
ijŠ£»Æѧъ¾æŠŌѧĻ°Š”×é²éŌÄ׏ĮĻĮĖ½āµ½ŅŌĻĀÄŚČŻ£ŗ
ŅŅ¶žĖį(HOOC-COOH£¬æɼņŠ“ĪŖH2C2O4)Ė×³Ę²ŻĖį£¬Ņ×ČÜÓŚĖ®£¬ŹōÓŚ¶žŌŖÖŠĒæĖį(ĪŖČõµē½āÖŹ)£¬ĒŅĖįŠŌĒæÓŚĢ¼Ėį£¬ĘäČŪµćĪŖ101.5”ę£¬ŌŚ157”ęÉż»Ŗ”£ĪŖĢ½¾æ²ŻĖįµÄ²æ·Ö»ÆѧŠŌÖŹ£¬½ųŠŠĮĖČēĻĀŹµŃé£ŗ
(1)ĻņŹ¢ÓŠ1 mL±„ŗĶNaHCO3ČÜŅŗµÄŹŌ¹ÜÖŠ¼ÓČė×ćĮæŅŅ¶žĖįČÜŅŗ£¬¹Ū²ģµ½ÓŠĪŽÉ«ĘųÅŻ²śÉś”£øĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ__________________________________________”£
(2)ĻņŹ¢ÓŠŅŅ¶žĖį±„ŗĶČÜŅŗµÄŹŌ¹ÜÖŠµĪČė¼øµĪĮņĖįĖį»ÆµÄKMnO4ČÜŅŗ£¬Õńµ“£¬·¢ĻÖĘäČÜŅŗµÄ×ĻŗģÉ«ĶŹČ„£¬ĖµĆ÷ŅŅ¶žĖį¾ßÓŠ_____________(Ģī”°Ńõ»ÆŠŌ”±”¢”°»¹ŌŠŌ”±»ņ”°ĖįŠŌ”±)£¬ĒėÅäĘ½øĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ
____ MnO4ØC + ____ H2C2O4 + _____ H+ = _____ Mn2+ + _____ CO2”ü + _____ H2O
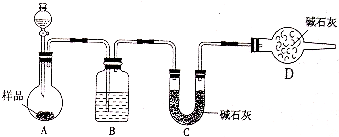
(3)½«Ņ»¶ØĮæµÄŅŅ¶žĖį·ÅÓŚŹŌ¹ÜÖŠ£¬°“ĻĀĶ¼ĖłŹ¾×°ÖĆ½ųŠŠŹµŃé(¼Š³Ö×°ÖĆĪ“±ź³ö)£ŗ
ŹµŃé·¢ĻÖ£ŗ×°ÖĆC”¢GÖŠ³ĪĒåŹÆ»ŅĖ®±ä»ė×Ē£¬BÖŠCuSO4·ŪÄ©±äĄ¶£¬FÖŠCuO·ŪÄ©±äŗģ”£¾Ż“Ė»Ų“š£ŗ
ÉĻŹö×°ÖĆÖŠ£¬DµÄ×÷ÓĆŹĒ__________________”£
ŅŅ¶žĖį·Ö½āµÄ»Æѧ·½³ĢŹ½ĪŖ_____________________________________”£
(4)øĆŠ”×éĶ¬Ń§½«2.52 g²ŻĖį¾§Ģå(H2C2O4”¤2H2O)¼ÓČėµ½100 mL 0.2 mol/LµÄNaOHČÜŅŗÖŠ³ä·Ö·“Ó¦£¬²āµĆ·“Ó¦ŗóČÜŅŗ³ŹĖįŠŌ£¬ĘäŌŅņŹĒ__________________”£(ÓĆĪÄ×Ö¼ņµ„±ķŹö)
(5)ŅŌÉĻČÜŅŗÖŠø÷Ąė×ÓµÄÅضČÓɓ󵽊”µÄĖ³ŠņĪŖ£ŗ_____________________________£»£ØÓĆĄė×Ó·ūŗűķŹ¾£©
(1)HCO3ØC + H2C2O4 = HC2O4ØC+ CO2”ü+ H2O(2·Ö)
(2) »¹ŌŠŌ(2·Ö) 2 5 6 2 10 8£Ø2·Ö)
(3) ³żČ„»ģŗĻĘųĢåÖŠµÄCO2(2·Ö) H2C2O4H2O+CO”ü+CO2”ü (2·Ö)
(4)·“Ó¦ĖłµĆČÜŅŗĪŖNaHC2O4ČÜŅŗ£¬ÓÉÓŚHC2O4ØCµÄµēĄė³Ģ¶Č±ČĖ®½ā³Ģ¶Č“󣬵¼ÖĀČÜŅŗÖŠc(H+) > c(OHØC)£¬ĖłŅŌČÜŅŗ³ŹĖįŠŌ(2·Ö)
(5)Na+>HC2O4->H+>C2O42->OH-(3·Ö)
½āĪö:ĀŌ



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā

| ||
| ||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
 ijŠ£»Æѧъ¾æŠŌѧĻ°Š”×éÉč¼ĘČēĻĀŹµŃé·½°ø£¬²ā¶Ø·ÅÖĆ¼ŗ¾ĆµÄŠ”ĖÕ“ņѳʷ֊“æ¼īµÄÖŹĮæ·ÖŹż£®
ijŠ£»Æѧъ¾æŠŌѧĻ°Š”×éÉč¼ĘČēĻĀŹµŃé·½°ø£¬²ā¶Ø·ÅÖĆ¼ŗ¾ĆµÄŠ”ĖÕ“ņѳʷ֊“æ¼īµÄÖŹĮæ·ÖŹż£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012ѧğŗŚĮś½¹ž¶ū±õŹŠµŚĮłÖŠŃ§øßČżÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©Ä³Š£»Æѧъ¾æŠŌѧĻ°Š”×éÉč¼ĘČēĻĀŹµŃé·½°ø£¬²ā¶Ø·ÅÖĆŅŃ¾ĆµÄŠ”ĖÕ“ņѳʷ֊“æ¼īµÄÖŹĮæ·ÖŹż”£
£Ø1£©·½°øŅ»£ŗ³ĘČ”Ņ»¶ØÖŹĮæµÄѳʷ£¬ÖĆÓŚŪįŪöÖŠ¼ÓČČÖĮŗćÖŲŗó£¬ĄäČ“£¬³ĘĮæŹ£Óą¹ĢĢåÖŹĮ棬¼ĘĖć”£ŹµŃéÖŠ¼ÓČČÖĮŗćÖŲµÄÄæµÄŹĒ ”£
£Ø2£©·½°ø¶ž£ŗ³ĘČ”Ņ»¶ØĮæѳʷ£¬ÖĆÓŚŠ”ÉÕ±ÖŠ£¬¼ÓŹŹĮæĖ®Čܽā£¬ĻņŠ”ÉÕ±ÖŠ¼ÓČė×ćĮæĀČ»Æ±µČÜŅŗ£¬¹żĀĖĻ“µÓ£¬øÉŌļ³Įµķ£¬³ĘĮæ¹ĢĢåÖŹĮ棬¼ĘĖć£ŗ
¢Ł¹żĀĖ²Ł×÷ÖŠ£¬³żĮĖÉÕ±”¢Ā©¶·Ķā»¹ÓƵ½µÄ²£Į§ŅĒĘ÷ÓŠ______________________£»
¢ŚŹŌŃéÖŠÅŠ¶Ļ³ĮµķŹĒ·ńĶźČ«µÄ·½·ØŹĒ_______________________________________
¢ŪČō¼ÓČėŹŌ¼ĮøÄĪŖĒāŃõ»Æ±µ£¬ŅŃÖŖ³ĘµĆѳʷ9.5g£¬øÉŌļµÄ³ĮµķÖŹĮæĪŖ19.7g£¬Ōņѳʷ֊Ģ¼ĖįÄʵÄÖŹĮæ·ÖŹżĪŖ_________________£Ø±£ĮōŅ»Ī»Š”Źż£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģŗŚĮś½¹ž¶ū±õŹŠøßČżÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©Ä³Š£»Æѧъ¾æŠŌѧĻ°Š”×éÉč¼ĘČēĻĀŹµŃé·½°ø£¬²ā¶Ø·ÅÖĆŅŃ¾ĆµÄŠ”ĖÕ“ņѳʷ֊“æ¼īµÄÖŹĮæ·ÖŹż”£
£Ø1£©·½°øŅ»£ŗ³ĘČ”Ņ»¶ØÖŹĮæµÄѳʷ£¬ÖĆÓŚŪįŪöÖŠ¼ÓČČÖĮŗćÖŲŗó£¬ĄäČ“£¬³ĘĮæŹ£Óą¹ĢĢåÖŹĮ棬¼ĘĖć”£ŹµŃéÖŠ¼ÓČČÖĮŗćÖŲµÄÄæµÄŹĒ ”£
£Ø2£©·½°ø¶ž£ŗ³ĘČ”Ņ»¶ØĮæѳʷ£¬ÖĆÓŚŠ”ÉÕ±ÖŠ£¬¼ÓŹŹĮæĖ®Čܽā£¬ĻņŠ”ÉÕ±ÖŠ¼ÓČė×ćĮæĀČ»Æ±µČÜŅŗ£¬¹żĀĖĻ“µÓ£¬øÉŌļ³Įµķ£¬³ĘĮæ¹ĢĢåÖŹĮ棬¼ĘĖć£ŗ
¢Ł¹żĀĖ²Ł×÷ÖŠ£¬³żĮĖÉÕ±”¢Ā©¶·Ķā»¹ÓƵ½µÄ²£Į§ŅĒĘ÷ÓŠ______________________£»
¢ŚŹŌŃéÖŠÅŠ¶Ļ³ĮµķŹĒ·ńĶźČ«µÄ·½·ØŹĒ_______________________________________
¢ŪČō¼ÓČėŹŌ¼ĮøÄĪŖĒāŃõ»Æ±µ£¬ŅŃÖŖ³ĘµĆѳʷ9.5g£¬øÉŌļµÄ³ĮµķÖŹĮæĪŖ19.7g£¬Ōņѳʷ֊Ģ¼ĖįÄʵÄÖŹĮæ·ÖŹżĪŖ_________________£Ø±£ĮōŅ»Ī»Š”Źż£©”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com