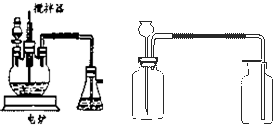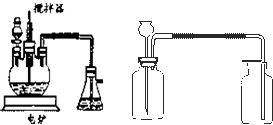
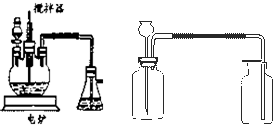
����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�
�����ף�ȡ2ҩ��ϸС������ͭ��������
�в�
�в�
��������������������������
������
������
����С���������Ȳ��ò�������ͣ���裬�����������
������
������
�н�����ȴ����ѡ�ú����������豸��գ��������в����ձ���ͨ������ԹܼС��������������ǣ���
�����ң�ȡ2ҩ�����������ͭ������С�ձ��У�����20mlŨ���ᣨ��������������98%�������ò��������裬����5min����ȥŨ���ᣬ����ˮ�Ҵ�ϴ�����Σ�������ֽ�����ɣ�
���������ۣ�
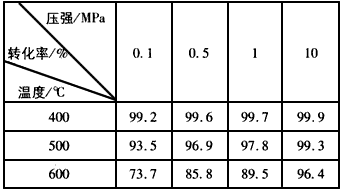
��1���������У������¶��Ը�ʱ����ֱ������ԭ����
CuSO
4?5H
2O
CuO+SO
3+5H
2O��CuSO
4?5H
2O
CuO+H
2SO
4+4H
2O
CuSO
4?5H
2O
CuO+SO
3+5H
2O��CuSO
4?5H
2O
CuO+H
2SO
4+4H
2O
���û�ѧ����ʽ��ʾ����
��2���������У�Ũ�����������
��ˮ��
��ˮ��
��Ϊ�˲��˷�ҩƷ������ˮ�Ҵ�ϴ��Һ���������ķ�����
��CaO������
��CaO������
�����õ���Ҫ����������������ƿ���¶ȼơ��нӹܣ�ţ�ǹܣ�����ƿ
�ƾ��ơ�������
�ƾ��ơ�������
�ȣ�
��3�����Ƶõ���ˮ����ͭ����ij˫��ˮ���Ƿ�ˮʱ�����˷��ֹ�������⣬�����ָ�˫��ˮ�������ݲ������Դ����кβ���
ͭ���ӣ�������ͭ����˫��ˮ�ֽ��д�����
ͭ���ӣ�������ͭ����˫��ˮ�ֽ��д�����
��
��4��Ŀǰ��ҵ��������̽����ŨHNO
3������������Cu��ŨH
2SO
4��ŨHNO
3��Ӧ����ȡ��Ъ���ȡ�����ŨHNO
3�ķ������Ʊ�CuSO
4?5H
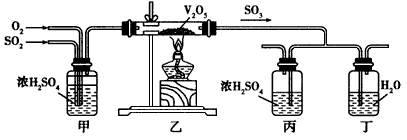
2O ���¹��գ�ģ���Ʊ�װ����ͼ��ʾ��
����һ��ͼװ���У���Һ©����װ��Һ����
Ũ����
Ũ����
����Ӧ����ʱ����ȡCuSO
4?5H
2O�IJ�����������
�ȳ�ȥ����
�ȳ�ȥ����
����
ֹͣ����
ֹͣ����
�����Ƚ�����ƿ�е�Һ�嵹���ձ�����ȴ����������CuSO
4?5H
2O�����ˡ����ɣ�
�������ҵ����ʯ��������β�������˷�ֹ������Ⱦ�⣬���ܵõ����о���ʵ�ü�ֵ�ĸ���Ʒ----������ƣ�β����������������ƵĻ�ѧ����ʽ�ǣ�
NO2+NO+Ca��OH��2=Ca��NO2��2+H2O ��4NO2+2Ca��OH��2=Ca��NO3��2+Ca��NO2��2+2H2O
NO2+NO+Ca��OH��2=Ca��NO2��2+H2O ��4NO2+2Ca��OH��2=Ca��NO3��2+Ca��NO2��2+2H2O
��
��������ʯ����ϡ�ͣ��ɵõ�����ʯ��ˮ�� ����ʯ��ˮ��CO
2�����ܹ�������ɫ������ijͬѧ������ͼ��ʾװ���Դ���ʯ��ϡ���ᷴӦ��ȡCO
2����ʦָ����ȡ���������壬��װ����Ҫ̫������ᣬ����˷ѣ���ͬѧ�Ը�װ��ij��λ����һ��С�Թܣ������������⣮����ѸĽ�����ͼ�к��ʵ�λ�ã�
��


 ����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�
����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�

 ����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�
����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�