
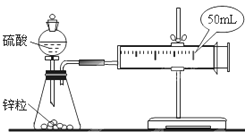
”¾ĢāÄæ”æĄūÓĆĻĀĶ¼×°ÖĆ²ā¶ØÖŠŗĶČȵďµŃé²½ÖčČēĻĀ£ŗ

¢ŁÓĆĮæĶ²ĮæČ”50 mL 0.25 mol/LĮņĖįµ¹ČėŠ”ÉÕ±ÖŠ£¬²ā³öĮņĖįČÜŅŗĪĀ¶Č£»
¢ŚÓĆĮķŅ»ĮæĶ²ĮæČ”50 mL 0.55 mol/L NaOHČÜŅŗ£¬²¢²ā³öĘäĪĀ¶Č£»
¢Ū½«NaOHČÜŅŗµ¹ČėŠ”ÉÕ±ÖŠ£¬Éč·ØŹ¹Ö®»ģŗĻ¾łŌČ£¬²ā³ö»ģŗĻŅŗ×īøßĪĀ¶Č”£
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)µ¹ČėNaOHČÜŅŗµÄÕżČ·²Ł×÷ŹĒ_________
A£®ŃŲ²£Į§°ō»ŗĀżµ¹Čė B£®·ÖČż“ĪÉŁĮæµ¹Čė C£®Ņ»“ĪŃøĖŁµ¹Čė
(2)Ź¹ĮņĖįÓėNaOHČÜŅŗ»ģŗĻ¾łŌȵÄÕżČ·²Ł×÷ŹĒ__________
A£®ÓĆĪĀ¶Č¼ĘŠ”ŠÄ½Į°č B£®½ŅæŖÓ²Ö½Ę¬ÓĆ²£Į§°ō½Į°č C£®ĒįĒįµŲÕńµ“ÉÕ± D£®ÓĆĢ×ŌŚĪĀ¶Č¼ĘÉĻµÄ»·ŠĪ²£Į§°ōÉĻĻĀĒįĒįµŲ³é¶Æ
(3)ŹµŃ鏿¾ŻČēĻĀ±ķ£ŗ
ĪĀ¶Č ŹµŃé“ĪŹż”” | ĘšŹ¼ĪĀ¶Čt1”ę | ÖÕÖ¹ĪĀ¶Čt2/”ę | ĪĀ¶Č²īĘ½¾łÖµ (t2£t1)/”ę | ||
H2SO4 | NaOH | Ę½¾łÖµ | |||
1 | 26.2 | 26.0 | 26.1 | 29.5 | |
2 | 27.0 | 27.4 | 27.2 | 32.3 | |
3 | 25.9 | 25.9 | 25.9 | 29.2 | |
4 | 26.4 | 26.2 | 26.3 | 29.8 | |
¢Łøł¾Ż±ķÖŠŹż¾Ż¼ĘĖć³öĄ“ĪĀ¶Č²īµÄĘ½¾łÖµĪŖ______”ę£»
¢Ś½üĖĘČĻĪŖ0.55 mol/L NaOHČÜŅŗŗĶ0.25 mol/LĮņĖįČÜŅŗµÄĆܶȶ¼ŹĒ1 g/cm3£¬ÖŠŗĶŗóÉś³ÉČÜŅŗµÄ±ČČČČŻc£½4.18 J/(g”¤”ę)”£ŌņÖŠŗĶČȦ¤H£½_______( Č”Š”ŹżµćŗóŅ»Ī»)”£
¢ŪÉĻŹöŹµŃ鏿ֵ½į¹ūÓėÖŠŗĶČČĪŖ57.3 kJ/molÓŠĘ«²ī£¬²śÉśĘ«²īµÄŌŅņæÉÄÜŹĒ____”£
a£®ŹµŃé×°ÖƱ£ĪĀ”¢øōČČŠ§¹ū²ī b£®ÓĆĪĀ¶Č¼Ę²ā¶ØNaOHČÜŅŗĘšŹ¼ĪĀ¶ČŗóÖ±½Ó²ā¶ØH2SO4ČÜŅŗµÄĪĀ¶Č c£®ĮæČ”NaOHČÜŅŗµÄĢå»żŹ±ŃöŹÓ¶ĮŹż d£®·Ö¶ą“Ī°ŃNaOHČÜŅŗµ¹ČėŹ¢ÓŠĮņĖįµÄŠ”ÉÕ±ÖŠ
”¾“š°ø”æC D 3.4 -56.8kJ/mol abcd
”¾½āĪö”æ
±¾ŹµŃéµÄÄæµÄŹĒ²ā¶ØÖŠŗĶČČ£¬ÖŠŗĶČČŹĒÖøŹĒĒæĖįŗĶĒæ¼īµÄĻ”ČÜŅŗĶźČ«·“Ӧɜ³É1molĖ®·Å³öµÄČČĮ棻±¾ŹµŃéÖŠŹ×ĻČ²ā¶ØĖįŗĶ¼ī·“Ó¦Ē°µÄĪĀ¶Č£¬Č»ŗó²ā¶Ø·“Ó¦ÖÕÖ¹ĪĀ¶Č£¬Č»ŗóĄūÓƱČČČČŻ½«ĪĀ¶Č×Ŗ»ÆĪŖČČĮæ¼ĘĖć³öÖŠŗĶČČ”£±¾ŹµŃéÖŠĪŖ±£Ö¤ĖįŗĶ¼īĶźČ«·“Ó¦£¬NaOH¹żĮ棻ŹµŃéµÄ¹Ų¼üŹĒŅŖ±£ĪĀ”£
(1)ĪŖĮĖ¼õÉŁČČĮæµÄÉ¢Ź§£¬ŹµŃé¹ż³ĢÖŠµ¹ČėNaOHČÜŅŗŹ±£¬±ŲŠėŅ»“ĪŃøĖŁµÄµ¹Čė£¬ĖłŅŌŃ”C£»
(2)ĪĀ¶Č¼ĘŹĒ²āĮæĪĀ¶ČµÄ£¬²»ÄÜŹ¹ÓĆĪĀ¶Č¼Ę½Į°č£»Ņ²²»ÄÜĒįĒįµŲÕńµ“ÉÕ±£¬·ńŌņæÉÄܵ¼ÖĀŅŗĢ彦³ö»ņČČĮæÉ¢Ź§£¬Ó°Ļģ²ā¶Ø½į¹ū£»øü²»ÄÜ“ņæŖÓ²Ö½Ę¬ÓĆ²£Į§°ō½Į°č£¬·ńŌņ»įÓŠČČĮæÉ¢Ź§£»Ź¹ĮņĖįÓėNaOHČÜŅŗ»ģŗĻ¾łŌȵÄÕżČ·²Ł×÷·½·ØŹĒ£ŗÓĆĢ×ŌŚĪĀ¶Č¼ĘÉĻµÄ»·ŠĪ²£Į§½Į°č°ōĒįĒįµŲ½Į¶Æ£¬ĖłŅŌŃ”D£»
(3)¢ŁĖÄ“ĪµÄĪĀ²ī·Ö±šĪŖ3.4”ę£¬5.1£¬3.3”ę£¬3.5”ę£¬µŚ2×鏿¾ŻĘ«²ī½Ļ“óÉįČ„£¬ĖłŅŌĘ½¾łĪĀ²īĪŖ![]() =3.4”ę£»
=3.4”ę£»
¢Ś50mL0.25mol/LĮņĖįÓė50mL0.55mol/LNaOHČÜŅŗ½ųŠŠÖŠŗĶ·“Ó¦£¬NaOH¹żĮ棬ĖłŅŌÉś³ÉĖ®µÄĪļÖŹµÄĮæĪŖ0.05L”Į0.25mol/L”Į2=0.025mol£¬ČÜŅŗµÄÖŹĮæĪŖ100ml”Į1g/cm3=100g£¬ĪĀ¶Č±ä»ÆµÄÖµ”÷T=3.4”ę£¬ŌņÉś³É0.025molĖ®·Å³öµÄČČĮæĪŖQ=mc”÷T=100g”Į4.18J/(g”ę)”Į3.4”ę=1421.2J£¬¼“1.4212kJ£¬ĖłŅŌŹµŃé²āµĆµÄÖŠŗĶČČ”÷H=-![]() =-56.8kJ/mol£»
=-56.8kJ/mol£»
¢ŪŹµŃé½į¹ūµÄ¾ų¶ŌÖµŠ”ÓŚÖŠŗĶČČµÄ¾ų¶ŌÖµ£»
a£®ŹµŃé×°ÖƱ£ĪĀ”¢øōČČŠ§¹ū²ī£¬ČČĮæÉ¢Ź§½Ļ“ó£¬ĖłµĆÖŠŗĶČČµÄ¾ų¶ŌֵʫŠ”£¬¹Źa·ūŗĻ£»
b£®ÓĆĪĀ¶Č¼Ę²ā¶ØNaOHČÜŅŗĘšŹ¼ĪĀ¶ČŗóÖ±½Ó²ā¶ØH2SO4ČÜŅŗµÄĪĀ¶Č£¬ĪĀ¶Č¼ĘÉĻÕ“ÓŠµÄNaOHÓėĮņĖį·“Ó¦·Å³öČČĮ棬Ź¹µĆ²ā¶ØµÄ³õŹ¼ĪĀ¶ČĘ«øߣ¬ĒŅŌģ³ÉČČĮæµÄÉ¢Ź§£¬²ā¶ØµÄĪĀ²īĘ«Š”£¬ÖŠŗĶČČµÄ¾ų¶ŌֵʫŠ”£¬¹Źb·ūŗĻ”£
c£®ĮæČ”NaOHČÜŅŗµÄĢå»żŹ±ŃöŹÓ¶ĮŹż£¬»įµ¼ÖĀĒāŃõ»ÆÄĘĢå»żĘ«“󣬵«ĮņĖįÉŁĮ棬ĖłŅŌÉś³ÉµÄĖ®µÄĪļÖŹµÄĮæ²»±ä£¬·Å³öµÄ×ÜČČĮæ²»±ä£¬¶ų¼ÓČėµÄNaOHČÜŅŗĢå»żĘ«“󣬻įŹ¹»ģŗĻŅŗµÄÖŹĮæĘ«“ó£¬Ōņ²āµĆµÄĪĀ²ī»įĘ«Š”£¬ÖŠŗĶČČµÄ¾ų¶ŌֵʫŠ”£¬¹Źc·ūŗĻ£»
d£®·Ö¶ą“Ī°ŃNaOHČÜŅŗµ¹ČėŹ¢ÓŠĮņĖįµÄŠ”ÉÕ±ÖŠ£¬ČČĮæÉ¢Ź§½Ļ“ó£¬ĖłµĆÖŠŗĶČČµÄ¾ų¶ŌֵʫŠ”£¬¹Źd·ūŗĻ£»
¹Ź“š°øĪŖ£ŗabcd£»


 ĆūŠ£æĪĢĆĻµĮŠ“š°ø
ĆūŠ£æĪĢĆĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijӊ»śĪļµÄ½į¹¹¼ņŹ½ČēĻĀ£¬ĻĀĮŠÓŠ¹ŲøĆÓŠ»śĪļµÄŠšŹöÖŠ²»ÕżČ·µÄŹĒ
![]()
A. Ęä·Ö×ÓŹ½ĪŖ C9H10O
B. ÄÜŹ¹ĖįŠŌøßĆĢĖį¼ŲČÜŅŗĶŹÉ«
C. Ņ»¶ØĢõ¼žĻĀ£¬1mol øĆÓŠ»śĪļĄķĀŪÉĻ×ī¶ąÄÜÓė 4mol H2 ·¢Éś¼Ó³É·“Ó¦
D. Ņ»¶ØĢõ¼žĻĀ£¬1mol øĆÓŠ»śĪļĄķĀŪÉĻ×ī¶ąÄÜÓė 4mol Br2 ·¢Éś¼Ó³É·“Ó¦
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijĶ¬Ń§½ųŠŠSO2µÄŠŌÖŹŹµŃ锣ŌŚµćµĪ°åa”¢b”¢c”¢d“¦·Ö±šµĪÓŠ²»Ķ¬µÄŹŌ¼Į£¬ŌŁĻņNa2SO3¹ĢĢåÉĻµĪ¼ÓŹżµĪÅØH2SO4ŗó£¬ŌŚÕūøöµćµĪ°åÉĻøĒÉĻÅąŃųĆó£¬Ņ»¶ĪŹ±¼äŗó¹Ū²ģµ½µÄŹµŃéĻÖĻóČē±ķĖłŹ¾”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ£Ø £©
ŠņŗÅ | ŹŌ¼Į | ŹµŃéĻÖĻó |
a | Ę·ŗģČÜŅŗ | ŗģÉ«ĶŹČ„ |
b | ĖįŠŌKMnO4ČÜŅŗ | ×ĻÉ«ĶŹČ„ |
c | NaOHČÜŅŗ£Øŗ¬2µĪ·ÓĢŖ£© | ŗģÉ«ĶŹČ„ |
d | H2SČÜŅŗ | »ĘÉ«»ė×Ē |
A.ŌŚÅØĮņĖįÓėNa2SO3¹ĢĢå·“Ó¦ÖŠ£¬ÅØĮņĖį±ķĻÖµÄĒæŃõ»ÆŠŌ
B.a”¢b¾ł±ķĆ÷SO2¾ßÓŠĘư׊Ō
C.cÖŠÖ»æÉÄÜ·¢Éś·“Ó¦£ŗSO2+2OH-=SO32-+H2O
D.dÖŠ±ķĆ÷SO2¾ßÓŠŃõ»ÆŠŌ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æAs2O3ŌŚŅ½Ņ©”¢µē×ÓµČĮģÓņÓŠÖŲŅŖÓ¦ÓĆ”£Ä³ŗ¬ÉéŌŖĖŲ£ØAs£©µÄ¹¤Ņµ·ĻĖ®¾ČēĶ¼1Į÷³Ģ×Ŗ»ÆĪŖ“Ö²śĘ·”£
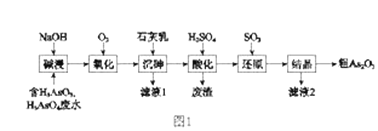
£Ø1£©”°¼ī½ž”±µÄÄæµÄŹĒ½«·ĻĖ®ÖŠµÄH3AsO3ŗĶH3AsO4×Ŗ»ÆĪŖŃĪ”£H3AsO4×Ŗ»ÆĪŖNa3AsO4·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ_______________________________”£
£Ø2£©”°Ńõ»Æ”±Ź±£¬1molAsO33-×Ŗ»ÆĪŖAsO43-ÖĮÉŁŠčŅŖO2______ mol”£
£Ø3£©”°³ĮÉé”±ŹĒ½«ÉéŌŖĖŲ×Ŗ»ÆĪŖCa5(AsO4)3OH³Įµķ£¬·¢ÉśµÄÖ÷ŅŖ·“Ó¦ÓŠ£ŗ
a£®Ca(OH)2£Øs£©![]() Ca2+£Øaq£©+2OH-£Øaq£© ”÷H£¼0
Ca2+£Øaq£©+2OH-£Øaq£© ”÷H£¼0
b£®5Ca2++OH-+3AsO43-![]() Ca5(AsO4)3OH ”÷H£¾0
Ca5(AsO4)3OH ”÷H£¾0
ŃŠ¾æ±ķĆ÷£ŗ”°³ĮÉé”±µÄ×ī¼ŃĪĀ¶ČŹĒ85”ę”£ ÓĆ»ÆŃ§Ę½ŗāŌĄķ½āŹĶĪĀ¶ČøßÓŚ85”ęŗó,ĖęĪĀ¶ČÉżøß³ĮµķĀŹĻĀ½µµÄŌŅņŹĒ_____________________”£
£Ø4£©”°»¹Ō”±¹ż³ĢÖŠH3AsO4×Ŗ»ÆĪŖH3AsO3£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ_______________________”£
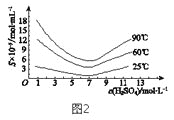
£Ø5£©”°»¹Ō”±ŗó¼ÓČČČÜŅŗ£¬H3AsO3·Ö½āĪŖAs2O3£¬Ķ¬Ź±½į¾§µĆµ½“ÖAs2O3”£As2O3ŌŚ²»Ķ¬ĪĀ¶ČŗĶ²»Ķ¬ÅضČĮņĖįÖŠµÄČܽā¶Č£ØS£©ĒśĻßČēĶ¼2ĖłŹ¾”£ĪŖĮĖĢįøß“ÖAs2O3µÄ³ĮµķĀŹ£¬”°½į¾§”±¹ż³Ģ½ųŠŠµÄ²Ł×÷ŹĒ_______”£
£Ø6£©ĻĀĮŠĖµ·ØÖŠ£¬ÕżČ·µÄŹĒ ______ £ØĢī×ÖÄø£©”£
a£®“ÖAs2O3ÖŠŗ¬ÓŠCaSO4
b£®¹¤ŅµÉś²śÖŠ£¬ĀĖŅŗ2æÉŃ»·Ź¹ÓĆ£¬ĢįøßÉéµÄ»ŲŹÕĀŹ
c£®ĶعżĻČ”°³ĮÉé”±ŗó”°Ėį»Æ”±µÄĖ³Šņ£¬æÉŅŌ“ļµ½ø»¼ÆÉéŌŖĖŲµÄÄæµÄ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æČēĶ¼ŹĒŅ»øö»Æѧ¹ż³ĢµÄŹ¾ŅāĶ¼”£
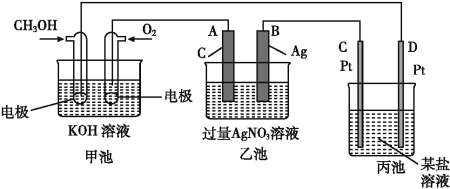
(1)C(Pt)µē¼«µÄĆū³ĘŹĒ____”£
(2)Š“³öĶØČėO2µÄµē¼«ÉĻµÄµē¼«·“Ó¦Ź½:_______________”£
(3)Š“³öĶØČėCH3OHµÄµē¼«ÉĻµÄµē¼«·“Ó¦Ź½:_________”£
(4)Čō±ū³ŲŹĒµē½ā±„ŗĶŹ³ŃĪĖ®ČÜŅŗ,ŌŚ____(Ģī”°Ńō¼«”±»ņ”°Ņõ¼«”±)ø½½üµĪČė·ÓĢŖČÜŅŗ±äŗģ”£
(5)ŅŅ³ŲÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ____”£
(6)µ±ŅŅ³ŲÖŠB(Ag)¼«µÄÖŹĮæŌö¼Ó5.40 gŹ±,¼×³ŲÖŠĄķĀŪÉĻĻūŗÄO2____mL(±ź×¼×“æöĻĀ);Čō±ū³ŲÖŠ±„ŗĶŹ³ŃĪĖ®ČÜŅŗµÄĢå»żĪŖ500 mL,µē½āŗó,ČÜŅŗµÄpH=_____”£(25 ”ę,¼ŁÉčµē½āĒ°ŗóČÜŅŗµÄĢå»żĪŽ±ä»Æ)”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æŌŚŅ»¶ØĢå»żµÄĆܱÕČŻĘ÷ÖŠ£¬½ųŠŠČēĻĀ»Æѧ·“Ó¦£ŗCO2(g)+H2(g)CO(g)+H2O(g)£¬Ęä»ÆŃ§Ę½ŗā³£ŹżKŗĶĪĀ¶ČtµÄ¹ŲĻµČē±ķ£ŗ
t/”ę | 700 | 800 | 830 | 1000 | 1200 |
K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)øĆ·“Ó¦µÄ»ÆŃ§Ę½ŗā³£Źż±ķ“ļŹ½ĪŖK=___”£ŅŃÖŖ£ŗK1000”ę£¾K800”ę£¬ŌņøĆ·“Ó¦ŹĒ__·“Ó¦”£(Ģī”°ĪüČČ”±»ņ”°·ÅČČ”±)£»
(2)ŅŃÖŖŌŚ800”ꏱ£¬øĆ·“Ó¦µÄĘ½ŗā³£ŹżK1=0.9£¬ŌņøĆĪĀ¶ČĻĀ·“Ó¦CO(g)£«H2O(g)![]() CO2(g)£«H2(g)µÄĘ½ŗā³£ŹżK2=___”£
CO2(g)£«H2(g)µÄĘ½ŗā³£ŹżK2=___”£
(3)ÄÜÅŠ¶ĻøĆ·“Ó¦ŹĒ·ń“ļµ½»ÆŃ§Ę½ŗāדĢ¬µÄŅĄ¾ŻŹĒ__”£
A£®ČŻĘ÷ÖŠŃ¹Ēæ²»±ä
B£®»ģŗĻĘųĢåÖŠc(CO)²»±ä
C£®vÕż(H2)=vÄę(H2O)
D£®c(CO2)=c(CO)
(4)ijĪĀ¶ČĻĀ£¬Ę½ŗāÅØ¶Č·ūŗĻĻĀŹ½£ŗc(CO2)c(H2)=c(CO)c(H2O)£¬ŹŌÅŠ¶Ļ“ĖŹ±µÄĪĀ¶ČĪŖ__”ę”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĮ½Ģ×ČēĶ¼ĖłŹ¾×°ÖĆ£¬·Ö±šĪŖ×°ÖĆ¢ŁŗĶ×°ÖĆ¢Ś£¬ø÷Ź¢ÓŠ2gŠæĮ£(æÅĮ£“óŠ”ĻąĶ¬)”£
ŹµŃé¢Ł£ŗŌŚ×°ÖĆ¢ŁÖŠ¼ÓČė40mL1mol/LµÄĮņĖį
ŹµŃé¢Ś£ŗŌŚ×°ÖĆ¢ŚÖŠ¼ÓČė40mL4mol/LµÄĮņĖį”£
±Č½Ļ¶žÕßŹÕ¼Æ10mLH2Ź±ĖłÓƵď±¼ä”£
(1)µ±ŹÕ¼Æµ½10mLH2Ź±£¬ÄÄøöŹµŃéĖłŗÄŹ±¼ä½Ļ³¤£æ__(ĢīŠ“ŹµŃéŠņŗÅ)ĪŖŹ²Ć“£æ__”£
(2)»īČūĶāŅʵÄĒéæöŹĒ__”£
A£®¾łŌČĶāŅĘ B£®ĻČæģŗóĀż C£®ĻČĀżŗóæģ D£®ĻČĀżŗóæģ£¬Č»ŗóÓÖÖš½„¼õĀż
ÄćŃ”ŌńµÄĄķÓÉŹĒ__”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĀĮ¼°Ęä»ÆŗĻĪļŌŚÉś²śÉś»īÖŠ¾ßÓŠÖŲŅŖµÄ×÷ÓĆ”£
(1)ĀĮŹōÓŚ»īĘĆ½šŹōČ“ÄÜŌŚæÕĘųÖŠĪČ¶Ø“ęŌŚ£¬ŌŅņŹĒ£ØÓĆ»ÆѧÓĆÓļ¼°Ļą¹ŲĪÄ×ÖĖµĆ÷£©___________
(2)ĀĮµē³ŲŠŌÄÜÓÅŌ½£¬ŌŚĻÖ“śÉś²ś”¢Éś»īÖŠÓŠ¹ć·ŗµÄÓ¦ÓĆ”£ĀĮ-æÕĘųµē³ŲŅŌĘä»·±£”¢°²Č«¶ųŹÜµ½Ō½Ą“Ō½¶ąµÄ¹Ų×¢£¬ĘäŌĄķČēĻĀĶ¼ĖłŹ¾”£
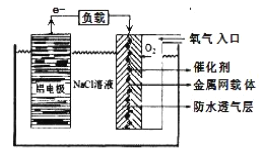
øƵē³ŲµÄÕż¼«·“Ó¦·½³ĢŹ½ĪŖ _____£»µē³ŲÖŠNaClČÜŅŗµÄ×÷ÓĆŹĒ ______£»ŅŌøƵē³ŲĪŖµēŌ“£¬ÓƶčŠŌµē¼«µē½āNa2SO4ČÜŅŗ£¬µ±Alµē¼«ÖŹĮæ¼õÉŁ1.8gŹ±£¬µē½ā³ŲŅõ¼«Éś³ÉµÄĘųĢåŌŚ±ź×¼×“æöĻĀµÄĢå»żĪŖ_______L”£
(3)AlCl3ÓėNaN3ŌŚøßĪĀĻĀ·“Ó¦æÉÖʵĆøßĪĀ½į¹¹ĢՓɵŖ»ÆĀĮ(AlN)£¬ĒŅÉś³ÉN2”£NaN3¾§ĢåÖŠŅõ”¢ŃōĄė×ÓøöŹż±ČĪŖ______£¬Š“³ö·“Ó¦»Æѧ·½³ĢŹ½ĪŖ___________
(4)Ķ¬Ö÷×åµÄŌŖĖŲÓ¦ÓĆ¹ć·ŗ”£2019Äź1ŌĀ3ČÕÉĻĪē£¬ęĻ¶šĖÄŗÅĢ½²āĘ÷ōęČ»ĀäŌĀ£¬Ź×“ĪŹµĻÖČĖĄą·ÉŠŠĘ÷ŌŚŌĀĒņ±³ĆęµÄČķ×ÅĀ½”£Ėł“īŌŲµÄ”°ÓńĶƶžŗÅ”±ŌĀĒņ³µ£¬ĶعżÉé»ÆļŲ£ØGaAs£©Ģ«ŃōÄܵē³ŲĢį¹©ÄÜĮæ½ųŠŠ¹¤×÷”£»Ų“šĻĀĮŠĪŹĢā£ŗ
¢Ł»łĢ¬GaŌ×Ó¼Ūµē×ÓÅŲ¼Ź½____£¬ŗĖĶāµē×ÓÕ¼¾Ż×īøßÄܼ¶µÄµē×ÓŌĘŠĪדĪŖ____£»»łĢ¬AsŌ×Ó×īøßÄܲćÉĻÓŠ____øöµē×Ó”£
¢ŚļŲŹ§Č„µē×ÓµÄÖš¼¶µēĄėÄܣص„Ī»£ŗkJ/mol£©µÄŹżÖµŅĄ“ĪĪŖ577”¢1985”¢2962”¢6192£¬-1ÓÉ“ĖæÉĶĘÖŖļŲµÄÖ÷ŅŖ»ÆŗĻ¼ŪĪŖ_____ŗĶ+3£¬ÉéµÄµŚŅ»µēĄėÄܱČļŲ_____Ģī”°“ó”±»ņ”°Š””±£©”£
¢ŪµŚĖÄÖÜĘŚŌŖĖŲÖŠ£¬Óė»łĢ¬AsŌ×ÓŗĖĶāĪ“³É¶Ōµē×ÓŹżÄæĻąĶ¬µÄŌŖĖŲ·ūŗÅĪŖ____”£
¢ÜÉé»ÆļŲæÉÓÉ£ØCH3£©3GaŗĶAsH3ŌŚ700”ęÖĘµĆ£¬£ØCH3£©3GaÖŠCŌ×ÓµÄŌӻƷ½Ź½ĪŖ ______£¬AsH3·Ö×ÓµÄæռ乹ŠĶĪŖ______”£
¢ŻĻąĶ¬Ń¹ĒæĻĀ£¬AsH3µÄ·Šµć_______NH3£ØĢī”°“óÓŚ”±»ņ”°Š”ÓŚ”±£©£¬ŌŅņĪŖ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ£Ø15·Ö£©ŗ¬Ģ¼ĪļÖŹµÄ¼ŪÖµŠĶ×Ŗ»Æ£¬ÓŠĄūÓŚ”°¼õĢ¼”±ŗĶæɳ֊ųŠŌ·¢Õ¹£¬ÓŠ×ÅÖŲŅŖµÄŃŠ¾æ¼ŪÖµ”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ŅŃÖŖCO·Ö×ÓÖŠ»Æѧ¼üĪŖC”ŌO”£Ļą¹ŲµÄ»Æѧ¼ü¼üÄÜŹż¾ŻČēĻĀ£ŗ
»Æѧ¼ü | H”ŖO | C”ŌO | C=O | H”ŖH |
E/(kJ”¤mol1) | 463 | 1075 | 803 | 436 |
CO(g)£«H2O(g)![]() CO2(g)£«H2(g) ¦¤H=___________kJ”¤mol1”£ĻĀĮŠÓŠĄūÓŚĢįøßCOĘ½ŗā×Ŗ»ÆĀŹµÄ“ėŹ©ÓŠ_______________£ØĢī±źŗÅ£©”£
CO2(g)£«H2(g) ¦¤H=___________kJ”¤mol1”£ĻĀĮŠÓŠĄūÓŚĢįøßCOĘ½ŗā×Ŗ»ÆĀŹµÄ“ėŹ©ÓŠ_______________£ØĢī±źŗÅ£©”£
a£®Ōö“óŃ¹Ēæ b£®½µµĶĪĀ¶Č
c£®ĢįøßŌĮĻĘųÖŠH2OµÄ±ČĄż d£®Ź¹ÓĆøߊ§“߻ƼĮ
£Ø2£©ÓƶčŠŌµē¼«µē½āKHCO3ČÜŅŗ£¬æɽ«æÕĘųÖŠµÄCO2×Ŗ»ÆĪŖ¼×Ėįøł(HCOO)£¬Č»ŗó½ųŅ»²½æÉŅŌÖʵĆÖŲŅŖÓŠ»ś»Æ¹¤ŌĮĻ¼×Ėį”£CO2·¢Éś·“Ó¦µÄµē¼«·“Ó¦Ź½ĪŖ________________£¬Čōµē½ā¹ż³ĢÖŠ×ŖŅĘ1 molµē×Ó£¬Ńō¼«Éś³ÉĘųĢåµÄĢå»ż£Ø±ź×¼×“æö£©ĪŖ_________L”£
£Ø3£©ŅŅ±½“ß»ÆĶŃĒāÖĘČ”±½ŅŅĻ©µÄ·“Ó¦ĪŖ£ŗ![]() (g)£«CO2(g)
(g)£«CO2(g)![]()
![]() (g)£«CO(g)£«H2O(g)£¬Ęä·“Ó¦Ąś³ĢČēĻĀ£ŗ
(g)£«CO(g)£«H2O(g)£¬Ęä·“Ó¦Ąś³ĢČēĻĀ£ŗ
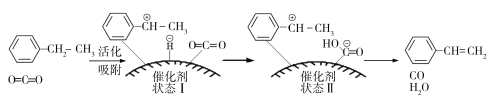
¢ŁÓÉŌĮĻµ½×“Ģ¬¢ń____________ÄÜĮæ£ØĢī”°·Å³ö”±»ņ”°ĪüŹÕ”±£©”£
¢ŚŅ»¶ØĪĀ¶ČĻĀ£¬ĻņŗćČŻĆܱÕČŻĘ÷ÖŠ³äČė2 molŅŅ±½ŗĶ2 mol CO2£¬ĘšŹ¼Ń¹ĒæĪŖp0£¬Ę½ŗāŹ±ČŻĘ÷ÄŚĘųĢå×ÜĪļÖŹµÄĮæĪŖ5 mol£¬ŅŅ±½µÄ×Ŗ»ÆĀŹĪŖ_______£¬ÓĆĘ½ŗā·ÖŃ¹“śĢęĘ½ŗāÅØ¶Č±ķŹ¾µÄ»ÆŃ§Ę½ŗā³£ŹżKp=_______”£[ĘųĢå·ÖŃ¹(p·Ö)=ĘųĢå×ÜŃ¹(p×Ü)”ĮĘųĢåĢå»ż·ÖŹż]
¢ŪŅŅ±½Ę½ŗā×Ŗ»ÆĀŹÓėp(CO2)µÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£¬Ēė½āŹĶŅŅ±½Ę½ŗā×Ŗ»ÆĀŹĖę×Åp(CO2)±ä»Æ¶ų±ä»ÆµÄŌŅņ________________________________”£

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com