
��12�֣�����к͵ζ�����ѧ��ѧ����ʵ�顣
ijͬѧ���ⶨijŨ������Ʒ�����ʵ���Ũ�ȣ�����������ʵ�������
| A����ȴ�����º���100mL����ƿ�ж������100mLϡ���ᡣ |
| B����ȡ20.00mLϡ��������ƿ�в����뼸��ָʾ���� |
C������ʽ�ζ��ܺͼ�ʽ�ζ��� ������ˮϴ�Ӹɾ������ø���ʢ��Һ��ϴ�� ������ˮϴ�Ӹɾ������ø���ʢ��Һ��ϴ�� |
| D�������ʵ���Ũ��Ϊ1.50mol��L��1��NaOH��Һװ���ʽ�ζ��ܣ�����Һ����¶���V1�� |
| �ζ� ��� | ����ϡ���� �����(mL) | ������NaOH����ҺҺ�����(mL) | |
| V1 | V2 | ||
| �� | 20��00 | 0��50 | 22��60 |
| �� | 20��00 | 6��00 | 27��90 |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
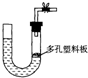
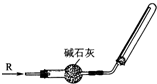
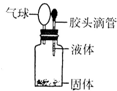
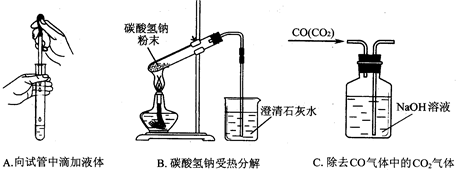
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
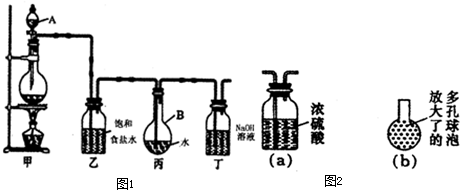
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��1��50mL 0.50mol/L������50mL 0.55mol/L NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺
��1��50mL 0.50mol/L������50mL 0.55mol/L NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺| �ζ����� | ������Һ�����mL�� | ������� | |
| �ζ�ǰ�Ŀ̶ȣ�mL�� | �ζ���Ŀ̶ȣ�mL�� | ||
| ��һ�� | 10.00 | 0.40 | 20.50 |
| �ڶ��� | 10.00 | 4.10 | 24.00 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��© | B�����ʵ��������60mL��ϡ�������Һ������ʱӦѡ��100mL����ƿ | C����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ�� | D���ü�����ָʾ�����ζ��յ�ʱ����Һ��ɫ�ӳ�ɫ��Ϊ��ɫ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com