
��ҵ���õ�ⱥ��ʳ��ˮ�ķ�����ȡ�������ơ������������������ǰҪ���д��ξ��ơ��Իش��������⣺
���Ͽ�Ƭ��

(1)ѡ���Լ��Գ�ȥ�±����еĿ��������ʡ�
|
���� |
������Լ� |
|
CaCl2 |
��________ |
|
MgCl2 |
��________ |
|
������ |
��________ |
(2)���ƹ��̼�Ҫ����������Ca2����Mg2����SO42�� ��ȫ��������Ҫ��֤�������µ����ʣ�Ϊ������Ϊ������ѡ�������ĺ���˳��Ϊ����________��________(�����)��
(3)���������������Ե�����Һ�����Բ���ȥ������CO32��.���������ȥCO32���Ļ�ѧ����ʽΪ______________________________________________________________________��
��1����.Na2CO3 ��.NaOH ��.BaCl2 ��2����
��3��Na2CO3+2HCl=2NaCl+CO2��+H2O
����������1�����ڲ����������µ����ʣ����Կ���̼����������ȥCa2������������������ȥMg2�����Ȼ���������ȥSO42����
��2�����ڹ������Ȼ�����Ҫͨ��̼��������ȥ�����̼���Ʊ�������Ȼ����ĺ��棬����ȷ˳���Ǣ�
��3�����������ǿ��̼��ģ����������̼���Ʒ�Ӧ�ķ���ʽΪ
Na2CO3+2HCl=2NaCl+CO2��+H2O��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
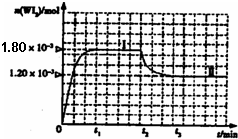 ��2010?��ׯһģ��±�ص��ʵ����ʻ��ã�±�صĻ�����Ӧ�ù㷺���о�±�ص��ʼ��仯���������Ҫ���壮
��2010?��ׯһģ��±�ص��ʵ����ʻ��ã�±�صĻ�����Ӧ�ù㷺���о�±�ص��ʼ��仯���������Ҫ���壮

| ||
| ||
| 0.036 |
| t1 |
| 0.036 |
| t1 |
| [WI2] |
| [W][I2] |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
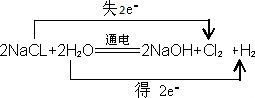
| A���Ӻ�ˮ����ȡ�嵥�ʵĹ������漰������ԭ��Ӧ | B���Ӻ�������ȡ�ⵥ�ʵĹ������漰�û���Ӧ | C����ҵ���õ�ⱥ��ʳ��ˮ�ķ�����ȡ�ȵ��ʣ�����������һ�����ڵ�Դ������ | D���Ӻ�ˮ����ȡ�Ȼ��ƵĹ������漰��ѧ��Ӧ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com