
| A�� | 10 mL 0.5 mol•L-1 CH3COONa ��Һ�� 6 mL1 mol•L-1 �����ϣ�c��Cl-����c��Na+����c��CH3COO-����c��H+����c��OH-�� | |
| B�� | 0.1 mol•L-1pH Ϊ 4 �� NaHB ��Һ�У�c��HB-����c��H2B����c��B2-�� | |
| C�� | ���������Һ�еμ�������������Һǡ�ó����ԣ�c��Na+����c��SO42-����c��NH4+����c��OH-��=c��H+�� | |
| D�� | pH ��ȵģ�NH4��2SO4����NH4��2Fe��SO4��2�� NH4Cl ��Һ�У�c[��NH4��2SO4]��c[��NH4��2Fe��SO4��2]��c��NH4Cl�� |
���� A����Ӧ������Ϊ0.005molCH3COOH��0.005molNaCl��0.001molHCl��������Ũ��������Ϊ�����ӣ�������Ũ�ȴ��ڴ�������ӣ�
B��NaHB��Һ��pH=4��˵��HB-�ĵ���̶ȴ�����ˮ��̶ȣ���c��B2-����c��H2B����
C�������������NaOH�����ʵ������ʱ����Ӧ��������狀������ƣ���Һ�����ԣ���Ϊ���ԣ�������NaOH�����ʵ���Ӧ���Թ������ݴ��жϸ�����Ũ�ȴ�С��
D����������ˮ������ԣ���NH4��2Fe��SO4��2��Һ��Ũ���������狀��Ȼ����Һ����笠�����ˮ�����Һ�����ԣ�������Һ��笠�����Ũ����ͬ����϶���ѧʽ���ж���Ũ�ȴ�С��
��� �⣺A.10 mL 0.5 mol•L-1 CH3COONa ��Һ�� 6 mL1 mol•L-1 �����ϣ���Ӧ������Ϊ0.005molCH3COOH��0.005molNaCl��0.001molHCl������Ϊ���ᣬ����̶Ƚ�С����c��H+����c��CH3COO-������ȷ������Ũ�ȴ�СΪ����c��Cl-����c��Na+����c��H+����c��CH3COO-����c��OH-������A����
B.0.1 mol•L-1pH=4�� NaHB��Һ�����ԣ�˵��HB-�ĵ���̶ȴ�����ˮ��̶ȣ���c��B2-����c��H2B������ȷ������Ũ�ȴ�СΪ��c��HB-����c��B2-����c��H2B������B����
C�����������Һ�еμ�������������Һǡ�ó����ԣ������NaOH�����ʵ����Դ���c��Na+����c��SO42-����c��OH-��=c��H+��������笠����Ӳ���ˮ�⣬��c��SO42-����c��NH4+������Һ������Ũ�ȴ�СΪ��c��Na+����c��SO42-����c��NH4+����c��OH-��=c��H+������C��ȷ��
D��������Һ�������ԣ���NH4��2Fe��SO4��2��Һ����������Ҳˮ�����������ӣ�pH��ͬʱ��Ũ����С����NH4��2SO4��NH4Cl��Һ����笠�����ˮ��ʹ��Һ�����ԣ�����Һ��笠�����Ũ����ͬ����������淋Ļ�ѧʽ�к���2��笠����ӣ�������淋�Ũ�Ƚ�С������Һ��Ũ�ȴ�СΪ��c[��NH4��2SO4]��c��NH4Cl����c[��NH4��2Fe��SO4��2]����D����
��ѡC��
���� ���⿼������Ũ�ȴ�С�Ƚϣ���Ŀ�Ѷ��еȣ���ȷ�ε�ˮ��ԭ������Ӱ��Ϊ���ؼ���ע�������ж�����Ũ�ȴ�С�ķ������������������ѧ���ķ����������ۺ�Ӧ��������


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ѧ������Ӧ���Ͱ������Ϸ�Ӧ���ֽⷴӦ���û���Ӧ���ֽⷴӦ���� | |
| B�� | �������Ӫ�����ǵ����ʡ����ࡢ��֬��ά���ء����κ�ˮ | |
| C�� | ��ͬ��Ԫ����ɵ�����һ���Ǵ����� | |
| D�� | ��������ʱ����ʪë����ס�ڱǣ�������������ֳ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 1��1��1 | B�� | 2��3��4 | C�� | 6��4��3 | D�� | 6��3��4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����Ȳ��ܷ����ķ�Ӧһ�������ȷ�Ӧ | |
| B�� | ���ȷ�Ӧ������Ҫ���Ⱦ��ܷ��� | |
| C�� | ��ѧ��Ӧ�Ĺ�����һ�������ʱ仯������û�������仯 | |
| D�� | ��ѧ��Ӧ�Ƿ��Ȼ������ȣ�ȡ��ȡ��Ӧ����е�����������������е�����������Դ�С |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| ѡ�� | ʵ�����ģ�ʡ�Լг�װ�ã� | ��Ӧʵ�� |
| A | �ձ����������������� | CuSO4��Һ��Ũ���ᾧ |
| B | �ձ�������������ͷ�ιܡ���ֽ | �������ȥBaSO4��������BaCO3 |
| C | �ձ�������������ͷ�ιܡ�����ƿ | �ù���NaCl����0.5mol/L����Һ |
| D | �ձ�������������ͷ�ιܡ���Һ©�� | ����ˮ��CCl4��ȥNaBr��Һ��������NaI |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��С�մ�NaHCO3������θ�ᣨHC1�����ࣺCO32-+2H+�TCO2��+H2O | |
| B�� | �Ȼ�����Һ��ʴͭ����ӡˢ��·�壺Fe3++Cu�TFe2++Cu2+ | |
| C�� | ������þ�������Mg��OH��2+2H+�TMg2++2H2O | |
| D�� | Fe3+��I-��Ӧ�����ӷ���ʽΪ��Fe3++2I-�TFe2++I2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ˮ | B�� | ��ˮ | C�� | ˮ�� | D�� | ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���� | B�� | 1��2��3������ | C�� | ����� | D�� | 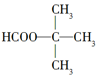 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
 ��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺
��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com