
| A£®b<a<7 |
| B£®Ņõ¼«µē¼«·“Ó¦Ź½ĪŖ£ŗCu2++2e-="Cu" |
| C£®ĻņČÜŅŗÖŠ¼ÓČĖ9.8x(10-b-10-a)gCu(OH)2æÉŹ¹ČÜŅŗ»Öø“µ½µē½āĒ°µÄÅØ¶Č |
| D£®Ńō¼«²śÉśµÄĘųĢåŹĒO2£¬ĘäĢå»ż(±ź×¼×“æöĻĀ)ĪŖ1.12x(10-b-10-a)L |
 ”£Ņņ“ĖCĻī“ķĪó£¬ĘäӹєĻī¾łÕżČ·”£“š°øŃ”C”£
”£Ņņ“ĖCĻī“ķĪó£¬ĘäӹєĻī¾łÕżČ·”£“š°øŃ”C”£

 ĆūŠ£æĪĢĆĻµĮŠ“š°ø
ĆūŠ£æĪĢĆĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®ČÜŅŗµÄÅØ¶Č±äĪŖ0.08 mol”¤L-1 | B£®Ńō¼«ÉĻ²śÉś112 mL O2£Ø±ź×¼×“æö£© |
| C£®×ŖŅʵĵē×ÓŹżŹĒ1.204”Į1023 | D£®·“Ó¦ÖŠÓŠ0.02 molµÄAg±»Ńõ»Æ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā

| A£®×°ÖĆ¼×ÖŠ¶ŌÓŚĢśÕ¢Ćŵı£»¤ÓƵďĒ”°ĪžÉüŃō¼«µÄŅõ¼«±£»¤·Ø”±£¬ŗø½ÓŌŚĢśÕ¢ÉĻµÄ½šŹōRæÉŅŌŹĒĪżæé |
| B£®×°ÖĆŅŅÖŠ¶ŌÓŚĢśÕ¢Ćŵı£»¤²ÉÓƵďĒ”°Ķā¼ÓµēĮ÷µÄŅõ¼«±£»¤·Ø”±£¬ĢśÕ¢ĆÅÓ¦ÓėÖ±Į÷µēŌ“µÄøŗ¼«ĻąĮ¬ |
| C£®×°ÖƱūæÉŹµĻÖµē½ā¾«Į¶Ķ£¬µē¼«AµÄ²ÄĮĻÓĆ“æĶ£¬µē¼«BµÄ²ÄĮĻÓĆ“ÖĶ |
| D£®×°Öƶ”æÉŹµĻÖ¶ĘĶ£¬Ķʬ½ÓŌŚµēŌ“µÄÕż¼«£¬“ż¶Ę½šŹō½ÓŌŚµēŌ“µÄøŗ¼«£¬XČÜŅŗÓ¦ÓĆŗ¬ÓŠ“ż¶Ę½šŹōŃōĄė×ÓµÄČÜŅŗ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
 Na2S4+3NaBr”£±ÕŗĻæŖ¹ŲKŹ±£¬b¼«ø½½üĻȱäŗģÉ«”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
Na2S4+3NaBr”£±ÕŗĻæŖ¹ŲKŹ±£¬b¼«ø½½üĻȱäŗģÉ«”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ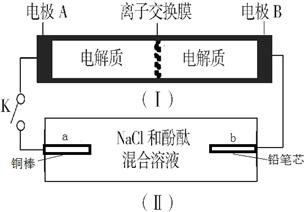
| A£®µ±ÓŠ0.01 mol Na+ĶعżĄė×Ó½»»»Ä¤Ź±£¬bµē¼«ÉĻĪö³ö±ź×¼×“æöĻĀµÄĘųĢå112 mL |
B£®øŗ¼«·“Ó¦ĪŖ4Na 4e-=4Na+ 4e-=4Na+ |
| C£®±ÕŗĻKŗó£¬bµē¼«ø½½üµÄpH±äŠ” |
| D£®±ÕŗĻKŗó£¬aµē¼«ÉĻÓŠ²śÉśĒāĘų |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®3.36 L | B£®4.48 L | C£®5.60 L | D£®6.72 L |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
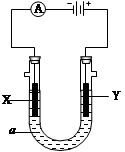
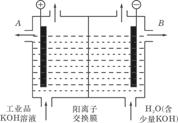
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗ¼ĘĖćĢā
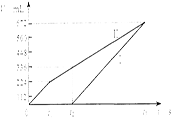
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com