
���� ��1������������Һ���ѡ������ƿ���������Һϡ���������ʵ����ʵ������������ҪŨ���������
��2����������һ�����ʵ���Ũ����Һһ�㲽��ѡ����Ҫ������
��3������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ���ݴ�����
��4���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��5���ȸ���n=$\frac{V}{Vm}$�������״����aL�Ȼ�������ʵ�����Ȼ�����m=nM������Ȼ����������1Lˮ������ԼΪ1000g���Ӷ���֪��Һ�������ٸ���V=$\frac{m}{��}$�����������Һ�����������c=$\frac{n}{V}$���������������ʵ���Ũ�ȣ�
��� �⣺��1����12.0mol/LŨ��������230mL 0.3mol/L��ϡ���ᣬӦѡ��250mL����ƿ������ҪŨ�������V����������Һϡ���������ʵ����ʵ����������ã�12.0mol/L��V=250mL��0.3mol/L�����V=6.3mL��
�ʴ�Ϊ��6.3��
��2������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ�����������Ͳ����ͷ�ιܡ��ձ���������������ƿ������230mL 0.3mol/L��ϡ���ᣬӦѡ��250mL�������ƿ����ȱ�ٵ�������250mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��250mL����ƿ����ͷ�ιܣ�
��3������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�ҡ�ȡ�װƿ����ǩ��������ȷ��˳��Ϊ��baedc��
�ʴ�Ϊ��baedc��
��4��a����ȡŨ����ʱ���ӣ�������ȡ��Ũ�������ƫС�����ʵ����ʵ���ƫС����ҺŨ��ƫС��
�ʴ�Ϊ��ƫС��
b����ȡŨ�������ϴ����Ͳ����ϴ��Һת��������ƿ��������ȡ��Ũ�������ƫ�����ʵ����ʵ���ƫ����ҺŨ��ƫ��
�ʴ�Ϊ��ƫ��
c��ʵ��ǰ������ƿ����������������ˮ�������ʵ����ʵ�������Һ�����������Ӱ�죬��ҺŨ�Ȳ��䣻
�ʴ�Ϊ�����䣻
��5����״���µ�a L �Ȼ�����������ʵ���Ϊ��n��HCl��=$\frac{aL}{22.4L/mol}$=$\frac{a}{22.4}$mol����HCl������Ϊ��36.5g/mol��$\frac{a}{22.4}$mol=$\frac{36.5a}{22.4}$g��
1Lˮ������ԼΪ1000g�������������Ϊ��1000g+$\frac{36.5a}{22.4}$g��
������������$\frac{1000+\frac{36.5a}{22.4}}{b}$=$\frac{22400+36.5a}{22.4b}$mL��
���Ը���������ʵ���Ũ��Ϊ��c��HCl��=$\frac{\frac{a}{22.4}}{\frac{22400+36.5a}{22.4b��1{0}^{-3}}}$=$\frac{1000ab}{22400+36.5a}$mol/L��
�ʴ�Ϊ��$\frac{1000ab}{22400+36.5a}$��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ��й����ʵ���Ũ�ȼ��㣬��ȷ���ʵ���Ũ�ȶ��弰����ԭ���Ͳ��������ǽ���ؼ�����Ŀ�ѶȲ���


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����ʵʩ����ȼ�ϵ����������������Լ���SO2��NOx���ŷ� | |
| B�� | PM2.5��ֱ����2.5��ϸ������������������ж��к����� | |
| C�� | O3������ɱ����������O3Ũ��Խ��Խ���������彡�� | |
| D�� | ����β���е�CO��Ҫ�������͵IJ���ȫȼ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ڸ������£���Ӧǰ���ѹǿ֮��Ϊ6��5.3 | |
| B�� | ����Ӧ��ʼʱ�������Ϊ2 L����0��2 min��v��SO3��=0.35 mol/��L•min�� | |
| C�� | ���ѡ����º�ѹ�¡���Ϊ����ѹ���������¡���Ӧ��ƽ���n��SO3����1.4 mol | |
| D�� | ���ѡ����º�ѹ�¡���Ϊ�����º����¡���Ӧ����ƽ��ʱ�ų�����������Q kJ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 1905��¹���ѧ�ҹ��������˺ϳɰ��ķ���������˻����1918���ŵ������ѧ�������ĺϳɲ�������˵���������ʳ��������µļ������������⣬�ڹ�������Դ���Ṥҵ����Ҳ�й㷺��;��
1905��¹���ѧ�ҹ��������˺ϳɰ��ķ���������˻����1918���ŵ������ѧ�������ĺϳɲ�������˵���������ʳ��������µļ������������⣬�ڹ�������Դ���Ṥҵ����Ҳ�й㷺��;���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
| ʵ����� | Ԥ������ | ��Ӧ�����ӷ���ʽ |
| ��ʢ���¸�FeSO4��Һ���Թ��е�������Ũ���ᣬ�� | �Թ��в�������ɫ���壬��Һ��ɫ��� | Fe2++NO3-+2H+=Fe3++NO2��+H2O |
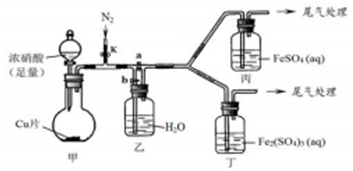
| ���� | ���������� | ���ͻ���� |
| �� | �ر�k��b����a����ʼ���з�Ӧ��һ��ʱ��۲쵽������Һ��Ϊ����ɫ����������Һ�����Ա仯 | ��Һ����ɫ��Fe2+��NO��NO2��Ӧ���ĵõ� |
| �� | ֹͣ���з�Ӧ����k��b���ر�a������ͨ��N2һ��ʱ�� | �ò�����Ŀ���ž���װ�ü��������е�NO2 |
| �� | �����µı�����װ�ã���ͨһ��ʱ��N2��ر�k��ʹ���з�Ӧ�������۲쵽�������벽�������ͬ | ��ƿ�з����Ļ�ѧ��Ӧ����ʽΪ3NO2+H2O=2HNO3+NO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ڳ��³�ѹ�£�11.2 L O2���еķ�����Ϊ0.5NA | |
| B�� | �ڳ��³�ѹ�£�1 mol�������е�ԭ����Ϊ2NA | |
| C�� | 71 g Cl2����ԭ����Ϊ2NA | |
| D�� | ��״���£�1 mol H2O��1 mol H2�������ԼΪ22.4 L |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
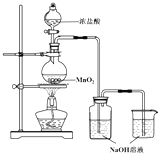 ijʵ��С������ͼװ���Ʊ���������Һ����̽�������ʣ���Ӧֹͣ��ȡϴ��ƿ����ɫ��Һ5mL�ֱ����������ʵ�飺
ijʵ��С������ͼװ���Ʊ���������Һ����̽�������ʣ���Ӧֹͣ��ȡϴ��ƿ����ɫ��Һ5mL�ֱ����������ʵ�飺| ���� | ���� |
| a������ҺpH���������еμ�2�η�̪ | pH=13����Һ��죬5min����ɫ |
| b����������μ������� | ��Һ��ɻ���ɫ |
| ���� | ���� |
| ȡ5mL pH=13NaOH��Һ�������еμ�2�η�̪ | ��Һ��죬30min����ɫ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com