




| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| Ʒ������ | Ũ������֭ |
| �䡡���� | ��Ũ������֭ ������ �����ʻ� ��ɽ����� ��ˮ |
| ��֭���� | ��80% |
| �������� | ���ڰ�װ��ڴ� |
| ���� |
| ���� |
| ���� |
| ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ѧ�뼼����
20-1����6�֣�����Ϊ����ѡ���⣬ȫѡ�Ե�6�֣�©ѡ���������֣���ѡ���ѡΪ0�֣������йع�ҵ������������ȷ���� �� ��
A���ϳɰ����������н�NH3Һ�����룬�������N2��H2ת����
B�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������SO3ת��ΪH2SO4ʱ�ų�������
C����ⱥ��ʳ��ˮ���ռ�������ӽ���Ĥ�����ɷ�ֹ�����Ҳ�����Cl2����������
D����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С
E������������̼������������������������ã��������CO������
20-2����14�֣�����ʡ�Ǻ����ʡ����ˮ�����౦�����Ȼ��Դ���Ӻ�ˮ�п�����ȡ���ֻ���ԭ�ϣ���ͼ��ij�����Ժ�ˮ��Դ�ۺ����õ�ʾ��ͼ��
��1�����оٺ�ˮ���������ַ��� �� ��
��2����������ѻ��Br2����������ֽ�Br2��ԭΪBr -����Ŀ���ǣ� ��
�������SO2ˮ��Һ����Br2�������ʿɴ�95%���йط�Ӧ�����ӷ���ʽ
��3������ȡ��ˮ�Ȼ�þʱ��Ҫ�ڸ����HCl�����м���MgCl2��6H2O��ԭ��
��4�������ˮ�Ȼ�þ���õ�þ�������ض��Ļ�������ȴ��Ϊ����þ�����������п���
����þ��������ȴ�����ǣ� ��
A��H2 B��CO2
C������ D��O2 E��ˮ����
��5���Ӹ��־�Ӫ�ۺϿ�����������ǿ���ִ���ҵ�Լ�������������Ҫ��ԭ�ϵȷ�����
��������Ϊ�û����������������ӵ�������Ŀ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꺣��ʡ������У������ѧ���� ���ͣ������
����ѧ�뼼����
20-1����6�֣�����Ϊ����ѡ���⣬ȫѡ�Ե�6�֣�©ѡ���������֣���ѡ���ѡΪ0�֣������йع�ҵ������������ȷ���� �� ��
A���ϳɰ����������н�NH3Һ�����룬�������N2��H2ת����
B�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������SO3ת��ΪH2SO4ʱ�ų�������
C����ⱥ��ʳ��ˮ���ռ�������ӽ���Ĥ�����ɷ�ֹ�����Ҳ�����Cl2����������
D����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С
E������������̼������������������������ã��������CO������
20-2����14�֣�����ʡ�Ǻ����ʡ����ˮ�����౦�����Ȼ��Դ���Ӻ�ˮ�п�����ȡ���ֻ���ԭ�ϣ���ͼ��ij�����Ժ�ˮ��Դ�ۺ����õ�ʾ��ͼ��
��1�����оٺ�ˮ���������ַ��� �� ��
��2����������ѻ��Br2����������ֽ�Br2��ԭΪBr -����Ŀ���ǣ� ��
�������SO2ˮ��Һ����Br2�������ʿɴ�95%���йط�Ӧ�����ӷ���ʽ
��3������ȡ��ˮ�Ȼ�þʱ��Ҫ�ڸ����HCl�����м���MgCl2��6H2O��ԭ��
��4�������ˮ�Ȼ�þ���õ�þ�������ض��Ļ�������ȴ��Ϊ����þ�����������п���
����þ��������ȴ�����ǣ� ��
A��H2 B��CO2
C������ D��O2 E��ˮ����
��5���Ӹ��־�Ӫ�ۺϿ�����������ǿ���ִ���ҵ�Լ�������������Ҫ��ԭ�ϵȷ�����
��������Ϊ�û����������������ӵ�������Ŀ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꺣��ʡ������У������ѧ���� ���ͣ������
����ѧ�뼼����
20-1����6�֣�����Ϊ����ѡ���⣬ȫѡ�Ե�6�֣�©ѡ���������֣���ѡ���ѡΪ0�֣������йع�ҵ������������ȷ���� �� ��
A���ϳɰ����������н�NH3Һ�����룬�������N2��H2ת����
B�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������SO3ת��ΪH2SO4ʱ�ų�������
C����ⱥ��ʳ��ˮ���ռ�������ӽ���Ĥ�����ɷ�ֹ�����Ҳ�����Cl2����������
D����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С
E������������̼������������������������ã��������CO������
20-2����14�֣�����ʡ�Ǻ����ʡ����ˮ�����౦�����Ȼ��Դ���Ӻ�ˮ�п�����ȡ���ֻ���ԭ�ϣ���ͼ��ij�����Ժ�ˮ��Դ�ۺ����õ�ʾ��ͼ��
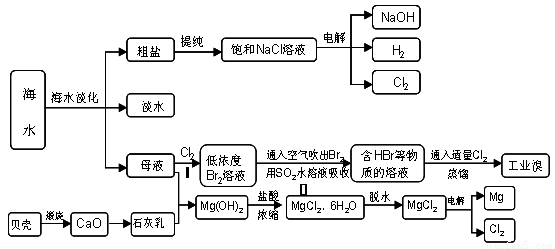
��1�����оٺ�ˮ���������ַ��� �� ��
��2����������ѻ��Br2����������ֽ�Br2��ԭΪBr -����Ŀ���ǣ� ��
�������SO2ˮ��Һ����Br2�������ʿɴ�95%���йط�Ӧ�����ӷ���ʽ
��3������ȡ��ˮ�Ȼ�þʱ��Ҫ�ڸ����HCl�����м���MgCl2��6H2O��ԭ��
��4�������ˮ�Ȼ�þ���õ�þ�������ض��Ļ�������ȴ��Ϊ����þ�����������п���
����þ��������ȴ�����ǣ� ��
A��H2 B��CO2
C������ D��O2 E��ˮ����
��5���Ӹ��־�Ӫ�ۺϿ�����������ǿ���ִ���ҵ�Լ�������������Ҫ��ԭ�ϵȷ�����
��������Ϊ�û����������������ӵ�������Ŀ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
����ѧ�뼼����
20-1����6�֣�����Ϊ����ѡ���⣬ȫѡ�Ե�6�֣�©ѡ���������֣���ѡ���ѡΪ0�֣������йع�ҵ������������ȷ���� �� ��
A���ϳɰ����������н�NH3Һ�����룬�������N2��H2ת����
B�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������SO3ת��ΪH2SO4ʱ�ų�������
C����ⱥ��ʳ��ˮ���ռ�������ӽ���Ĥ�����ɷ�ֹ�����Ҳ�����Cl2����������
D����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С
E������������̼������������������������ã��������CO������
20-2������ʡ�Ǻ����ʡ����ˮ�����౦�����Ȼ��Դ���Ӻ�ˮ�п�����ȡ���ֻ���ԭ�ϣ���ͼ��ij�����Ժ�ˮ��Դ�ۺ����õ�ʾ��ͼ��

��1�����оٺ�ˮ���������ַ��� �� ��
��2����������ѻ��Br2����������ֽ�Br2��ԭΪBr -����Ŀ���ǣ� ��
�������SO2ˮ��Һ����Br2�������ʿɴ�95%���йط�Ӧ�����ӷ���ʽ
��3������ȡ��ˮ�Ȼ�þʱ��Ҫ�ڸ����HCl�����м���MgCl2��6H2O��ԭ��
��4�������ˮ�Ȼ�þ���õ�þ�������ض��Ļ�������ȴ��Ϊ����þ�����������п���
����þ��������ȴ�����ǣ� ��
A��H2 B��CO2
C������ D��O2 E��ˮ����
��5���Ӹ��־�Ӫ�ۺϿ�����������ǿ���ִ���ҵ�Լ�������������Ҫ��ԭ�ϵȷ�����
��������Ϊ�û����������������ӵ�������Ŀ�� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com