
���� ��1������m=CVM������Ҫ���ʵ�������
��2����������һ�����ʵ���Ũ����Һ��һ�㲽��ѡ����Ҫ��������ȷ��ȱ�ٵ�������
��3����������ƿ�Ĺ��켰��ȷʹ�÷������
��4����������һ�����ʵ���Ũ����Һ��һ�㲽������
��� �⣺��1����NaOH��������1.0mol/L��NaOH��Һ480mL��Ӧѡ��500mL����ƿ��ʵ������500mL��Һ����Ҫ���ʵ�����m=0.1mol/L��40g/mol��0.5L=2.0g��
�ʴ�Ϊ��2.0��
��2������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݵȣ��õ���������
������ƽ��ҩ�ס�500mL����ƿ���ձ�������������ͷ�ιܣ�
����ʹ�õIJ������������ձ� ��500mL����ƿ �ݲ���������ȱ�ٵ���������ͷ�ιܣ�
�ʴ�Ϊ���٢ܢݣ���ͷ�ιܣ�
��3������ƿ���л�����ʹ��ǰ��Ҫ����Ƿ�©ˮ��
�ʴ�Ϊ����©��
��4������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ�ȣ�������ȷ��˳��Ϊ���ݢڢ٢ۢܣ�
�ʴ�Ϊ���ݢڢ٢ۢܣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ���Ͳ��������ǽ���ؼ���ע������ƿ�Ĺ��켰ʹ�÷�������Ŀ�ѶȲ���



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �٢ڢ� | B�� | �ڢۢ� | C�� | �٢ڢ� | D�� | �٢ڢۢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����Ni���������CO��ת���ʣ�Ni��ת���ʽ��� | |
| B�� | ��С�����ݻ���ƽ�����ƣ���H��С | |
| C�� | ��Ӧ�ﵽƽ�����CO�ٴδﵽƽ��ʱ��CO������������� | |
| D�� | ��4v��[Ni��CO��4]=v����CO��ʱ�������л�������ܶȲ���ʱ������˵����Ӧ�Ѵﻯѧƽ��״̬ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Al����������չ�ԺͿ���ʴ�ԣ����Ƴ�������װ��Ʒ | |
| B�� | KAl��SO4��2ˮ�������Al��OH��3���壬����������ˮ�ľ��� | |
| C�� | NH3����Cl2����NH4Cl������Ũ��ˮ�������������Ĺܵ��Ƿ���й© | |
| D�� | NaHCO3����Ӧ��ʳƷ��ҵ�������Ƹ������ɼ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
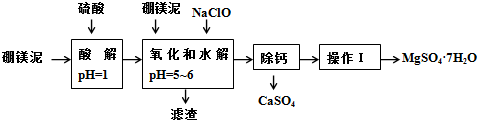
| �������� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
| Mg��OH��2 | 9.3 | 10.8 |
| Fe��OH��2 | 7.6 | 9.6 |
| Fe��OH��3 | 2.7 | 3.7 |
| Al��OH��3 | 3.7 | 4.7 |
| �¶ȣ��棩 | 40 | 50 | 60 | 70 |
| MgSO4 | 30.9 | 33.4 | 35.6 | 36.9 |
| CaSO4 | 0.210 | 0.207 | 0.201 | 0.193 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
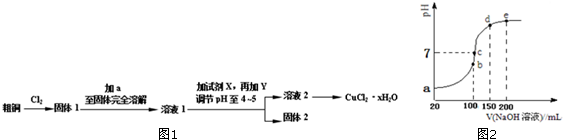
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
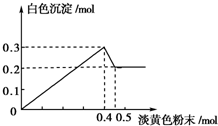 ��һ����Һ����֪���п��ܺ���Fe3+��Mg2+��Cu2+��Al3+��NH4+������һ�ֵ���ɫ��ĩ����ʱ�������д̼�����ζ�Ļ������ų���ͬʱ���ɰ�ɫ������������0.4mol����ɫ��ĩʱ����������0.3mol���������뵭��ɫ��ĩʱ�������̼�����ζ�����壬�Ҽ��뵭��ɫ��ĩʱ������ɫ������������ͼ��ʾ������֪��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O����
��һ����Һ����֪���п��ܺ���Fe3+��Mg2+��Cu2+��Al3+��NH4+������һ�ֵ���ɫ��ĩ����ʱ�������д̼�����ζ�Ļ������ų���ͬʱ���ɰ�ɫ������������0.4mol����ɫ��ĩʱ����������0.3mol���������뵭��ɫ��ĩʱ�������̼�����ζ�����壬�Ҽ��뵭��ɫ��ĩʱ������ɫ������������ͼ��ʾ������֪��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���ά����Ϣ���ز����ٵ��л��ϳɲ��� | |
| B�� | ʳ�μӵ�ʵ������ʳ���м���KIO3 | |
| C�� | ����ɻ��ϵ��մɷ���Ƭ�����������ǽ������� | |
| D�� | �������������������Ⱦ���������ʣ��ʲ��˲��ô˷� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com