
ijС�������֪:������I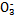 >Fe3+>I2,3Br2+6FeCl2
>Fe3+>I2,3Br2+6FeCl2 2FeBr3+4FeCl3;I2+2S2
2FeBr3+4FeCl3;I2+2S2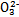
 S4
S4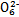 +2I-;CuI��һ�ְ�ɫ����(Ksp=1.3��1
+2I-;CuI��һ�ְ�ɫ����(Ksp=1.3��1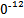 )��
)��
��.��С��Ϊȷ��һ�ݼӵ���(���ܺ���KIO3��KI��Mg2+��Fe3+)�ijɷ�,��ƶ���ʵ����Բ������֤��
(1)ʵ���������:
| ʵ�鲽�� | ʵ����̺����� | ��Ӧ���� | |
| ����1 | ȡһ��������,����������ˮ�ܽ�,����ϡ�����ữ,��������Һ��Ϊ3�� | _____________ | |
| �� �� 2 | �ڢٷ� ��Һ | 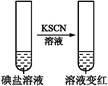 | �����п϶����������� |
| �ڢڷ� ��Һ | 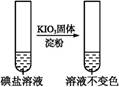 | _____________ | |
| �ڢ۷� ��Һ | 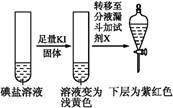 | XΪ������(�ѧʽ) | |
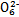 ��I2��Br2����������ǿ������˳������������������������
��I2��Br2����������ǿ������˳������������������������  �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д� ����ѧ��Ӧ�����ϵ�д�
����ѧ��Ӧ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ҫ������������
(1)д���������ʵĵ��뷽��ʽ:
Fe2(SO4)3_____________________________________________��
NaHCO3______________________________________________��
(2)д�����з�Ӧ�����ӷ���ʽ��
ϡ������̼��Ʒ�Ӧ___________________________________��
����������Һ��ϡ���ᷴӦ_______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ɫ��Һ�������п��ܴ����������ӣ�Na+��Ag+��Ba2+��Al3+��AlO2-��S2-��CO32-��SO32-��SO42-����ȡ����Һ�����й����飬�������£�
�ش��������⣺
��1�����ɳ��������ӷ���ʽ�� ��
��֪�������������������ɣ�������ˮ������HBr�����������ʵ����һ���������ֳɷ֣��������Լ��Լ����������±��У��ɲ�����������ÿһ�ж�Ӧ��ȷ���ɵ÷֣�
| ���� | ʵ��Ŀ�� | �Լ� | ���� |
| 1 | | | |
| 2 | | | |
| 3 | | | |
| 4 | | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ŀǰ������Դ���ϱ����õ��ִ��������У�������ؼ������ǿ��л��������о���һ������
��1��������أ�п��أ����Խ��ʣ���һ�ֵ��͵ĸ�����أ�����ֵ�ظ��������� ��
��2����ҵ�ϳ�����NaClO����������������أ�K2FeO4����K2FeO4�ڼ��Ի������ȶ��������˺����������²��ȶ�����Ӧԭ��Ϊ��
���ڼ��������£�����NaClO����Fe(NO3)3�Ƶ�Na2FeO4
3NaClO + 2Fe(NO3)3 + 10NaOH��2Na2FeO4��+ 3NaCl + 6NaNO3 + 5H2O
�� Na2FeO4��KOH��Ӧ����K2FeO4��Na2FeO4 + 2KOH��K2FeO4 + 2NaOH
��Ҫ�������������£�
��д����Ӧ�ٵ����ӷ���ʽ ��
������ͼ�С�ת��������Ӧ�ۣ�����ij�����½��еģ�˵�����¶���Ksp��K2FeO4�� Ksp��Na2FeO4���������������������
��3����֪K2FeO4��ˮ��Һ�п��Է�����4FeO42����10H2O 4Fe(OH)3����8OH��+3O2��,��K2FeO4������ˮ�����е������� ��
4Fe(OH)3����8OH��+3O2��,��K2FeO4������ˮ�����е������� ��
��4��FeO42����ˮ��Һ�еĴ�����̬ͼ��ͼ��ʾ��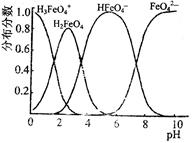
������pH=10��������Һ�м�������pH=2��HFeO4-�ķֲ������ı仯����� ��
������pH=6��������Һ�еμ�KOH��Һ������Һ�к���Ԫ�ص����У� ת��Ϊ ���ѧʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��ˮ��������ʱ������˫����(H2Dz����Ԫ����)�ѽ���������ϳɵ����Ե����ʣ�����CCl4��ȡ�����Ӷ��ѽ������Ӵ�ˮ��Һ����ȫ�������������˫����(H2Dz)��CCl4������ˮ�е�Cu2+ʱ���ȷ�����Ϸ�Ӧ��Cu2++2H2DZ Cu (HDZ)2+2H+���ټ���CCl4��Cu (HDZ)2�ͺ����ױ���ȡ��CCl4�С�
Cu (HDZ)2+2H+���ټ���CCl4��Cu (HDZ)2�ͺ����ױ���ȡ��CCl4�С�
��1��д��˫�����Fe3+��ϵ����ӷ���ʽ��_____________________����ȡFe3+�Ĺ�����Ҫ�������˵���ȣ������Һ��pH����������___________________________��
��2����ͼ����˫����(HzDz)��CCl4�����ȡijЩ�������ӵ�������ߣ�����ӳ����ȡijЩ��������ʱ���˵�pH��Χ��E����ʾij�ֽ����������������ʽ��ȡ����İٷ��ʡ�
ij��ҵ��ˮ�к���Hg2+��Bi3+��Zn2+����˫���꣨H2Dz��~ CCl4�����ȡ��������ˮ��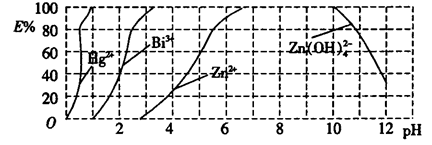
������ȫ����ˮ�е�Hg2+����������������Һ��pH=________��
�ڵ�����pH=2ʱ����(Bi)�Ĵ�����ʽ��_________________��
��3����ˮ�е��ǹ�����(Hg2+ 2)����ת���ɹ�����(Hg2+)������˫������ϡ�ij������ˮ�к��н϶���Ȼ��ǹ�(Hg2Cl2)������������(K2S2O8)������(Hg2+ 2)��д���÷�Ӧ�Ļ�ѧ����ʽ��________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ȣ�ClO2���ڳ�������һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ��59�棬�е�Ϊ11��0�棬������ˮ����ҵ�����Գ�ʪ��KClO3�Ͳ��ᣨH2C2O4����60��ʱ��Ӧ�Ƶá�ijѧ��������ͼ��ʾװ��ģ�ҵ��ȡ���ռ�ClO2�����ڸ÷�Ӧ�����ȷ�Ӧ������������������ڸ÷�Ӧ�Է�����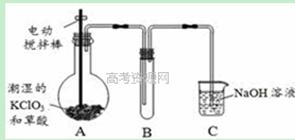
��1��A�з�Ӧ������K2CO3��ClO2��CO2�ȣ���д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��2��A���������¶ȿ���װ�ã����ƾ����⣬����Ҫ�IJ����������ձ��� ��Bװ�ñ�����ڱ�ˮԡ�У���ԭ���� ��
��3����Ӧ����װ��C�пɵ�NaClO2��Һ����֪NaClO2������Һ���¶ȵ���38��ʱ����������NaClO2��3H2O�����¶ȸ���38��ʱ����������NaClO2���벹���NaClO2��Һ���Ƶ�NaClO2����IJ������裺�� ���� ����ϴ�ӣ��ܸ��
��4��ClO2�ܲ��ȶ������������ƣ�������ˮ���յõ�ClO2��Һ��Ϊ�ⶨ������Һ��ClO2�ĺ���������������ʵ�飺
����1��ȷ��ȡClO2��Һ10.00 mL��ϡ�ͳ�100.00 mL��������ȡV1 mL�������뵽��ƿ�У�
����2����ϡ�������������pH��2.0������������KI���壬����Ƭ�̣�
����3���������ָʾ������c mol/L Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��ҺV2 mL������֪2 Na2S2O3 + I2��Na2S4O6 + 2NaI��
������100 mL c mol/LNa2S2O3����Һʱ���õ��IJ����������ձ�����Ͳ����������У� ��
��д������2�з�����Ӧ�����ӷ���ʽ ��
��ԭClO2��Һ��Ũ��Ϊ g / L���ò����е���ĸ����ʽ��ʾ����
�����ζ�ǰ�ζ��ܼ����������ݣ��ζ���������ʧ����ⶨ��� ��
���ζ���ʼ���Ӷ������ζ��յ�ʱ��ȷ��������ⶨ��� ��
���ƫ�ߡ���ƫ�͡����䡱 ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����Fe2����I����Br������Һ��ͨ��������������Һ�и������ӵ����ʵ����仯��ͼ��ʾ���й�˵������ȷ����( )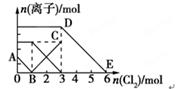
| A���߶�BC����Fe3�����ʵ����ı仯��� |
| B��ԭ�����Һ��n(FeBr2)=3mol |
| C����ͨ��2molCl2ʱ����Һ���ѷ��������ӷ�ӦΪ��2Fe2��+2I��+2Cl2=2Fe3��+I2+4Cl�� |
| D��ԭ��Һ��n(Fe2��)��n(I��)��n(Br��)=2��1��3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�Թ��ڱڵ���ǿ������ȵ�KOH��Һ��ϴ�ӣ�3S+6KOH==K2SO3+2K2S+3H2O���÷�Ӧ�У��������뱻��ԭ����ԭ�Ӹ�����Ϊ
| A��1��2 | B��2��1 | C��1��1 | D��3��2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ͭ����Ҫ���ջ�ͭ��ӦΪ:CuFeS2+O2 Cu2S+FeS+SO2 ������˵����ȷ����
Cu2S+FeS+SO2 ������˵����ȷ����
| A��CuFeS2�������������ǻ�ԭ������Ԫ�ؼȱ������ֱ���ԭ | B��ÿ����1molSO2ת��6mol���� |
| C��SO2���������������ǻ�ԭ���FeSֻ�ǻ�ԭ���� | D��ÿת��1.2 mol���ӣ���0.3 mol��������ԭ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com