
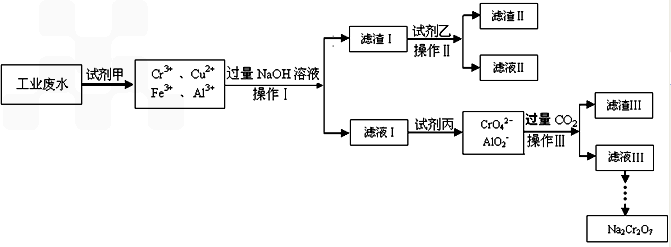
 NaCrO2
NaCrO2  Na2CrO4
Na2CrO4 Na2Cr2O7
Na2Cr2O7 
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| NaOH |
| Cl2 |
| �ữ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
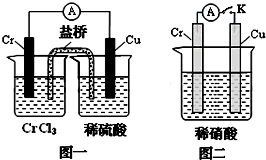 2011��6�£���������½������ʵҵ����˾5000��ֹ�ҵ���ϸ����Ƿ��㵹���µ���������Ⱦ��+6�۸��ױ��������գ����°���+3�۸����ױ��������գ�����С����ҵ������ˮ�Ĵ�������֮һ�ǽ���+6�۸��ķ�ˮ��������ڣ�����������������������NaCl����е�⣺���������ɵ�Fe2+��Cr2O72-������Ӧ�����ɵ�Fe3+��Cr3+����������OH-�������Fe��OH��3��Cr��OH��3������ȥ��
2011��6�£���������½������ʵҵ����˾5000��ֹ�ҵ���ϸ����Ƿ��㵹���µ���������Ⱦ��+6�۸��ױ��������գ����°���+3�۸����ױ��������գ�����С����ҵ������ˮ�Ĵ�������֮һ�ǽ���+6�۸��ķ�ˮ��������ڣ�����������������������NaCl����е�⣺���������ɵ�Fe2+��Cr2O72-������Ӧ�����ɵ�Fe3+��Cr3+����������OH-�������Fe��OH��3��Cr��OH��3������ȥ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
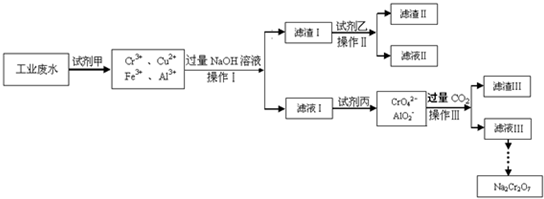
 NaCrO2
NaCrO2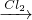 Na2CrO4
Na2CrO4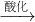 Na2Cr2O7
Na2Cr2O7�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�걱���з�̨���������ϣ���ĩ��ѧ�Ծ��������棩 ���ͣ������
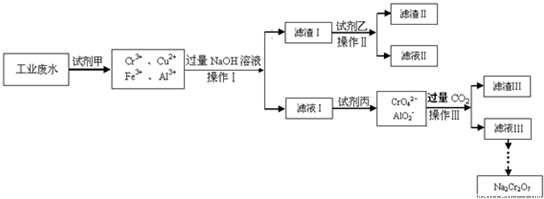
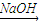 NaCrO2
NaCrO2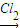 Na2CrO4
Na2CrO4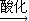 Na2Cr2O7
Na2Cr2O7�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com