
ѧϰ��ѧ��Ŀ�������û�ѧ֪ʶ�����ճ������е��������ͽ�����������е�ʵ�����⡣����˵��������Ϊ������ǣ� ��
A�����ڷ��õľƾ�û��ȼ�յ�ԭ����û�дﵽ�ƾ����Ż��
B���Ӻ�ˮ����ȡʳ�εĹ��̣�ʵ���Ͼ���������ˮ��ʳ�ξ���ᾧ�����Ĺ���
C�����죬ͭ���۾��ܱ�����ֵ���ɫ����[��Ҫ�ɷ�Cu2(OH)2CO3],����ϡ�����ȥ
D�������ܼӿ�������ˮ�е��ܽ⣬��ԭ�������������ǵ��ܽ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
��13�֣�
[̽��һ]Ϊ�ⶨ������FeTiO3�������������п���ȡ��������������ij�о���ѧϰС���������������ʵ��װ�ã���ע������ȡ��ָ���ǿɱ�H2��Cת��ΪH2O��CO2����ԭ�ӣ�
��1����֪FeTiO3��H2��ԭ������Fe��TiO2��ˮ��ѧ����������ͼ��1װ�ã�AΪ�����װ�ã�������������ȡ������д��FeTiO3�ڼ���ʱ��H2��ԭ�Ļ�ѧ����ʽ��
___________________________________________________________________
��2����֪FeTiO3��C��ԭ������Fe��TiO2��CO2��ѧ����������ͼ��2װ�ã�AΪ�����װ�ã�������������ȡ����������Ϊ�÷������������ڣ���Ӧ������CO2��ͨ����ɫֲ��Ĺ������ת��ΪӪ�����ʣ�ͬʱ����������
6CO2+6H2OC6H12O6�������ǣ�+6O2��
ʵ������У��Ƶ÷�Ӧǰװ��A�������������Ϊag��̿�۵�����Ϊbg����Ӧ��װ��B�в���CaCO3������Ϊcg�����������п���ȡ�������������ı���ʽΪ ��
��3����ר��������϶������������������壬��ʵ����Ʒ��滹���ڲ���֮��������ͼ��2װ���ڼ���ʱ�����ɵ�CO2������ȫ��������ʯ��ˮ���գ�ͬʱ�������̿���ڼ���ʱ�������CO����ʵ�������
�����ѧ���ҵ���������Ľ���ʩ������ͼ��1װ�ü����������̿�ۣ�����U��B֮ǰ����ʢ�� __________��ȼ�չܣ���B֮������ ��
�ڸĽ���ʵ����ʼ�����������װ��A��ͨ��N2��Ŀ���� ��
[̽����]����TiO2��Ϊһ�ֹ����Խ��Խ�ܵ����ǵĹ�ע�������㷺�������á�
��4��ij����С���ڲ�ͬ�����壨��Ƭ����Ƭ���մɣ������Ʊ��������ѱ�Ĥ�������첻ͬ����TiO2��Ĥ���ʹ������ɫ��ÿ�ι���20minȡһ������ʵ������ͼ��ʾ������˵����ȷ���� ����˫ѡ��
��a����ͬ���壬���ۺ����¶�һ������Ƭ���
��b��Լ��520��ʱ����Ƭ����Ĺ���������
��c�����ۺ������壬�������������¶ȵ����߶�����
��d����ͬ����TiO2��Ĥ�Ĺ�����Բ�ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��2011ѧ�긣��ʡ��Ϫһ�С�������ѧ�߶���ѧ����ĩ������ѧ�Ծ������� ���ͣ�ʵ����
��13�֣�
[̽��һ]Ϊ�ⶨ������FeTiO3�������������п���ȡ��������������ij�о���ѧϰС���������������ʵ��װ�ã���ע������ȡ��ָ���ǿɱ�H2��Cת��ΪH2O��CO2����ԭ�ӣ�
��1����֪FeTiO3��H2��ԭ������Fe��TiO2��ˮ��ѧ����������ͼ��1װ�ã�AΪ�����װ�ã�������������ȡ������д��FeTiO3�ڼ���ʱ��H2��ԭ�Ļ�ѧ����ʽ��
___________________________________________________________________
��2����֪FeTiO3��C��ԭ������Fe��TiO2��CO2��ѧ����������ͼ��2װ�ã�AΪ�����װ�ã�������������ȡ����������Ϊ�÷������������ڣ���Ӧ������CO2��ͨ����ɫֲ��Ĺ������ת��ΪӪ�����ʣ�ͬʱ����������
6CO2+6H2O  C6H12O6�������ǣ�+6O2��
C6H12O6�������ǣ�+6O2��
ʵ������У��Ƶ÷�Ӧǰװ��A�������������Ϊag��̿�۵�����Ϊbg����Ӧ��װ��B�в���CaCO3������Ϊcg�����������п���ȡ�������������ı���ʽΪ ��
��3����ר��������϶������������������壬��ʵ����Ʒ��滹���ڲ���֮��������ͼ��2װ���ڼ���ʱ�����ɵ�CO2������ȫ��������ʯ��ˮ���գ�ͬʱ�������̿���ڼ���ʱ�������CO����ʵ�������
�����ѧ���ҵ���������Ľ���ʩ������ͼ��1װ�ü����������̿�ۣ�����U��B֮ǰ����ʢ�� __________��ȼ�չܣ���B֮������ ��
�ڸĽ���ʵ����ʼ�����������װ��A��ͨ��N2��Ŀ���� ��
[̽����]����TiO2��Ϊһ�ֹ����Խ��Խ�ܵ����ǵĹ�ע�������㷺�������á�
��4��ij����С���ڲ�ͬ�����壨��Ƭ����Ƭ���մɣ������Ʊ��������ѱ�Ĥ�������첻ͬ����TiO2��Ĥ���ʹ������ɫ��ÿ�ι���20minȡһ������ʵ������ͼ��ʾ������˵����ȷ���� ����˫ѡ��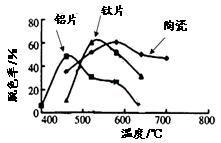
��a����ͬ���壬���ۺ����¶�һ������Ƭ���
��b��Լ��520��ʱ����Ƭ����Ĺ���������
��c�����ۺ������壬�������������¶ȵ����߶�����
��d����ͬ����TiO2��Ĥ�Ĺ�����Բ�ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�콭��ʡ�����и�����һ����Ͽ��Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
��16�֣����������(Na2S2O3)������Ҫ����������Σ��׳ơ������������������մ�������ˮ���������Ҵ�������֯��Ư�ס������������й㷺Ӧ�á�
ijУ��ѧ�о���ѧϰС���������ѧϰ��˼�룬ͨ��ʵ��̽��Na2S2O3�Ļ�ѧ���ʡ�
����Ʒ�Ʊ���ʵ�����г����������ƺ�����Ʊ�Na2S2O3��5H2O��д����Ӧ�Ļ�ѧ����ʽ ��
��ӦҺ����ɫ�����ˡ�Ũ���ᾧ�����ˡ�ϴ�ӡ����T�ò�Ʒ�����þ������Ҵ�ϴ�ӵ�Ŀ���� ��
��������⡿Na2S2O3�Ƿ���Na2SO4�߱����Ƶ����������أ�
����٣� ��
����ڣ���Һ�����ԣ��Ҳ����ᷴӦ��
����ۣ���ԭ�ԣ����ܱ�������������
������̽����������������ڡ��ۣ����ʵ�鷽����
|
|
ʵ����� |
ʵ������� Ԥ��ʵ������ |
������ͣ��� ���ӷ���ʽ��ʾ�� |
|
����� |
������ֽ�����ɫ������
|
��ҺpH=8 |
|
|
��pH=2�������� �μ�Na2S2O3��Һ |
|
2S2O32- +2H+ �T�T S��+ SO2 ��+ H2O |
|
|
����� |
��������ˮ�еμ�����Na2S2O3��Һ |
��ˮ��ɫ��dz |
|
��ʵ����ۡ�
Na2S2O3�����ᷴӦ�����л�ԭ�ԣ���Na2SO4�Ļ�ѧ���ʲ����ơ�
���������ۡ�
��1����ͬѧ��̽��������ۡ���Ӧ�����Һ�еμ���������Һ���۲쵽�а�ɫ�������������ݴ���Ϊ��ˮ�ɽ�Na2S2O3����������Ϊ�÷����Ƿ���ȷ��˵�����ɣ� ��
��2�����������һ��ʵ�鷽����֤��Na2S2O3���л�ԭ�ԡ����ʵ�鷽���ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�츣��ʡ�߶���ѧ����ĩ������ѧ�Ծ������� ���ͣ�ʵ����
��13�֣�
[̽��һ]Ϊ�ⶨ������FeTiO3�������������п���ȡ��������������ij�о���ѧϰС���������������ʵ��װ�ã���ע������ȡ��ָ���ǿɱ�H2��Cת��ΪH2O��CO2����ԭ�ӣ�

��1����֪FeTiO3��H2��ԭ������Fe��TiO2��ˮ��ѧ����������ͼ��1װ�ã�AΪ�����װ�ã�������������ȡ������д��FeTiO3�ڼ���ʱ��H2��ԭ�Ļ�ѧ����ʽ��
___________________________________________________________________
��2����֪FeTiO3��C��ԭ������Fe��TiO2��CO2��ѧ����������ͼ��2װ�ã�AΪ�����װ�ã�������������ȡ����������Ϊ�÷������������ڣ���Ӧ������CO2��ͨ����ɫֲ��Ĺ������ת��ΪӪ�����ʣ�ͬʱ����������
6CO2+6H2O
 C6H12O6�������ǣ�+6O2��
C6H12O6�������ǣ�+6O2��
ʵ������У��Ƶ÷�Ӧǰװ��A�������������Ϊag��̿�۵�����Ϊbg����Ӧ��װ��B�в���CaCO3������Ϊcg�����������п���ȡ�������������ı���ʽΪ ��
��3����ר��������϶������������������壬��ʵ����Ʒ��滹���ڲ���֮��������ͼ��2װ���ڼ���ʱ�����ɵ�CO2������ȫ��������ʯ��ˮ���գ�ͬʱ�������̿���ڼ���ʱ�������CO����ʵ�������
�����ѧ���ҵ���������Ľ���ʩ������ͼ��1װ�ü����������̿�ۣ�����U��B֮ǰ����ʢ�� __________��ȼ�չܣ���B֮������ ��
�ڸĽ���ʵ����ʼ�����������װ��A��ͨ��N2��Ŀ���� ��
[̽����]����TiO2��Ϊһ�ֹ����Խ��Խ�ܵ����ǵĹ�ע�������㷺�������á�
��4��ij����С���ڲ�ͬ�����壨��Ƭ����Ƭ���մɣ������Ʊ��������ѱ�Ĥ�������첻ͬ����TiO2��Ĥ���ʹ������ɫ��ÿ�ι���20minȡһ������ʵ������ͼ��ʾ������˵����ȷ���� ����˫ѡ��
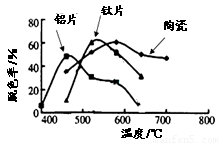
��a����ͬ���壬���ۺ����¶�һ������Ƭ���
��b��Լ��520��ʱ����Ƭ����Ĺ���������
��c�����ۺ������壬�������������¶ȵ����߶�����
��d����ͬ����TiO2��Ĥ�Ĺ�����Բ�ͬ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com