

| 10.6g |
| 11.0g |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A���������м���Ũ�������ַ�������˵��Ũ���������ˮ�� |
| B��Ũ�����ڹ�������ɫ��ƣ�˵��Ũ����ȶ� |
| C����ˮ���������������Ϊ������Һ�� |
| D�����ˮ�еμ�CCl4�����ú�ֲ㣬CCl4����Ϻ�ɫ��˵������CCl4�ӵ�ˮ����ȡ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A�� |
| B��HOCH2CH2OH |
C�� |
D��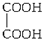 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A������������� |
| B�������¶� |
| C��������� |
| D������H2���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| Ԫ�� | ��ؽṹ������ |
| A | �䵥�ʵ�һ�־����䲻�ǽ������壬���ǵ�������壬�����ۡ������������ԣ�������ԭ�Ӻ˷�Ӧ�ѵ��������������������ܺ͵缫���ϵȣ� |
| B | �䵥�ʼ�����ǿ����Һ��Ӧ��������ǿ����Һ��Ӧ����������ڵ������ڵ��������а뾶��С�� |
| C | �ǵ����ʵ����Ԫ��֮һ��ԭ�Ӻ�����3�ֲ�ͬ�����ĵ��ӣ���δ�ɶԵ�������࣮ |
| D | ԭ�Ӻ������������Ų�ʽΪnsnnp2n+1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A����״���£�1L������ȫȼ�������ɵ���̬����ķ�����Ϊ
| ||
| B��1 mol����-CH3�������ĵ�������Ϊ10NA | ||
| C��7 g CnH2n�к��е���ԭ����ĿΪNA | ||
| D��1mol���ʯ�����У�ƽ������̼̼��������Ϊ4 NA |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com