
��ҵ��������淋�����ͼ����ͼ����ش�
��1����֪ ��N2(g)+3H2(g)![]() 2NH3(g) ��H����92 kJ?mol��1��
2NH3(g) ��H����92 kJ?mol��1��
����500�桢2.02��107Pa��������������һ�ܱ������г���1molN2��3molH2,��ַ�Ӧ�ų�������______(�<����>����=��)92.4kJ��������________ _________��
��Ϊ��Ч���������ת���ʣ�ʵ���������˲�ȡ�Ĵ�ʩ��____________
A.�����¶� B.���ʺϴ������Ե��ʵ����� C.����ѹǿ
D.����ѹǿ E.ѭ�����úͲ��ϲ��䵪�� F.��ʱ�Ƴ���
��2�� ��֪����Ͻ���δԤ��Ҳ�ᷢ�ȡ�д�����������Ļ�ѧ����ʽ��_____________________________________________ _____________________��
�÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽK=___________�����¶�����ʱ��Kֵ____������С����Ӱ�죩��
 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
| ||
| �� |
| ||
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| �� |
| ||
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
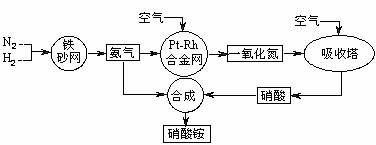
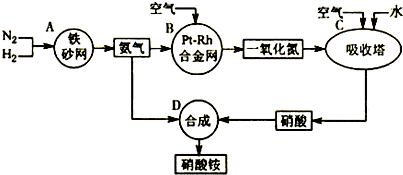
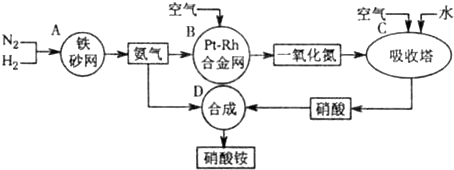
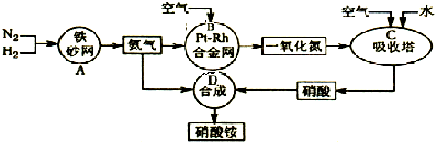
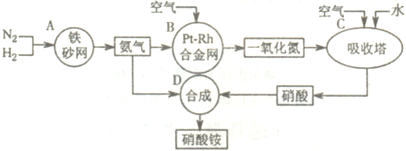
��10�֣���ͼ�ǹ�ҵ��������淋����̡�
��1��������C��ͨ�������Ŀ���� ��
A��B��C��D�ĸ������еķ�Ӧ������������ԭ��Ӧ���� ������ĸ����
��2����֪��4NH3��g��+3O2��g��=2N2��g��+6H2O��g�� ��H = ��1266��8kJ��mol
N2��g��+O2��g��=2NO��g�� ��H= +180��5 kJ��mol
д�������´��������Ȼ�ѧ����ʽ�� ������������Ӧ�Ļ�ѧƽ�ⳣ������ʽK= ��
��3����֪�� ��H= �� 92 kJ��mol��Ϊ���������ת���ʣ��˲�ȡ�Ĵ�ʩ�� ��������ĸ��
A�������¶� B��ʹ�ô��� C������ѹǿ
D��ѭ�����úͲ��ϲ��䵪�� E����ʱ�Ƴ���
��4����һ���¶Ⱥ�ѹǿ�£���H2��N2��3��1������ȣ����ܱ������л�ϣ����÷�Ӧ�ﵽƽ��ʱ�����ƽ��������NH3�������������Ϊ17��6������ʱH2��ת����Ϊ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com