
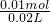 =0.5mol/L
=0.5mol/L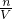 ����FeSO4��ҺŨ�ȣ�
����FeSO4��ҺŨ�ȣ�

 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ѧ��ȤС����100 mLij���ʵ���Ũ�ȵ�FeSO4��Һ�����Լ������������ɫ���Ƶ������������塢Ũ���ᡢϡ���ᡢKMnO4��Һ����ˮ��KSCN��Һ��ʯ���̪����ѧ��ѧ���û�ѧ������ʵ��������£� ��.����FeSO4��Һ
(1)���й���FeSO4��Һ���Ʋ�������ĺ���˳��Ϊ________(����ĸ)��
A����ʢ����ˮ���ձ��еμ�����ŨH2SO4�������Ȳ���ȴ������
B����ȡһ��������FeSO4������Ʒ
C������Ʒ���������Ƶ�ϡH2SO4�У��ò�������������Ʒ����ܽ�
D�����˺���Һת�Ƶ�����ƿ�У���ˮϡ����ָ���̶�
E��������������Һ�м����Թ��������ۣ���ֽ��裬����һ��ʱ��������������ð��Ϊֹ
(2)�ش��������⣺
�ٽ���Ʒ����ϡH2SO4������ֱ������ˮ��������__________________________��
������������������Һʱ����������۵�������__________________________��
��.�궨FeSO4��Һ��Ũ��
(1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______________���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ________mol��L��1��
(2)�ζ�ʱѡ��________(A.ʯ�B����̪��C������ָʾ��������ĸ)Ϊָʾ�����ζ��յ���ɫ�仯��_________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ������Э����߶���ѧ�����п��Ի�ѧ�Ծ� ���ͣ������
(15��)ij��ѧ��ȤС����100 mLij���ʵ���Ũ�ȵ�FeSO4��Һ�����Լ������������ɫ���Ƶ������������塢Ũ���ᡢϡ���ᡢKMnO4��Һ����ˮ��KSCN��Һ��ʯ���̪����ѧ��ѧ���û�ѧ������ʵ��������£�
��.����FeSO4��Һ
(1)���й���FeSO4��Һ���Ʋ�������ĺ���˳��Ϊ________(����ĸ)��
A����ʢ����ˮ���ձ��еμ�����ŨH2SO4�������Ȳ���ȴ������
B����ȡһ��������FeSO4������Ʒ
C������Ʒ���������Ƶ�ϡH2SO4�У��ò�������������Ʒ����ܽ�
D�����˺���Һת�Ƶ�����ƿ�У���ˮϡ����ָ���̶�
E��������������Һ�м����Թ��������ۣ���ֽ��裬����һ��ʱ��������������ð��Ϊֹ
(2)�ش��������⣺
�ٽ���Ʒ����ϡH2SO4������ֱ������ˮ��������_____________________________________________________________________��
������������������Һʱ����������۵������� __________________________________________________
__________________________________________________ ______________________��
______________________��
��.�궨FeSO4��Һ��Ũ��
( 1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______ __���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ______ __mol��L��1��
1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______ __���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ______ __mol��L��1��
(2)�ζ�ʱѡ��________(A.ʯ�B����̪��C������ָʾ��������ĸ)Ϊָʾ����������____________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��������һ�и߶���ѧ�ڵ����ν��Կ��Ի�ѧ�Ծ� ���ͣ�ʵ����
ij��ѧ��ȤС����100 mLij���ʵ���Ũ�ȵ�FeSO4��Һ�����Լ������������ɫ���Ƶ������������塢Ũ���ᡢϡ���ᡢKMnO4��Һ����ˮ��KSCN��Һ��ʯ���̪����ѧ��ѧ���û�ѧ������ʵ��������£�
��.����FeSO4��Һ
(1)���й���FeSO4��Һ���Ʋ�������ĺ���˳��Ϊ________(����ĸ)��
A����ʢ����ˮ���ձ��еμ�����ŨH2SO4�������Ȳ���ȴ������
B����ȡһ��������FeSO4������Ʒ
C������Ʒ���������Ƶ�ϡH2SO4�У��ò�������������Ʒ����ܽ�
D�����˺���Һת�Ƶ�����ƿ�У���ˮϡ����ָ���̶�
E��������������Һ�м����Թ��������ۣ���ֽ��裬����һ��ʱ��������������ð��Ϊֹ
(2)�ش��������⣺
�ٽ���Ʒ����ϡH2SO4������ֱ������ˮ��������__________________________��
������������������Һʱ����������۵�������__________________________��
��.�궨FeSO4��Һ��Ũ��
(1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______________���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ________mol��L��1��
(2)�ζ�ʱѡ��________(A.ʯ�B����̪��C������ָʾ��������ĸ)Ϊָʾ�����ζ��յ���ɫ�仯��_________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013������Э����߶���ѧ�����п��Ի�ѧ�Ծ� ���ͣ������
(15��)ij��ѧ��ȤС����100 mLij���ʵ���Ũ�ȵ�FeSO4��Һ�����Լ������������ɫ���Ƶ������������塢Ũ���ᡢϡ���ᡢKMnO4��Һ����ˮ��KSCN��Һ��ʯ���̪����ѧ��ѧ���û�ѧ������ʵ��������£�
��.����FeSO4��Һ
(1)���й���FeSO4��Һ���Ʋ�������ĺ���˳��Ϊ________(����ĸ)��
A����ʢ����ˮ���ձ��еμ�����ŨH2SO4�������Ȳ���ȴ������
B����ȡһ��������FeSO4������Ʒ
C������Ʒ���������Ƶ�ϡH2SO4�У��ò�������������Ʒ����ܽ�
D�����˺���Һת�Ƶ�����ƿ�У���ˮϡ����ָ���̶�
E��������������Һ�м����Թ��������ۣ���ֽ��裬����һ��ʱ��������������ð��Ϊֹ
(2)�ش��������⣺
�ٽ���Ʒ����ϡH2SO4������ֱ������ˮ��������_____________________________________________________________________��
������������������Һʱ����������۵�������
________________________________________________________________________��
��.�궨FeSO4��Һ��Ũ��
(1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______ __���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ______ __mol��L��1��
(2)�ζ�ʱѡ��________(A.ʯ�B����̪��C������ָʾ��������ĸ)Ϊָʾ����������____________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��������һ�и߶���ѧ�ڵ����ν��Կ��Ի�ѧ�Ծ� ���ͣ�ʵ����
ij��ѧ��ȤС����100 mLij���ʵ���Ũ�ȵ�FeSO4��Һ�����Լ������������ɫ���Ƶ������������塢Ũ���ᡢϡ���ᡢKMnO4��Һ����ˮ��KSCN��Һ��ʯ���̪����ѧ��ѧ���û�ѧ������ʵ��������£� ��.����FeSO4��Һ
(1)���й���FeSO4��Һ���Ʋ�������ĺ���˳��Ϊ________(����ĸ)��
A����ʢ����ˮ���ձ��еμ�����ŨH2SO4�������Ȳ���ȴ������
B����ȡһ��������FeSO4������Ʒ
C������Ʒ���������Ƶ�ϡH2SO4�У��ò�������������Ʒ����ܽ�
D�����˺���Һת�Ƶ�����ƿ�У���ˮϡ����ָ���̶�
E��������������Һ�м����Թ��������ۣ���ֽ��裬����һ��ʱ��������������ð��Ϊֹ
(2)�ش��������⣺
�ٽ���Ʒ����ϡH2SO4������ֱ������ˮ��������__________________________��
������������������Һʱ����������۵�������__________________________��
��.�궨FeSO4��Һ��Ũ��
(1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______________���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ________mol��L��1��
(2)�ζ�ʱѡ��________(A.ʯ�B����̪��C������ָʾ��������ĸ)Ϊָʾ�����ζ��յ���ɫ�仯��_________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com