ���ǿ���ϡ��Һ��ȫ��Ӧ����1molˮ�ų�����������ע���ʾۼ�״̬�Ͷ�Ӧ�ʱ�д���Ȼ�ѧ����ʽ��
��3������������������Ʒ�Ӧ��ӦѸ�پ��ң������ͷŴ����ȣ�һ���Կ��ٵ�����Լ�������ɢ������������
��4�����ݻ��β���������ʹ������NaOH��Һ��Ͼ��ȣ�
��5�������ж��¶Ȳ����Ч�ԣ�Ȼ������¶Ȳ�ƽ��ֵ��
�ڸ���Q=m•c•��T���㷴Ӧ�ų��������������ݡ�H=-$\frac{Q}{n}$kJ/mol�������Ӧ�ȣ�
��a��ʵ��װ�ñ��¡�����Ч������ã�
b����ȡNaOH��Һ�����ʱ����Ҫ�Ͱ�Һ����ƽ��
c�������ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У�
d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�Ҫ���¶ȼƻ���ֱ�ٲⶨH2SO4��Һ���¶ȣ�
��6����������к�ϵʽ��NaOH��HCl������������Ũ�ȣ�
��� �⣺��1������A������Ϊ���β�����������ʴ�Ϊ�����β����������
��2��ǿ��ǿ����к���Ϊ-57.3kJ/mol���к�����ǿ���ǿ���ϡ��Һ��ȫ��Ӧ����1molˮ�ų���������ϡ�����ϡ����������Һ��Ӧ���Ȼ�ѧ����ʽΪ��
$\frac{1}{2}$H2SO4��l��+NaOH��l��=$\frac{1}{2}$Na2SO4��l��+H2O��l����H=-57.3KJ/mol��
�ʴ�Ϊ��$\frac{1}{2}$H2SO4��l��+NaOH��l��=$\frac{1}{2}$Na2SO4��l��+H2O��l����H=-57.3KJ/mol��
��3������������������Ʒ�Ӧ��ӦѸ�پ��ң������ͷŴ����ȣ�һ���Կ��ٵ�����Լ�������ɢ��������������ѡ��C��
��4���������¶ȼ��ϵĻ��β���������ؽ���ʹ������NaOH��Һ��Ͼ��ȣ���ѡ��D��
��5����4���¶Ȳ�ֱ�Ϊ��3.4�棬5.1�棬3.3�棬3.5�棬��2��������Ч���¶Ȳ�ƽ��ֵΪ3.4�棬�ʴ�Ϊ��3.4�棻
��50mL0.25mol/L������50mL0.55mol/L NaOH��Һ�����кͷ�Ӧ����ˮ�����ʵ���Ϊ0.05L��0.25mol/L��2=0.025mol����Һ������Ϊ��100ml��1g/ml=100g���¶ȱ仯��ֵΪ��T=3.4�棬������0.025molˮ�ų�������ΪQ=m•c•��T=100g��4.18J/��g•�棩��3.4��=1421.2J����1.4212KJ������ʵ���õ��к��ȡ�H=-$\frac{1.4212kJ}{0.025mol}$=-56.8kJ/mol��
�ʴ�Ϊ��-56.8kJ/mol��
��a��װ�ñ��¡�����Ч�����õ�����ƫС���к��ȵ���ֵƫС����a��ȷ��
b����ȡNaOH��Һ�����ʱ���Ӷ������ᵼ�������������������ƫ�ų�������ƫ�ߣ��к��ȵ���ֵƫ��b����
c���ֶ�ΰ�NaOH��Һ����ʢ��ϡ�����С�ձ��У�����ɢʧ�ϴ������к��ȵ���ֵƫС����c��ȷ��
d���¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳ���ϡH2SO4���¶ȣ��������ʼ�¶�ƫ�ߣ���õ�����ƫС���к��ȵ���ֵƫС����d��ȷ��
�ʴ�Ϊ��acd��
��6����ͼ��V1Ϊ30mLʱ�¶���ߣ�˵����ʱ����Ѿ���Ӧ��ϣ���ʵ����ʼ�ձ���V1+V2=50mL������V2Ϊ20mL��������к�ϵʽ��NaOH��HCl����ʽ��30mL��1mol•L-1=20mL��C��NaOH��������NaOH��Ũ����1.5 mol•L-1��
�ʴ�Ϊ��1.5 mol•L-1��
���� ���⿼���Ȼ�ѧ����ʽ�Լ���Ӧ�ȵļ��㣬��Ŀ�Ѷȴ�ע�������к��ȵĸ�������Ȼ�ѧ����ʽ����д�������Լ��ⶨ��Ӧ�ȵ��������⣮


 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����Ӧǰ���Ƿ�����ԭ�ӵĵ�ʧ | |
| B�� | ����Ӧǰ���Ƿ���ԭ�ӵ�������� | |
| C�� | ����Ӧǰ���Ƿ���Ԫ�صĻ��ϼ۷����ı� | |
| D�� | ����Ӧ���Ƿ����������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
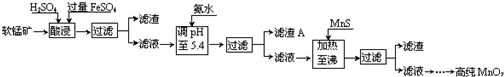
| ������ | Al��OH��3 | Fe��OH��3 | Fe��OH��2 | Mn��OH��2 | Cu��OH��2 | Zn��OH��2 | CuS | ZnS | MnS | FeS |
| pH | 5.2 | 3.2 | 9.7 | 10.4 | 6.7 | 8.0 | ��-0.42 | ��2.5 | ��7 | ��7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ܳ��ⶼ���� | B�� | ���ڢ��ⶼ���� | C�� | ֻ�Т٢ݢ��� | D�� | ȫ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
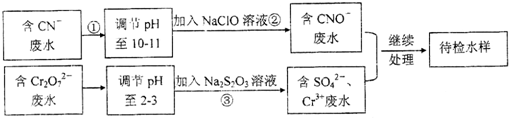
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
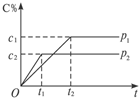 ��һ���¶��µĿ��淴Ӧ��mA��g��+nB��g��?pC��g��+qD��g����������C�����������ѹǿp1��p2��ʱ��t1��t2�Ĺ�ϵ��ͼ��ʾ���������й�ϵ����ȷ���ǣ�������
��һ���¶��µĿ��淴Ӧ��mA��g��+nB��g��?pC��g��+qD��g����������C�����������ѹǿp1��p2��ʱ��t1��t2�Ĺ�ϵ��ͼ��ʾ���������й�ϵ����ȷ���ǣ�������| A�� | p1��p2 | B�� | ��Ӧ���� | C�� | m+n��p+q | D�� | m+n��p+q |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 0.1mol•L-1NaHCO3��Һ�У�c��Na+��+c��H+��=c��HCO3-��+2c��CO32-��+c��OH-�� | |
| B�� | ������������ʵ���Ũ�ȵ�NaX������HX��Ϻ�����Ե���Һ�У�c��X-����c��Na+����c��HX����c��H+����c��OH-�� | |
| C�� | ��0.2 mol•L-1 NaA��Һ��0.1 mol•L-1��������������ü�����Һ�У�c��Na+��+c��H+��=c��A-��+c��Cl-��+c��OH-�� | |
| D�� | 1.5 L 0.1 mol•L-1 NaOH��Һ�л���ͨ��CO2����Һ����4.4 gʱ����Һ�У�c��Na+����c��CO32-����c��HCO3-����c��OH-����c��H+�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com