
����Ŀ������ʽΪC3H6O2���л����ж���ͬ���칹�壬�������е�����X��Y��Z��W�����ǵķ����о������������Ƿֱ��������ʵ���Լ�����ʵ���¼���£�
NaOH��Һ | ������Һ | ����Cu(OH)2 | ������ | |
X | �кͷ�Ӧ | ������ | �ܽ� | �������� |
Y | ������ | ������ | ���Ⱥ���ש��ɫ���� | �������� |
Z | ˮ�ⷴӦ | ������ | ���Ⱥ���ש��ɫ���� | ������ |
W | ˮ�ⷴӦ | ������ | ������ | ������ |
�ش��������⣺
��1��д��X�Ľṹ��ʽ________�� W��ϵͳ������________.
��2����Y��һ�������·��������ڵ���ˮ��Ӧ�Ļ�ѧ����ʽ_________________________
��Z��NaOH��Һ��Ӧ�Ļ�ѧ����ʽ_______________________________
���𰸡� CH3CH2COOH ������� ![]()
![]() CH2=CHCHO��H2O HCOOCH2CH3��NaOH
CH2=CHCHO��H2O HCOOCH2CH3��NaOH![]() HCOONa��CH3CH2OH
HCOONa��CH3CH2OH
������������ʽΪC3H6O2���л���X��Y��Z��W�����ǵķ����о���������X����NaOH�����кͷ�Ӧ�����ܽ�Cu(OH)2��˵������-COOH����XΪCH3CH2COOH��Y�ܷ���������Ӧ��˵������-CHO������Na��������������˵������-OH��YΪCH3CHOHCHO��Z�ܷ���ˮ�⣬���ܷ���������Ӧ��˵������-OOCH��Ϊ����������ZΪHCOOCH2CH3��W��ˮ�⣬����������WΪCH3COOCH3����
��1��������������֪��X��W�ֱ�ΪCH3CH2COOH��CH3COOCH3��W�������������������2����Y�����ǻ�����һ�������·��������ڵ���ˮ��Ӧ�Ļ�ѧ����ʽΪCH3CHOHCHO![]() CH2=CHCHO��H2O����Z��NaOH��Һ��Ӧ�Ļ�ѧ����ʽΪHCOOCH2CH3��NaOH
CH2=CHCHO��H2O����Z��NaOH��Һ��Ӧ�Ļ�ѧ����ʽΪHCOOCH2CH3��NaOH![]() HCOONa��CH3CH2OH��
HCOONa��CH3CH2OH��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������йػ�ѧ��Ӧ������ȷ���ǣ� ��
A. 1-������Ũ�����ᷴӦ�� CH3CH2 CH2OH + HBr ![]() CH3CH2 CH2Br + H2O
CH3CH2 CH2Br + H2O
B. ���ð�˾ƥ�ֹ�������ˮ���ᣨ![]() ���ж���Ӧ�� �ɾ���ע�� NaHCO3��Һ��
���ж���Ӧ�� �ɾ���ע�� NaHCO3��Һ��
![]() + 2 HCO3����
+ 2 HCO3����![]() + 2 CO2�� + 2 H2O
+ 2 CO2�� + 2 H2O
C. ��CH2BrCOOH�м�������������������Һ�����ȣ�CH2BrCOOH��OH��![]() CH2BrCOO����H2O
CH2BrCOO����H2O
D. �������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ѧ��ѧ�����и����ʼ䲻��ʵ��![]() (��������ʾһ�����)ת�����ǣ�
(��������ʾһ�����)ת�����ǣ�
A | B | C | D | |
a | CH3CH2OH | CH2===CH2 | Ӳ֬�� | HCOOCH3 |
b | CH3CHO | CH3CH2Br | Ӳ֬������� | CH3OH |
c | CH3COOH | CH3CH2OH | Ӳ֬���� | HCOOH |
A. A B. B C. C D. D
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij����˻��ϣ��и����˶�Ա������˷ܼ���ȡ���������ʸ���ͼ�Ǽ����˷ܼ���ij��ͬϵ��X�Ľṹ������X��˵����ȷ������ ��
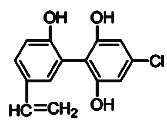
A��X�����в���������ԭ�Ӷ���ͬһƽ����
B��X ����FeCl3��Һʱ����ɫ��������ʹ������Ȼ�̼��Һ��ɫ
C��1 mol X ��������Ũ��ˮ��Ӧ���������5 mol Br2
D��1 mol X��һ����������������������Ӧ���������1 mol H2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ի���������ʶ��ȷ����
A.��ˮ���壺һ����Ҫ����Ũ������������ȡ��������
B.�ϳ��Ȼ��⣺ͨ�� H2 �����Դ��� Cl2������ʹƽ������
C.�ϳɰ������� 500��ĸ��£�����������Ӧ������еij̶�
D.�ȼ��ʹ�õĴ������Ȳ���Ҫ�������Ӵ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ���ҿ��þƾ���Ũ�������Լ�����ȡ��ϩ����ʵ��������������ั��Ӧ�������練Ӧ�л�����SO2��CO2��ˮ���������ij�о���ѧϰС��������ͼ��ʾ��װ���Ʊ���������ϩ��̽����ϩ�뵥�����ܷ�Ӧ����Ӧ���͡��ش��������⣺
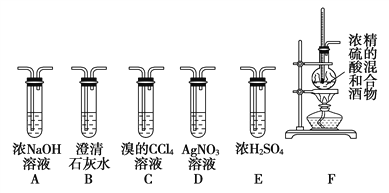
��1��д���Ʊ���ϩ��Ӧ�Ļ�ѧ����ʽ��________________________��ʵ���У����Ũ�������Ҵ��ķ����ǽ�___________����������һ�������У�����Fװ��ʱ����ʹҺ���¶�______________��
��2��Ϊʵ������ʵ��Ŀ�ģ�װ�õ�����˳��ΪF��__________��_______��_______��
_______��D (��װ������һ��)��Bװ�õ�������_________________��
��3����C�й۲쵽____________________ʱ������������������ϩ��Ӧ����D��_________________������C�з�������ȡ����Ӧ����Dû�г���ǰ��������������ʱ������C�з�������_______________��Ӧ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������������ˮ��ɱ����������������ʱ�����һЩ����Ӱ����������ǿ�ʼ�о�һЩ��������ˮ��������ijѧϰС��������Ϸ���NCl3����Ϊɱ������������С��������ͼ��ʾ��װ���Ʊ�NCl3����̽��NCl3��Ư������
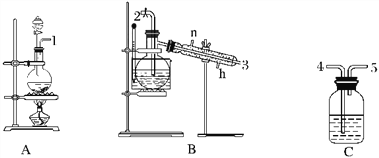
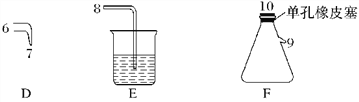
NCl3���������������
�������� | �Ʊ�ԭ�� | ��ѧ���� |
��ɫ��״Һ�壬�۵�Ϊ��40 �����е�Ϊ71 ������������ˮ���������л��ܼ����ܶ�Ϊ1.65 g��mL��1 | Cl2��NH4Clˮ��Һ�ڵ����·�Ӧ | 95 ����ը����ˮ������ˮ��Ӧ |
�ش�����������
(1)����ʵ��Ŀ�����ӿ����ӵ�˳��Ϊ1��____________________________________��
(2)Cװ��������_________________________________________________________________��
(3)Bװ���з�����Ӧ�Ļ�ѧ����ʽΪ____________________________________����Bװ��������ƿ�г��ֽ϶���״Һ������رսӿ�2���Ļ���������ˮԡ���ȵ��¶�Ϊ________________��
(4)��Fװ�õ���ƿ���н϶��ɫ��״Һ�����ʱ���ø������ྻ�IJ�����պȡ��Һ��ε�����ĺ�ɫʯ����ֽ������ֽ����ɫ����ȡ��Һ�����50��60 ����ˮ����Ƭ�̺�ȡ����ˮ�ٵε�����ĺ�ɫʯ����ֽ������ֽ�ȱ�������ɫ��д���йػ�ѧ��Ӧ����ʽ������ʵ��������___________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ����ѧ��ѧ�г������ʵ�ת����ϵ���������ʺͷ�Ӧ������ȥ��

(1)����F�Ļ�ѧʽ��_______________��
(2)д������B��ǿ����Һ��Ӧ�����ӷ���ʽ___________________________��д���ɳ���J����H��Һ�����ӷ���ʽ______________________________________��
(3)��ҺE�м��백ˮʱ�������ɰ�ɫ����L��д������L�����ӷ���ʽ��____________________________________________����ɫ����L���ձ�Ϊ���ɫ����G��д��L��ΪG�Ļ�ѧ��Ӧ����ʽ______________________________��
(4)��ҺI����������������__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ��ʾ(B����ȴװ��δ����)���������Ϳ���(�����뷴Ӧ)�������Լ1��3���ͨ�뺬ˮ8%��̼�������Ʊ�Cl2O������ˮ����Cl2O�Ʊ���������Һ��

��֪��Cl2O��������ˮ����ˮ��Ӧ����HClO��Cl2O�ķе�Ϊ3.8�棬42�����Ϸֽ�ΪCl2��O2��
��1����ʵ���п������������������ȵķ�����_____________________________________��
��ʹ�ö�����ݵ�������__________________________________________��
��2����װ��B�в���Cl2O�Ļ�ѧ����ʽΪ__________________________________________��
����B����ȴװ�ã�����C�е�Cl2O��������١���ԭ����___________________________��
��3��װ��C�в�����ɫԲ����ƿ����Ϊ______________________________________________��
��4����֪������ɱ�H2O2��FeCl2�����ʻ�ԭ��Cl�����ⶨC�����ô�������Һ�����ʵ���Ũ�ȵ�ʵ�鷽������_________��ȡ20.00 mL��������Һ���ձ��У�_________________________________________________________________________________________________________��(��ѡ�õ��Լ���H2O2��Һ��FeCl2��Һ��AgNO3��Һ����������������ʹ�õ������У�������ƽ����ո�����)
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com