
Ρ≥ΙΛ“ΒΖœΥ°ΫωΚ§œ¬±μ÷–ΒΡΡ≥–©άκΉ”Θ§«“Ης÷÷άκΉ”ΒΡΈο÷ ΒΡΝΩ≈®Ε»œύΒ»Θ§ΨυΈΣ0.1 mol/L(¥Υ ΐ÷ΒΚω¬‘Υ°ΒΡΒγάκΦΑάκΉ”ΒΡΥ°Ϋβ)ΓΘ
| ―τάκΉ” | KΘΪΓΓAgΘΪΓΓMg2ΘΪΓΓCu2ΘΪΓΓAl3ΘΪΓΓNH4+ |
| “θάκΉ” | ClΘ≠ΓΓCO32ΓΣΓΓNO3ΓΣΓΓSO42ΓΣΓΓIΘ≠ |

| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
AΓΔBΓΔCΓΔDΥΡ÷÷Ω…»ήΒΡΜ·ΚœΈο(ΥυΚ§άκΉ”Ης≤ΜœύΆ§)Θ§Ζ÷±π”…―τάκΉ”NaΘΪΓΔMg2ΘΪΓΔAl3ΘΪΓΔBa2ΘΪΚΆ“θάκΉ”OHΘ≠ΓΔClΘ≠ΓΔ ΓΔ
ΓΔ ΝΫΝΫΉιΚœΕχ≥…ΓΘΈΣΝΥ»ΖΕ®’βΥΡ÷÷Μ·ΚœΈοΒΡ≥…Ζ÷Θ§Ρ≥Ά§―ßΫχ––ΝΥ»γœ¬ Β―ι≤ΌΉςΘΚ
ΝΫΝΫΉιΚœΕχ≥…ΓΘΈΣΝΥ»ΖΕ®’βΥΡ÷÷Μ·ΚœΈοΒΡ≥…Ζ÷Θ§Ρ≥Ά§―ßΫχ––ΝΥ»γœ¬ Β―ι≤ΌΉςΘΚ
ΔΌΫΪΥΡ÷÷Μ·ΚœΈοΗς»Γ ΝΩ≈δ≥…»ή“ΚΘ§Ζ÷±πΉΑ»κΥΡ÷ß ‘ΙήΓΘ
ΔΎ»ΓA»ή“ΚΖ÷±πΒΈ»κΝμΆβ»ΐ÷÷»ή“Κ÷–Θ§Φ«¬Φ Β―ιœ÷œσ»γœ¬ΘΚ
B»ή“ΚΑΉ…Ϊ≥ΝΒμ≥ΝΒμ≤Μ»ήΫβ
C»ή“ΚΑΉ…Ϊ≥ΝΒμ≥ΝΒμ≤Μ»ήΫβ
D»ή“ΚΑΉ…Ϊ≥ΝΒμ≥ΝΒμ≤ΩΖ÷»ήΫβ
ΔέœρB»ή“Κ÷–ΒΈ»κD»ή“ΚΘ§ΈόΟςœ‘ Β―ιœ÷œσΓΘ
«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
(1)–¥≥ωΥϋΟ«ΒΡΜ·―ß ΫΘΚ
A ΘΜB ΘΜC ΘΜD ΘΜ
(2)…œ ωΔΎΒΡΒΎ»ΐ¥Έ Β―ιΘ§‘ΌΦ”»κAΘ§≥ΝΒμ≤ΩΖ÷»ήΫβΒΡάκΉ”ΖΫ≥Χ Ϋ ΓΘ
(3)Δέ÷–»τœρC»ή“Κ÷–ΒΈ»κD»ή“ΚΘ§Ω…Ρή≥ωœ÷ΒΡ Β―ιœ÷œσ « ΓΘ”ΟάκΉ”ΖΫ≥Χ Ϋ±μ ΨΤδ‘≠“ρΘΚ ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
―θΜ·ΜΙ‘≠Ζ¥”Π÷– ΒΦ …œΑϋΚ§―θΜ·ΚΆΜΙ‘≠ΝΫΗωΙΐ≥ΧΘ§œ¬Οφ «“ΜΗωΜΙ‘≠Ιΐ≥ΧΒΡΖ¥”Π ΫΘΚNO3Θ≠ΘΪ4HΘΪΘΪ3eΘ≠=NOΘΪ2H2OΘ§KMnO4ΓΔNa2CO3ΓΔCu2OΓΔFe2(SO4)3ΥΡ÷÷Έο÷ ÷–ΒΡ“Μ÷÷Έο÷ (ΦΉ)Ρή Ι…œ ωΜΙ‘≠Ιΐ≥ΧΖΔ…ζΓΘ
(1)–¥≥ω≤Δ≈δΤΫΗΟ―θΜ·ΜΙ‘≠Ζ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ________________________ΓΘ
(2)Ζ¥”Π÷–œθΥαΧεœ÷ΝΥ________ΓΔ________ΒΡ–‘÷ ΓΘ
(3)Ζ¥”Π÷–»τ≤ζ…ζ0.2 molΤχΧεΘ§‘ρΉΣ“ΤΒγΉ”ΒΡΈο÷ ΒΡΝΩ «________molΓΘ
(4)»τ1 molΦΉ”κΡ≥≈®Ε»œθΥαΖ¥”Π ±Θ§±ΜΜΙ‘≠œθΥαΒΡΈο÷ ΒΡΝΩ‘ωΦ”Θ§‘≠“ρ «_________________________________________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
(¥¥–¬‘Λ≤βΧβ)(1)ΔΌ‘ΎΒμΖέΒβΜ·ΦΊ»ή“Κ÷–Θ§ΒΈΦ”…ΌΝΩ¥Έ¬»ΥαΡΤΦν–‘»ή“ΚΘ§ΝΔΦ¥ΜαΩ¥ΒΫ»ή“Κ±δάΕ…ΪΘ§’β «“ρΈΣ________Θ§άκΉ”ΖΫ≥Χ ΫΈΣ__________________________ΓΘ
ΔΎ‘ΎΒβΚΆΒμΖέ–Έ≥…ΒΡάΕ…Ϊ»ή“Κ÷–Θ§ΒΈΦ”―«ΝρΥαΡΤΦν–‘»ή“ΚΘ§ΖΔœ÷άΕ…Ϊ÷πΫΞœϊ ßΘ§’β «“ρΈΣ______________________________ΓΘάκΉ”ΖΫ≥Χ Ϋ «_______________________________ΓΘ
ΔέΕ‘±»ΔΌΚΆΔΎ Β―ιΥυΒΟΒΡΫαΙϊΘ§ΫΪI2ΓΔClOΘ≠ΓΔSO42ΓΣ–‘”…«ΩΒΫ»θΒΡΥ≥–ρ≈≈Ν–ΈΣ_____________________________ΓΘ
(2)Ϋώ”–ΧζΤ§ΓΔΆ≠Τ§Θ§…ηΦΤ Β―ι÷ΛΟς“‘œ¬ ¬ ΒΘ§≤Δ–¥≥ωΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΓΘ
ΔΌ≈®ΝρΥαΒΡ―θΜ·–‘±»œΓΝρΥα«ΩΓΘ________________________________ΓΘ
ΔΎ¬»Μ·Χζ»ή“Κ÷–Fe3ΘΪΒΡ―θΜ·–‘±»ΝρΥαΆ≠»ή“Κ÷–ΒΡCu2ΘΪ«ΩΓΘ__________________________ΓΘ
ΔέΧζΒΡΜΙ‘≠–‘±»Ά≠«ΩΓΘ
___________________________________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
œ÷”–AΓΔBΓΔCΓΔDΓΔEΓΔFΝυ÷÷≥ΘΦϊΜ·ΚœΈοΘ§“―÷ΣΥϋΟ«ΑϋΚ§ΒΡ―τάκΉ””–KΘΪΓΔAgΘΪΓΔNaΘΪΓΔ
Ba2ΘΪΓΔFe2ΘΪΓΔAl3ΘΪΘ§“θάκΉ””–ClΘ≠ΓΔOHΘ≠ΓΔAlO2-ΓΔNO3-ΓΔSO42-ΓΔCO32-ΓΘΫΪΥϋΟ«Ζ÷±π≈δ≥…0.1 mol/LΒΡ»ή“ΚΫχ––»γœ¬ Β―ιΘΚ
ΔΌ≤βΒΟ»ή“ΚAΓΔCΓΔEΨυ≥ Φν–‘Θ§«“Φν–‘AΘΨEΘΨCΘ§EΒΡ―φ…Ϊ≥ «≥Ήœ…ΪΘ®ΆΗΙΐάΕ…Ϊνή≤ΘΝßΙέ≤λΘ©ΘΜΔΎœρB»ή“Κ÷–ΒΈΦ”œΓΑ±Υ°÷ΝΙΐΝΩΘ§œ»…ζ≥…≥ΝΒμΘ§Κσ≥ΝΒμ»Ϊ≤Ω»ήΫβΘΜΔέœρF»ή“Κ÷–ΒΈΦ”œΓœθΥαΘ§»ή“Κ±δ≥…ΉΊΜΤ…ΪΘ§«“”–Έό…ΪΤχΧε…ζ≥…ΘΜΔήœρD»ή“Κ÷–ΒΈΦ”BaΘ®NO3Θ©2»ή“ΚΈόΟςœ‘œ÷œσΓΘ
Θ®1Θ©–¥≥ωAΓΔDΓΔEΓΔFΒΡΜ·―ß ΫΘΚ
A________ΘΜD________ΘΜE________ΘΜF________ΓΘ
Θ®2Θ©”ΟάκΉ”ΖΫ≥Χ ΫΫβ ΆC»ή“Κ≥ Φν–‘ΒΡ‘≠“ρΘΚ__________________________________ΓΘ
Θ®3Θ©–¥≥ω Β―ιΔέ÷–Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΘΚ________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Ρ≥»ή“ΚΩ…ΡήΚ§”–NaΘΪΓΔKΘΪΓΔMg2ΘΪΓΔCu2ΘΪΒ»―τάκΉ”ΦΑMnO4ΓΣΓΔSiO32ΓΣΓΔAlO2ΓΣΓΔCO32ΓΣΓΔHCO3ΓΣΓΔSO42ΓΣΓΔClΘ≠Β»“θάκΉ”Θ§“―÷ΣΘΚΔΌΗΟ»ή“Κ≥ Έό…ΪΘΜΔΎΨ≠≤βΕ®»ή“ΚΒΡpHΘΫ12ΘΜΔέ»Γ…ΌΝΩ»ή“ΚΘ§Φ”»κ100 mL 2 molΓΛLΘ≠1œΓ―ΈΥαΫχ––ΥαΜ·Θ§”–ΑΉ…Ϊ≥ΝΒμ…ζ≥…Θ§ΜΙΒΟΒΫ“Μ÷÷Έό…ΪΈόΈΕΒΡΤχΧεΘ§ΗΟΤχΧε Ι≥Έ«ε ·Μ“Υ°(ΉψΝΩ)±δΜκΉ«ΓΘΕ‘ΥαΜ·ΚσΒΡ»ή“ΚΙΐ¬ΥΘ§ΒΟΒΫ¬Υ“ΚΦΉΓΘ
(1)”…ΔΌΔΎΔέΩ…≈–ΕœΘΚ‘≠»ή“Κ÷–“ΜΕ®≤Μ¥φ‘ΎΒΡάκΉ” «________Θ§“ΜΕ®¥φ‘ΎΒΡάκΉ” «________ΓΘ
(2)ΫΪ¬Υ“ΚΦΉΖ÷≥…ΝΫΒ»ΖίΘ§“ΜΖί÷–÷πΒΈΦ”»κΑ±Υ°ΓΔΉν÷’”–ΑΉ…ΪΫΚΉ¥≥ΝΒμΘ§ΥΒΟς‘≠»ή“Κ÷–“ΜΕ®”–________(ΧνάκΉ”ΖϊΚ≈)Θ§Η’ΩΣ ΦΦ”»κΑ±Υ° ±Θ§ΟΜ”–≥ΝΒμ≤ζ…ζΘ§‘≠“ρ «____________________________________(”ΟάκΉ”ΖΫ≥Χ Ϋ±μ Ψ)ΘΜΝμ“ΜΖί÷–Φ”»κΉψΝΩΒΡBa(NO3)2»ή“ΚΘ§”–ΑΉ…Ϊ≥ΝΒμ…ζ≥…Θ§ΥΒΟς‘≠»ή“Κ÷–“ΜΕ®”–________(ΧνάκΉ”ΖϊΚ≈)Θ§Ιΐ¬ΥΒΟΒΫ¬Υ“Κ““ΓΘ
(3)Άυ¬Υ“Κ““÷–Φ”»κΉψΝΩΒΡAgNO3»ή“ΚΘ§Ιΐ¬ΥΓΔœ¥Β”ΓΔΗ…‘οΒΟΙΧΧε26.5 gΘ§‘ρ‘≠»ή“Κ÷– «Ζώ”–ClΘ≠ΘΩ________(ΧνΓΑ «Γ±ΜρΓΑΖώΓ±)ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
ΉέΚœάϊ”ΟΉΣ¬·ΟΚΤχ[COΘ®60ΓΪ80%Θ©ΓΔCO2Θ®15ΓΪ20%Θ©ΦΑΈΔΝΩN2Β»]ΚΆΝρΥαΙΛ“ΒΈ≤Τχ÷–ΒΡSO2Θ§Φ»ΡήΨΜΜ·Έ≤ΤχΘ§”÷ΡήΜώΒΟ±Θœ’ΖέΘ®Na2S2O4Θ©Θ§Τδ≤ΩΖ÷ΙΛ“’Νς≥Χ»γœ¬ΘΚ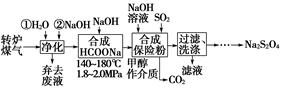
Θ®1Θ©ΉΣ¬·ΝΕΗ÷ ±Θ§¥φ‘ΎΖ¥”ΠΘΚFe3CΘ®sΘ©ΘΪCO2Θ®gΘ©??2COΘ®gΘ©ΘΪ3FeΘ®sΘ©Θ§ΤδΤΫΚβ≥Θ ΐ±μ¥ο ΫΈΣKΘΫ________ΓΘ
Θ®2Θ©ΟΚΤχΨΜΜ· ±Θ§œ»”ΟΥ°œ¥‘Ό”ΟNaOH»ή“Κœ¥Β”Θ§ΤδΡΩΒΡ «________ΓΘ
Θ®3Θ©¥”¬Υ“Κ÷–ΜΊ ’ΦΉ¥ΦΒΡ≤ΌΉςΖΫΖ® «____________________________ΘΜ
ΜΙΩ…ΜΊ ’ΒΡ―ΈάύΈο÷ «______________________________________Θ®÷Μ–¥“Μ÷÷Μ·―ß ΫΘ©ΓΘ
Θ®4Θ©Κœ≥…±Θœ’ΖέΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΈΣ_________________________ΓΘ
Θ®5Θ©±Θœ’ΖέΓΔH2O2ΨυΩ…”Ο”Ύ÷ΫΫ§Τ·ΑΉΘ§–¥≥ω±Θœ’Ζέ”κΙΐΝΩΒΡH2O2Θ§‘ΎΥ°»ή“Κ÷–Ζ¥”Π…ζ≥…ΝρΥα―ΈΒ»Έο÷ ΒΡάκΉ”ΖΫ≥Χ ΫΘΚ________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
δεΦΑΤδΜ·ΚœΈοΙψΖΚ”Π”Ο‘Ύ”–ΜζΚœ≥…ΓΔΜ·―ßΖ÷ΈωΒ»Νλ”ρΓΘ
Θ®1Θ©ΚΘΥ°ΧαδεΙΐ≥Χ÷–δε‘ΣΥΊΒΡ±δΜ·»γœ¬ΘΚ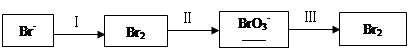
ΔΌΙΐ≥ΧΔώΘ§ΚΘΥ°œ‘Φν–‘Θ§ΒςΤδpHΘΦ3.5ΚσΘ§‘ΌΆ®»κ¬»ΤχΓΘ
ΔΓ.Ά®»κ¬»ΤχΚσΘ§Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ Ϋ «______ΓΘ
ΔΔ.ΒςΚΘΥ°pHΩ…ΧαΗΏCl2ΒΡάϊ”Ο¬ Θ§”ΟΤΫΚβ‘≠άμΫβ ΆΤδ‘≠“ρ «______ΓΘ
ΔΎΙΐ≥ΧΔρΘ§”Ο»»Ω’ΤχΫΪδεΗœ≥ωΘ§‘Ό”Ο≈®ΧΦΥαΡΤ»ή“ΚΈϋ ’ΓΘΆξ≥…≤Δ≈δΤΫœ¬Ν–ΖΫ≥Χ ΫΓΘ
Br2ΘΪ Na2CO3ΘΫ
Na2CO3ΘΫ NaBrO3ΘΪ
NaBrO3ΘΪ CO2ΘΪ
CO2ΘΪ ______
______
ΔέΙΐ≥ΧΔσΘ§”ΟΝρΥαΥαΜ·Ω…ΒΟBr2ΚΆNa2SO4ΒΡΜλΚœ»ή“ΚΓΘ
œύΆ§ΧθΦΰœ¬Θ§»τ”Ο―ΈΥαΥαΜ·Θ§‘ρΥυΒΟδεΒΡ÷ ΝΩΦθ…ΌΘ§‘≠“ρ «______ΓΘ
Θ®2Θ©NaBrO3 «“Μ÷÷Ζ÷Έω ‘ΦΝΓΘœρΝρΥαΥαΜ·ΒΡNaI»ή“Κ÷–÷πΒΈΦ”»κNaBrO3»ή“ΚΘ§Β±Φ”»κ2.6 mol NaBrO3 ±Θ§≤βΒΟΖ¥”ΠΚσ»ή“Κ÷–δεΚΆΒβΒΡ¥φ‘Ύ–Έ ΫΦΑΈο÷ ΒΡΝΩΖ÷±πΈΣΘΚ
| ΝΘΉ” | I2 | Br2 | IO3- |
| Έο÷ ΒΡΝΩ/mol | 0.5 | 1.3 | |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Ρ≥Na2CO3ΓΔNaAlO2ΒΡΜλΚœ»ή“Κ÷–÷πΒΈΦ”»κ1 molΓΛL-1ΒΡ―ΈΥα,≤βΒΟ»ή“Κ÷–ΒΡC ΓΔHC
ΓΔHC ΓΔAl
ΓΔAl ΓΔAl3+ΒΡΈο÷ ΒΡΝΩ”κΦ”»κ―ΈΥαΒΡΧεΜΐ±δΜ·ΙΊœΒ»γΆΦΥυ Ψ,‘ρœ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ® Θ©
ΓΔAl3+ΒΡΈο÷ ΒΡΝΩ”κΦ”»κ―ΈΥαΒΡΧεΜΐ±δΜ·ΙΊœΒ»γΆΦΥυ Ψ,‘ρœ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ® Θ©
AΘ°‘≠ΜλΚœ»ή“Κ÷–ΒΡC ”κAl ”κAl ΒΡΈο÷ ΒΡΝΩ÷°±»ΈΣ1ΓΟ2 ΒΡΈο÷ ΒΡΝΩ÷°±»ΈΣ1ΓΟ2 |
| BΘ°V1ΓΟV2=1ΓΟ5 |
| CΘ°MΒψ ±…ζ≥…ΒΡCO2ΈΣ0.05 mol |
DΘ°aœΏ±μ ΨΒΡάκΉ”ΖΫ≥Χ ΫΈΣ:Al +H++H2O +H++H2O Al(OH)3Γΐ Al(OH)3Γΐ |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΑΌΕ»÷¬–≈ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com