
����ѧ���������������ز��������;��ʯ�Ͳ�Ʒ��ϩΪ��ʼԭ�Ͻ��кϳɸ߷��ӻ�����F��G���ϳ�·����ͼ��ʾ��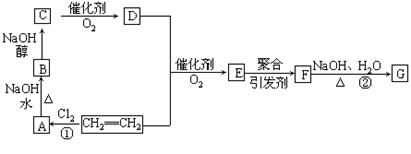
��֪��E�ķ���ʽΪC4H6O2��F�ķ���ʽΪ(C4H6O2)n���������齺������Ҫ�ɷ֣���G�ķ���ʽΪ(C2H4O)n���������ƻ�ѧ��������2CH2=CH2+2CH3COOH+O2  2C4H6O2��������ϩ����+2H2O
2C4H6O2��������ϩ����+2H2O
��֪���� �ṹ���Ƶ��л��ﲻ�ȶ�������������������
�ṹ���Ƶ��л��ﲻ�ȶ�������������������
��ش��������⣺
��1��д���ṹ��ʽ��D_______________��F___________________��
��2����Ӧ�١��ڵķ�Ӧ����______________��___________________________��
��3��д�������йط�Ӧ�ĵĻ�ѧ����ʽ��
A����B________________________________________________
B����C _______________________________________________
��1��DΪCH3COOH��FΪ ����2���ӳɷ�Ӧ��ˮ�ⷴӦ��
����2���ӳɷ�Ӧ��ˮ�ⷴӦ��
��3��ClCH2CH2Cl+H2O  ClCH2CH2OH+HCl��ClCH2CH2OH+ NaOH
ClCH2CH2OH+HCl��ClCH2CH2OH+ NaOH CH3CHO+NaCl+H2O��
CH3CHO+NaCl+H2O��
���������������ͼ�����ƶϳ�AΪClCH2CH2Cl��BΪClCH2CH2OH��CΪCH3CHO��DΪCH3COOH��FΪ ���ԣ�1���У�DΪCH3COOH��FΪ
���ԣ�1���У�DΪCH3COOH��FΪ ����2���У��١��ڵķ�Ӧ�ֱ�ΪCH2=CH2+Cl2
����2���У��١��ڵķ�Ӧ�ֱ�ΪCH2=CH2+Cl2 ClCH2CH2Cl��
ClCH2CH2Cl��
 (C2H4O)n+nCH3COOH�����䷴Ӧ���ͷֱ�Ϊ�ӳɷ�Ӧ��ˮ�ⷴӦ����3���У�A����B�ķ�Ӧ����ʽΪClCH2CH2Cl+H2O
(C2H4O)n+nCH3COOH�����䷴Ӧ���ͷֱ�Ϊ�ӳɷ�Ӧ��ˮ�ⷴӦ����3���У�A����B�ķ�Ӧ����ʽΪClCH2CH2Cl+H2O  ClCH2CH2OH+HCl��B����C�ķ�Ӧ����ʽΪClCH2CH2OH+ NaOH
ClCH2CH2OH+HCl��B����C�ķ�Ӧ����ʽΪClCH2CH2OH+ NaOH CH3CHO+NaCl+H2O��
CH3CHO+NaCl+H2O��
���㣺�л��ƶ���
������������һ���л��ƶ��⣬�Ǹ߿�������ص㡣���и�������Ϣ���ϴ��������˵���DZȽ����ġ�


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯�����ڹ�ũҵ������������������Ҫ���ã�
���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯�����ڹ�ũҵ������������������Ҫ���ã�| ��ѧ�� | H-H | N��N |
| ����/kJ?mol-1 | 435 | 943 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���㽭ʡ���ݵ�����У�߶���ѧ������������ѧ�Ծ����������� ���ͣ������
��8�֣����ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯�����ڹ�ũҵ������������������Ҫ���ã�
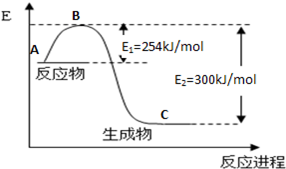
��1����ͼ��N2(g)��H2(g)��Ӧ����1mol NH3(g)�����������仯ʾ��ͼ����д��N2��H2��Ӧ���Ȼ�ѧ����ʽ�� ��
��2������֪�������ݣ�
| ��ѧ�� | H��H | N��N |
| ����/kJ��mol��1 | 435 | 943 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014��ɽ��ʡ�߶�3���¿���ѧ�Ծ��������棩 ���ͣ��ƶ���
����ѧ���������������ز��������;��ʯ�Ͳ�Ʒ��ϩΪ��ʼԭ�Ͻ��кϳɸ߷��ӻ�����F��G���ϳ�·����ͼ��ʾ��

��֪��E�ķ���ʽΪC4H6O2��F�ķ���ʽΪ(C4H6O2)n���������齺������Ҫ�ɷ֣���G�ķ���ʽΪ(C2H4O)n���������ƻ�ѧ��������2CH2=CH2+2CH3COOH+O2
 2C4H6O2��������ϩ����+2H2O
2C4H6O2��������ϩ����+2H2O
��֪���� �ṹ���Ƶ��л��ﲻ�ȶ�������������������
�ṹ���Ƶ��л��ﲻ�ȶ�������������������
��ش��������⣺
��1��д���ṹ��ʽ��D_______________��F___________________��
��2����Ӧ�١��ڵķ�Ӧ����______________��___________________________��
��3��д�������йط�Ӧ�ĵĻ�ѧ����ʽ��
A����B________________________________________________
B����C _______________________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꽭��ʡ������ʮһ�أ��У�������ѧ������������ѧ�Ծ��������棩 ���ͣ������
���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯�����ڹ�ũҵ������������������Ҫ���á��ش����й��ڵ�Ԫ�ص��й����⡣
��1����CH4����ԭ��������������������������Ⱦ����֪��
��CH4��g��+4NO2��g�� =4NO��g�� + CO2��g�� +2H2O��g�� ���SH=" -574" kJ��mol��1
��CH4��g�� +4NO��g�� =2N2��g�� + CO2��g�� + 2H2O��g�����SH=" -1160" kJ��mol��1
��H2O��g�� = H2O��l�� �� ��H= -44��0 kJ��mol��1
д��CH4��g����NO2��g����Ӧ����N2��g����CO2��g����H2O��1�����Ȼ�ѧ����ʽ ���÷�Ӧ���ر�SS 0�������������������="��" ����
��2���û���̿��ԭ��������������йط�ӦΪ��C��s��+2NO��g�� N2��g��+CO2��g�� ��ij�о�С����ij�ܱյ������������������������䣬��������������Բ��ƣ��м���NO�������Ļ���̿�����£�T1�棩�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����ұ���
N2��g��+CO2��g�� ��ij�о�С����ij�ܱյ������������������������䣬��������������Բ��ƣ��м���NO�������Ļ���̿�����£�T1�棩�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����ұ���
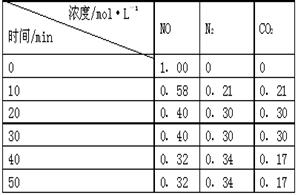
��10min~20min��V��CO2�� ��ʾ�ķ�Ӧ����Ϊ ��
�ڸ÷�Ӧ��ƽ�ⳣ���ı���ʽΪ��K= �����ݱ������ݣ�����T1��ʱ�÷�Ӧ��ƽ�ⳣ��Ϊ ���� ��������λС������
�����и�������Ϊ�жϸ÷�Ӧ�ﵽƽ����� ���������ĸ����
a��������ѹǿ���ֲ��䡡�� b��2V��NO��������= V��N2�����棩
c��������CO2������������� d�����������ܶȱ��ֲ���
��30min�ı�ijһ��������Ӧ���´ﵽƽ�⣬��ı������������ ��
��һ���¶��£�����NO����ʼŨ��������NO��ƽ��ת���� ������������䡱��С���� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com