
Ϊ�ⶨþ���Ͻ𣨲�������Ԫ�أ������������������ס��ҡ�������ѧϰС��������������ֲ�ͬ��ʵ�鷽������̽������ش��������⣺
![]() ��һ�����飺ʵ�鷽����þ���Ͻ�
��һ�����飺ʵ�鷽����þ���Ͻ�![]() �ⶨʣ���������
�ⶨʣ���������
ʵ�鲽�裺
�ٳ�����������ƽ����һ������þ���Ͻ��ĩ
���ܽ⣺������ҩƷ�����ձ��У����������NaOH��Һ�����Ͻ��裬��ַ�Ӧ��������Ӧ�����ӷ���ʽΪ
�۹��ˣ�
��ϴ�ӣ���δ�Թ������ù������ϴ�ӣ������������������ ���ƫ�ߡ�����ƫ�͡����䡱��
�ݸ������ʣ�����
![]() ���������飺ʵ�鷽����þ���Ͻ�
���������飺ʵ�鷽����þ���Ͻ�![]() �ⶨ������������
�ⶨ������������
ʵ��װ��������ͼ
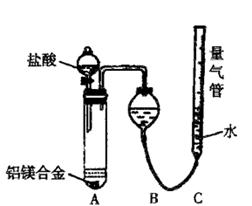
��1��ijͬѧ�����ʵ��װ�ò������ƣ�Ӧ��A��B֮������һ��װ�м�ʯ�ҵĸ���װ�á��������� �����Ҫ������Ҫ����
��2��Ϊʹ�ⶨ��������ܾ�ȷ��ʵ����Ӧע��������ǣ�Ҫ��д�����㣩 ��
���������飺ʵ�鷽����12 gþ���Ͻ�![]()
����l�������ˡ�ϴ�ӡ�����ͳ����������յõ���������1��45g����úϽ���������������Ϊ ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ����NaOH��Һ |
| ����ϡ���� |
| ����NaOH��Һ |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ϊ�ⶨþ���Ͻ𣨲�������Ԫ�أ������������������ס��ҡ�������ѧϰС��������������ֲ�ͬ��ʵ�鷽������̽������ش��������⣺
��һ�����飺ʵ�鷽����þ���Ͻ� �ⶨʣ���������
ʵ�鲽�裺
�ٳ�����������ƽ����һ������þ���Ͻ��ĩ
���ܽ⣺������ҩƷ�����ձ��У����������NaOH��Һ�����Ͻ��裬��ַ�Ӧ��������Ӧ�����ӷ���ʽΪ
�۹��ˣ�
��ϴ�ӣ���δ�Թ������ù������ϴ�ӣ������������������ ���ƫ�ߡ�����ƫ�͡����䡱��
�ݸ������ʣ�����
 ���������飺ʵ�鷽����þ���Ͻ�
���������飺ʵ�鷽����þ���Ͻ��ⶨ������������
ʵ��װ��������ͼ
��1��ijͬѧ�����ʵ��װ�ò������ƣ�Ӧ��A��B֮������һ��װ�м�ʯ�ҵĸ���װ�á��������� �����Ҫ������Ҫ����
��2��Ϊʹ�ⶨ��������ܾ�ȷ��ʵ����Ӧע��������ǣ�Ҫ��д�����㣩 ��
���������飺ʵ�鷽����12 gþ���Ͻ�
����l�������ˡ�ϴ�ӡ�����ͳ����������յõ���������1��45g����úϽ���������������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�긣��ʡ�����и�һ��ѧ��������⻯ѧ���� ���ͣ�ʵ����
Ϊ�ⶨþ���Ͻ𣨲�������Ԫ�أ������������������ס��ҡ�������ѧϰС��������������ֲ�ͬ��ʵ�鷽������̽������ش��������⣺ ��һ�����飺ʵ�鷽����þ���Ͻ�
��һ�����飺ʵ�鷽����þ���Ͻ�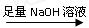 �ⶨʣ���������
�ⶨʣ���������
ʵ�鲽�裺
�ٳ������� ����ƽ����һ������þ���Ͻ��ĩ
����ƽ����һ������þ���Ͻ��ĩ
���ܽ⣺������ҩƷ�����ձ��У����������NaOH��Һ�����Ͻ��裬��ַ�Ӧ��������Ӧ�����ӷ���ʽΪ
�۹��ˣ�
��ϴ�ӣ���δ�Թ������ù������ϴ�ӣ������������������ ���ƫ�ߡ�����ƫ�͡����䡱��
�ݸ������ʣ����� ���������飺ʵ�鷽����þ���Ͻ�
���������飺ʵ�鷽����þ���Ͻ� �ⶨ������������
�ⶨ������������
ʵ��װ��������ͼ
��1��ijͬѧ�����ʵ��װ�ò������ƣ�Ӧ��A��B֮������һ��װ�м�ʯ�ҵĸ���װ�á��������� �����Ҫ������Ҫ����
��2��Ϊʹ�ⶨ��������ܾ�ȷ��ʵ����Ӧע��������ǣ�Ҫ��д�����㣩 ��
���������飺ʵ�鷽����12 gþ���Ͻ�
����l�������ˡ�ϴ�ӡ�����ͳ����� �����յõ���������1��45g����úϽ���������������Ϊ ��
�����յõ���������1��45g����úϽ���������������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�긣��ʡ�����и�һ��ѧ��������⻯ѧ���� ���ͣ�ʵ����
Ϊ�ⶨþ���Ͻ𣨲�������Ԫ�أ������������������ס��ҡ�������ѧϰС��������������ֲ�ͬ��ʵ�鷽������̽������ش��������⣺
��һ�����飺ʵ�鷽����þ���Ͻ�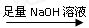 �ⶨʣ���������
�ⶨʣ���������
ʵ�鲽�裺
�ٳ�����������ƽ����һ������þ���Ͻ��ĩ
���ܽ⣺������ҩƷ�����ձ��У����������NaOH��Һ�����Ͻ��裬��ַ�Ӧ��������Ӧ�����ӷ���ʽΪ
�۹��ˣ�
��ϴ�ӣ���δ�Թ������ù������ϴ�ӣ������������������ ���ƫ�ߡ�����ƫ�͡����䡱��
�ݸ������ʣ�����
 ���������飺ʵ�鷽����þ���Ͻ�
���������飺ʵ�鷽����þ���Ͻ� �ⶨ������������
�ⶨ������������
ʵ��װ��������ͼ
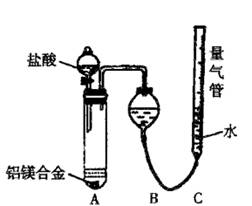
��1��ijͬѧ�����ʵ��װ�ò������ƣ�Ӧ��A��B֮������һ��װ�м�ʯ�ҵĸ���װ�á��������� �����Ҫ������Ҫ����
��2��Ϊʹ�ⶨ��������ܾ�ȷ��ʵ����Ӧע��������ǣ�Ҫ��д�����㣩 ��
���������飺ʵ�鷽����12 gþ���Ͻ�
����l�������ˡ�ϴ�ӡ�����ͳ����������յõ���������1��45g����úϽ���������������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
Ϊ�ⶨþ���Ͻ𣨲�������Ԫ�أ�������������������������ѧϰС����������ж��ֲ�ͬ��ʵ�鷽������̽������ش��������⣺
��һ�����飺
ʵ�鷽����þ���Ͻ�![]() �ⶨʣ���������
�ⶨʣ���������
ʵ�鲽�裺
�ٳ��� ��������ƽ����2.7gþ���Ͻ��ĩ
���ܽ� �Ѣ���ҩƷ�����ձ��У��ù��Ϊ mL����Ͳ��ȡһ�����1mol/L NaOH ��Һ�����ձ��У����Ͻ��裬��ַ�Ӧ
�۹���
��ϴ�� ��δ�Թ������ù������ϴ�ӣ������������������ ���ƫ�ߡ�����ƫ�͡����䡱 ����֤��������ϴ�Ӹɾ��ķ���Ϊ
�ݸ������ʣ�����
���������飺
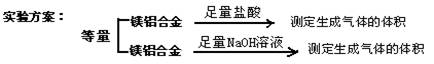
ʵ��װ�ã�
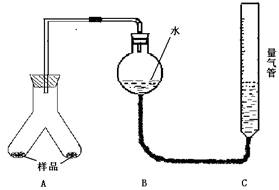
ʵ�鲽�裺
��ͼ���Ӻ�װ��
�ڳ�ȡ����������Ϊ0.3g ��þ���Ͻ���Ʒ��ĩ���ֱ����Aװ�������������У���Bװ���м���һ������ˮ����װ��A��B�Ľ������ã�Ȼ�����C�ĸ߶�ʹB��C�е�Һ����ƽ����¼�´�ʱ�����Ϊ112 mL
��ͨ��ע��������ͷ��װ��A������ע��������ϡ���ᣬ�Ȳ��������ݲ���ʱ������װ��C�ĸ߶ȣ�ʹB��C�е�Һ����ƽʱ��¼�´�ʱ�����Ϊ448mL
��ͨ����һ��ͷ��װ��A�Ҳ���м���������ϡNaOH��Һ���Ȳ��������ݲ���ʱ������װ��C�ĸ߶ȣ�ʹB��C�е�Һ����ƽʱ��¼�´�ʱ�����Ϊ672mL��
��������ۣ�
��1������ʵ�鲽��ٺ͢�֮������һ�� �IJ��������巽��Ϊ ��
��2��Ϊʹ��������ⶨ�������������ܴ�ƫ�����Ӧע��ʹB��C�е�Һ����ƽ�⣬�ڷ�Ӧ��ȫ��������֮ǰ������Ҫע��Ĺؼ������ǣ�ֻ��д��1�㣩 ��
��3������Ͻ���������������ʱ���Ƿ���Ҫ�������������Ϊ��״������� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com