
ij�Ȼ�����Ʒ��������FeCl2���ʡ���Ҫ�ⶨ������Ԫ�ص�����������ʵ�鰴���²�����У�
������������̣��ش��������⣺
��1������I��������Һ�����õ��IJ����������ձ����������⣬�������� �� .�����������ƣ�
��2�����в�������ʹ������ҺŨ��ƫС����________________����д��ţ���
��δϴ���ձ��Ͳ�����
�ڶ���ʱ��������ƿ�Ŀ̶���
������Һǰ����ƿ������������ˮ
��ҡ�Ⱥ���Һ����ڿ̶��ߺ������ˮ����Һ����̶�������
��3����д��������ˮ���������ӷ���ʽ ��
��4����������Ƿ��Ѿ�ϴ�Ӹɾ��IJ����� ��
��5����ԭ��Ʒ����aΪ50g�����Ⱥ����ɫ��������bΪ3g������Ʒ����Ԫ�ص����������� ��
��1��250mL����ƿ��1�֣�����ͷ�ιܣ�1�֣�
��2���٢� ��2�֣�
��3��2Fe 2+��Cl2��2Fe 3+�� 2Cl�� ��2�֣�
��4��ȡ����ϴ��Һ���μ�AgNO3��Һ�����������ɣ���֤��ϴ�Ӹɾ� ��2�֣�
��5����Ԫ�ص�����������42%
���������������1����ͼ��֪������I�ǽ��������ᷴӦ����Һϡ�ͳ�250.00mL��Һ������Ҫ250mL����ƿ����ͷ�ιܣ�
��2���٢�ƫС����ƫ�ߣ�����Ӱ��
��3������ˮ������+2������Ϊ+3�ۣ�������ӦΪ2Fe 2++Cl2=2Fe 3++2Cl-��
��4��ϴ�ӵ�������������������Һ�����Ȼ����Һ�����Լ���Cl���������Ƿ�ϴ�Ӹɾ���ȡ���һ��ϴ��Һ���μӵμ�AgNO3��Һ�����������ɣ���֤��ϴ�Ӹɾ���
��5����250ml��Һ��ȡ25ml��Һ����õ�����ɫ��������bΪ3g����ԭ����250ml��Һ�õ�����ɫ��������Ϊ30g,��������������������112/116������30g��������������112/116��30g. ��Ʒ����Ԫ�ص�����������112/116��30g/50g��100�G=42�G
���㣺��Һ���ƺͳ�������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʵ���Ҳⶨ̼������̼�����ƵĻ�����У�̼���Ƶ���������[�÷���w(Na2CO3)��ʾ]����ȡ�˻����5.lg������ˮ�У����250mL��Һ��
a��(10��)����һ����������w(Na2CO3)���û�ѧ��Ӧ��HCO3����CO32����ȫת��Ϊ��������ȡ�������������ɴ˼���������w (Na2CO3)��
��1����ȡ100 mL���ƺõ���Һ���ձ��У��μ�����������������Һ��HCO3����CO32����ȫת��Ϊ������Ӧѡ���Լ���___________ �����ţ���
| A��CaCl2 | B��MgSO4 | C����NaCI | D��Ba(OH)2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijͬѧ�������������������̼���Ʒ�ĩ�п��ܺ��������Ȼ��ƺ����������е�һ�ֻ��������ʡ�
(1)����1���貣��������______������3�IJ���������______��
(2)�Լ���̼���Ʒ�ĩ�п��ܺ��е���������������裺
����1��ֻ�����Ȼ��ƣ�
����2��ֻ����___________��
����3���Ȼ��ƺ��������ƶ����С�
(3)���ʵ�鷽��������ʵ�顣
��ѡ�����Լ����Ȼ�����Һ�����ᱵ��Һ����̪��Һ��ϡ���ᡢϡ���ᡢϡ���ᡢ��������Һ���ش��������⣺
�ټ�������Լ�A��������______��
������:
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����4: | |
| ����5: | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ҹ�����ר�Һ�°����ĺ����Ƽ�Ļ�ѧԭ���ǽ�������̼ͨ�백ˮ���Ȼ��Ʊ�����Һ�У��仯ѧ��Ӧ����ʽΪ��NaCl+NH3+CO2+H2O=NaHCO3��+NH4Cl��
��1����ʵ��������������ԭ���ӷ�Ӧ������Һ�з����̼�����ƾ��壬Ӧѡ������װ���е� ��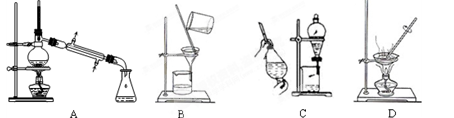
��2��ʵ������̼�����ƾ����У����ܺ��е�����������Cl����NH4+��ʵ���Ҽ���Cl����ѡ�õ��Լ��ǡ���������һ���������ӵķ����� ������ţ���
| A����ˮ����ʪ��ĺ�ɫʯ����ֽ�����Թܿ� |
| B��������������Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿ� |
| C��������������Һ�����ȣ������̪�Լ� |
| D��������������Һ�����ȣ�������ɫʯ���Լ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ѧ��ȤС���ͬѧΪ�ⶨijNa2CO3��NaCl�Ĺ���������Ʒ��Na2CO3��������������������ʵ�飬������벢��ɶ��й�����Ľ��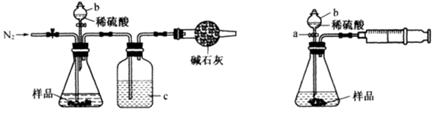
ͼ1 ͼ2
��1����ͬѧ��ͼ1��ʾװ�òⶨCO2��������ʵ��ʱϡ����������Ʒ�е� (�Na2CO3����"NaCl��)������Ӧ������b�������� ��ϴ��ƿc��ʢװ����Ũ���ᣬ��Ũ����������� ��
��2����ͬѧ��ͼ2��ʾװ�ã�ȡһ����������Ʒ(Ϊm g���Ѳ��)������ϡ���ᷴӦ����ʵ�飬�����Ʒ��Na2CO3�����������IJⶨ��
��ʵ��ǰ������װ�������Եķ������ȴ���a����bע��ˮ�����¶˲��������γ�һ��ˮ�����ٽ���Ͳ����������ѹ����b�¶˲������е� ��������װ�����������á�
����ʵ�����ʱ����ֱ�Ӳ�õ�������CO2�� (������������������)��
��3����ͬѧ����ͼ��ʾ�����Ͳ���ʵ�飺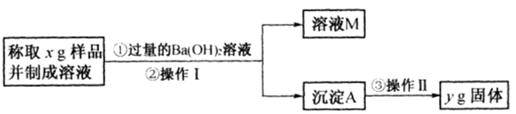
�ٲ���I�漰��ʵ�������� ��ϴ�ӣ��������漰��ʵ�������и�� ��
�ڱ���õ���Ʒ��Na2CO3���������ļ���ʽΪ ��
��4����״���£���672 mL CO2��ͨ��50 mL1mol/LKOH��Һ�У���ȫ��Ӧ��������Һ��K2CO3��KHCO3�����ʵ���֮��Ϊ(�跴Ӧǰ����Һ����仯���Բ���) ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ڳ����£�Fe��ˮ������Ӧ�����ڸ����£�Fe��ˮ�����ɷ�����Ӧ��Ӧ������װ�ã���Ӳ�ʲ������з��뻹ԭ���ۺ�ʯ���Ļ���ͨ��ˮ�����������ȣ��Ϳ�����ɸ����¡�Fe��ˮ�����ķ�Ӧʵ�顱��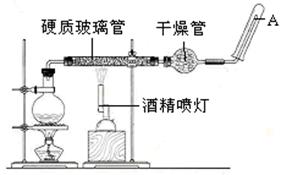
��ش��ʵ���е����⡣
��1��д���÷�Ӧ�ķ�Ӧ����ʽ�� ��
��2��ʵ��ǰ���������װ�ý��������Լ�飬���������� ��
��3��Բ����ƿ��ʢװ��ˮ����װ�����Ⱥ����Ҫ������ ����ƿ�ײ������˼�Ƭ���Ƭ�����Ƭ�������� ��
��4���ƾ��ƺ;ƾ���Ƶ�ȼ��˳�����ȵ�ȼ�ƾ��ƣ�����ˮ�������ٵ�ȼ�ƾ���ƣ�ԭ���� ��
��5���������ʢװ�ǵ������� ��
��6���Թ����ռ�������H2 �����Ҫ��A�������ܴ���ȼ�����壬�����Ը�������� ��
��һ������Ŀ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����Mg��CO2�ķ�Ӧ�Ʋ⣬NaҲ����CO2��ȼ�գ��ҹ���������ΪC��Na2O��Na2CO3�е����ֻ����֡�ij��ȤС��������װ�ÿ�չ�����ε�ʵ��̽����
��ʵ��I������CO2��Na��Ӧ����������Ϊ��
�ٰ�ͼ����װ�ã�����װ�������ԣ�������װ���м����Լ���
�ڴ�װ��a�ϻ���һ��ʱ�䣻
�۵�ȼd���ƾ��ƣ�ʹCO2��Na��ַ�Ӧ��ֹͣ���ȣ�����ͨ����ʹ˫ͨ����ȴ��
�ش��������⣺
��1��װ��a���õ��IJ����������Թܺ� ��
��2��װ��b��װ����Լ������������������������� ��
��3��װ��c������������������������������ ��
��4��������У����۲쵽������������������ ʱ�����ܽ��벽��ۡ�
��ʵ���̽����Ӧ���P��Ӧ������ȡ��Ӧ��˫ͨ���й�������29.2 g��������ʵ�飺
����ϸ�۲���壬�����к�ɫ������
�ڽ���������������ˮ�����ˡ�ϴ�ӣ��õ�1.8 g�����������
�۽���Һ��ˮϡ�����250 mL����Һ��
��ȡ�����۵���Һ���ȼ�����BaCl2��Һ���۲쵽��ɫ����,���ù�������Ϊ3.94g���ټӼ��η�̪��Һ����Һ��죻
��ȡ25.00 mL�۵���Һ���μӼ�����Ϊָʾ������3.0 mol��L-1����ζ��������������Ϊ20.00 mL��
�ش��������⣺
��5������жϢ��еĵζ��յ�����������������������
��6����Ӧ��װ��d�еĹ�������Ϊ���������������������� ��ͨ�����������29.2 g���������У�����ֵ��������Ƕ���?����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�̷���FeSO4��7H2O��������ȱ����ƶѪҩƷ����Ҫ�ɷ֡���������������м�����������������������ʣ�Ϊԭ�����������̷���һ�ַ�����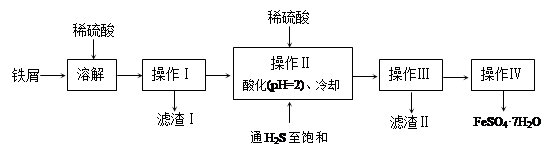
��֪�������±���H2S��Һ��pHԼΪ3.9��SnS������ȫʱ��Һ��pHΪ1.6��FeS��ʼ����ʱ��Һ��pHΪ3.0��������ȫʱ��pHΪ5.5��
��1������II�У�ͨ�����������͵�Ŀ���� _________________________ ������Һ���������ữ��pH��2��Ŀ���� _______________________________ ��
��2�������Ƶõ��̷��������Ƿ���Fe3+��ʵ������� ��
��3���ⶨ�̷���Ʒ��Fe2+�����ķ����ǣ�
a.��ȡ3.7200g�̷���Ʒ���ܽ⣬��250mL����ƿ�ж��ݣ�
b.��ȡ25.00mL������Һ����ƿ�У�
c.�������ữ��0.01000mol/LKMnO4��Һ�ζ����յ㣬����KMnO4��Һ�����ƽ��ֵΪ20.00mL��
����֪KMnO4������Һ��Fe2+��Ӧʱ����ԭΪMn2+����д���÷�Ӧ���� ��
���ڵζ�ʵ���в���ѡ�� ʽ�ζ��ܣ������� ��
�ۼ���������Ʒ��FeSO4��7H2O����������Ϊ ����С����ʾ��������λС������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ý����������������Ṥҵ�Ĵ�����ijͬѧ������������ַ����о�����ý����ɡ�
����һ��������װ�òⶨ����ý�ĺ�������ȷ������ɡ�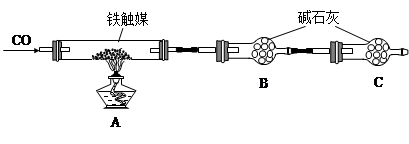
��������������ʵ�鷽���ⶨ����ý�ĺ�������ȷ������ɡ�
��1����������õ��IJ����������ձ�������������ͷ�ιܺ����� �� ��
��2����Ϊͨ��Cl2������������ҺB���л����� ��Ӱ��ⶨ�����
��3����Ϊͨ��Cl2�����Ҽ�����в���֣�����ҺB���п��ܺ���Cl2�������ʵ�鷽������Cl2���������ʵ�鱨�档
��ѡ�Լ���0.1mol��L��1����KMnO4��Һ����ɫʯ����Һ��Ʒ��ϡ��Һ������-KI��Һ��0.1moL��L��1KSCN��Һ
| ʵ����� | ʵ����������� |
| | |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com