
£Ø1£©”¢¢ŁÓĆėĀ(N2H4)ĪŖČ¼ĮĻ£¬ĖÄŃõ»Æ¶žµŖ×öŃõ»Æ¼Į£¬Į½Õß·“Ӧɜ³ÉµŖĘųŗĶĘųĢ¬Ė®”£
ŅŃÖŖ£ŗN2(g)£«2O2(g)£½N2O4(g) ¦¤H£½+10.7kJ”¤mol-1
N2H4(g)£«O2(g)£½N2(g)£«2H2O(g) ¦¤H£½-543kJ”¤mol-1
Š“³öĘųĢ¬ėĀŗĶN2O4·“Ó¦µÄČČ»Æѧ·½³ĢŹ½£ŗ ”£
¢ŚŅŃÖŖĖÄŃõ»Æ¶žµŖŌŚ“óĘųÖŠ»ņŌŚ½ĻøßĪĀ¶ČĻĀŗÜÄŃĪČ¶Ø“ęŌŚ£¬ĘäŗÜČŻŅ××Ŗ»ÆĪŖ¶žŃõ»ÆµŖ”£ŹŌĶʶĻÓɶžŃõ»ÆµŖÖĘČ”ĖÄŃõ»Æ¶žµŖµÄ·“Ó¦Ģõ¼ž(»ņ“ėŹ©)£ŗ ”£
£Ø2£©æĘѧ¼ŅÖĘŌģ³öŅ»ÖÖŹ¹ÓĆ¹ĢĢåµē½āÖŹµÄČ¼ĮĻµē³Ų£¬Ę䊧ĀŹøüøߣ¬æÉÓĆÓŚŗ½Ģģŗ½æÕ”£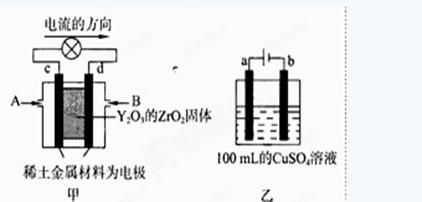
Ķ¼¼×ĖłŹ¾×°ÖĆÖŠ£¬ŅŌĻ”ĶĮ½šŹō²ÄĮĻĪŖ¶čŠŌµē¼«£¬ŌŚĮ½¼«ÉĻ·Ö±šĶØČėCH4ŗĶæÕĘų£¬ĘäÖŠ¹ĢĢåµē½āÖŹŹĒ²ōŌÓĮĖY2O3µÄZrO2¹ĢĢ壬ĖüŌŚøßĪĀĻĀÄÜ“«µ¼Ńō¼«Éś³ÉµÄO2-(O2+4e ”ś2O2-)
¢Łcµē¼«ĪŖ £¬dµē¼«ÉĻµÄµē¼«·“Ó¦Ź½ĪŖ ”£
¢ŚĶ¼ŅŅŹĒµē½ā100mL 0.5mol”¤L-1 CuSO4ČÜŅŗ£¬aµē¼«ÉĻµÄµē¼«·“Ó¦Ź½ĪŖ ”£Čōaµē¼«²śÉś56mL(±ź×¼×“æö)ĘųĢ壬ŌņĖłµĆČÜŅŗµÄpH= (²»æ¼ĀĒČÜŅŗĢå»ż±ä»Æ)£¬ČōŅŖŹ¹µē½āÖŹČÜŅŗ»Öø“µ½µē½āĒ°µÄדĢ¬£¬æɼÓČė (Ń”Ģī×ÖÄøŠņŗÅ)
a.CuO b.Cu(OH)2 c.CuCO3 d.Cu2(OH)2CO3
£Ø14·Ö£©
£Ø1£©¢Ł2 N2H4(g) + N2O4(g)= 3N£²(g)£«4H20(g) ”÷H£½£1096.7KJ”¤mol-1£Ø2·Ö£©
¢Ś¼ÓŃ¹”¢½µĪĀ£Øø÷1·Ö£©
£Ø2£©¢ŁÕż¼«£Ø2·Ö£© CH4 - 8e- + 402-=CO2+2H2O £Ø2·Ö£©
¢Ś4OH”Ŗ- 4e-=2H2O+O2 £Ø2·Ö£© 1 £Ø2·Ö£© a”¢c £Ø2·Ö£©
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©¢Ła”¢N2£Øg£©+2O2£Øg£©=N2O4£Øg£©”÷H=10.7kJ”¤mol£1£»b”¢N2H4£Øg£©+O2£Øg£©=N2£Øg£©+2H2O£Øg£©”÷H=-543kJ”¤mol£1
ŅĄ¾ŻøĒĖ¹¶ØĀÉb”Į2-aµĆµ½ 2N2H4£Øg£©+N2O4£Øg£©=3N2£Øg£©+4H2O£Øg£©”÷H=-1096.7KJ”¤mol£1£»
“š°øĪŖ£ŗ2N2H4£Øg£©+N2O4£Øg£©=3N2£Øg£©+4H2O£Øg£©”÷H=-1096.7KJ”¤mol£1£»
¢ŚĖÄŃõ»Æ¶žµŖŌŚ“óĘųÖŠ»ņŌŚ½ĻøßĪĀ¶ČĻĀŗÜÄŃĪČ¶Ø“ęŌŚ£¬ĖüŗÜČŻŅ××Ŗ»ÆĪŖ¶žŃõ»ÆµŖ£¬ÓɶžŃõ»ÆµŖÖĘČ”ĖÄŃõ»Æ¶žµŖ£¬2NO2 N2O4£¬·“Ó¦ŹĒ·ÅČČ·“Ó¦£¬·“Ó¦Ē°ŗóĘųĢåĢå»ż¼õŠ”ĖłŅŌ·“Ó¦Ģõ¼žĪŖ£ŗŌö“óŃ¹Ē攢½µĪĀ¶¼ÓŠĄūÓŚ·“Ó¦ÕżĻņ½ųŠŠ£»
N2O4£¬·“Ó¦ŹĒ·ÅČČ·“Ó¦£¬·“Ó¦Ē°ŗóĘųĢåĢå»ż¼õŠ”ĖłŅŌ·“Ó¦Ģõ¼žĪŖ£ŗŌö“óŃ¹Ē攢½µĪĀ¶¼ÓŠĄūÓŚ·“Ó¦ÕżĻņ½ųŠŠ£»
¹Ź“š°øĪŖ£ŗŌö“óŃ¹Ē攢½µĪĀ£»
£Ø2£©¢ŁĶ¼1ŹĒŌµē³Ų£¬ŅĄ¾ŻµēĮ÷Į÷ĻņŹĒ“ÓÕż¼«Į÷Ļņøŗ¼«£¬cµē¼«ĪŖÕż¼«£¬ŃõĘųµĆµ½µē×Ó·¢Éś»¹Ō·“Ó¦£¬dµē¼«ĪŖµē³Ųøŗ¼«¼×ĶéŹĒµē×Ó·¢Éś»¹Ō·“Ó¦£¬ŌŚĮ½¼«ÉĻ·Ö±šĶØČėCH4ŗĶæÕĘų£¬ĘäÖŠ¹ĢĢåµē½āÖŹŹĒ²ōŌÓĮĖY2O3µÄZrO2¹ĢĢ壬ĖüŌŚøßĪĀĻĀÄÜ“«µ¼Ńō¼«Éś³ÉµÄO2£Ąė×Ó£¬½įŗĻµē×ÓŹŲŗ抓³öµē¼«·“Ó¦ĪŖ£ŗCH4- 8e-+4O2£=CO2+2H2O£»
“š°øĪŖ£ŗÕż¼«£» CH4- 8e-+4O2£=CO2+2H2O£»
¢ŚČēĶ¼2ĖłŹ¾µē½ā100mL0.5mol?L-1CuSO4ČÜŅŗ£¬·¢ÉśµÄµē½ā³Ų·“Ó¦ĪŖ£ŗ2CuSO4+2H2O 2Cu+O2”ü+2H2SO4£¬ÓėµēŌ“Õż¼«ĻąĮ¬µÄĪŖŃō¼«£¬ČÜŅŗÖŠ ĒāŃõøłĄė×ÓŹĒµē×Ó·¢ÉśŃõ»Æ·“Ó¦£¬µē¼«·“Ó¦ĪŖ£ŗ4OH£-4e£=2H2O+O2”ü£»Čōaµē¼«²śÉś56mL£Ø±ź×¼×“æö£©ĘųĢåĪŖŃõĘų£¬ĪļÖŹµÄĮæĪŖ0.0025mol£¬ĻūŗÄĒāŃõøłĄė×ÓĪļÖŹµÄĮæĪŖ0.01mol£¬ČÜŅŗÖŠÉś³ÉĒāĄė×ÓĪļÖŹµÄĮæĪŖ0.01mol£¬c£ØH£«£©=
2Cu+O2”ü+2H2SO4£¬ÓėµēŌ“Õż¼«ĻąĮ¬µÄĪŖŃō¼«£¬ČÜŅŗÖŠ ĒāŃõøłĄė×ÓŹĒµē×Ó·¢ÉśŃõ»Æ·“Ó¦£¬µē¼«·“Ó¦ĪŖ£ŗ4OH£-4e£=2H2O+O2”ü£»Čōaµē¼«²śÉś56mL£Ø±ź×¼×“æö£©ĘųĢåĪŖŃõĘų£¬ĪļÖŹµÄĮæĪŖ0.0025mol£¬ĻūŗÄĒāŃõøłĄė×ÓĪļÖŹµÄĮæĪŖ0.01mol£¬ČÜŅŗÖŠÉś³ÉĒāĄė×ÓĪļÖŹµÄĮæĪŖ0.01mol£¬c£ØH£«£©=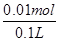 =0.1mol”¤L£1£¬PH=-lg0.1=1£»ŌņĖłµĆČÜŅŗµē½ā¹ż³ĢÖŠCuSO4ČÜŅŗĆæĖšŹ§2øöCuŌ×Ó£¬¾ĶĖšŹ§2øö OŌ×Ó£¬Ļąµ±ÓŚĖšŹ§Ņ»øöCuO£¬ĪŖĮĖŹ¹CuSO4ČÜŅŗ£¬»Öø“ŌÅØ¶Č£¬Ó¦¼ÓČėCuO£¬Ņ²æÉŅŌ¼ÓČėCuCO3£¬·ūŗĻ»Öø“ČÜŅŗÅØ¶ČµÄ¶ØĮæ¹ŲĻµ£¬µ«²»ÄܼÓČėCu£ØOH£©2”¢Cu2£ØOH£©2CO3£¬ŅņĪŖCuCO3+H2SO4
=0.1mol”¤L£1£¬PH=-lg0.1=1£»ŌņĖłµĆČÜŅŗµē½ā¹ż³ĢÖŠCuSO4ČÜŅŗĆæĖšŹ§2øöCuŌ×Ó£¬¾ĶĖšŹ§2øö OŌ×Ó£¬Ļąµ±ÓŚĖšŹ§Ņ»øöCuO£¬ĪŖĮĖŹ¹CuSO4ČÜŅŗ£¬»Öø“ŌÅØ¶Č£¬Ó¦¼ÓČėCuO£¬Ņ²æÉŅŌ¼ÓČėCuCO3£¬·ūŗĻ»Öø“ČÜŅŗÅØ¶ČµÄ¶ØĮæ¹ŲĻµ£¬µ«²»ÄܼÓČėCu£ØOH£©2”¢Cu2£ØOH£©2CO3£¬ŅņĪŖCuCO3+H2SO4 CuSO4+CO2”ü+H2O£¬Ļąµ±ÓŚ¼ÓCuO£¬¶ųCu£ØOH£©2+H2SO4
CuSO4+CO2”ü+H2O£¬Ļąµ±ÓŚ¼ÓCuO£¬¶ųCu£ØOH£©2+H2SO4 CuSO4+2H2O”¢Cu2£ØOH£©2CO3+2H2SO4=2CuSO4 +CO2”ü+3H2O£¬³żŌö¼ÓČÜÖŹĶā»¹Ōö¼ÓĮĖĖ®£»Ń”ac£®
CuSO4+2H2O”¢Cu2£ØOH£©2CO3+2H2SO4=2CuSO4 +CO2”ü+3H2O£¬³żŌö¼ÓČÜÖŹĶā»¹Ōö¼ÓĮĖĖ®£»Ń”ac£®
“š°øĪŖ£ŗ4OH”Ŗ- 4e-=2H2O+O2 £»1£»ac£»
æ¼µć£ŗČČ»Æѧ·½³ĢŹ½£»»ÆѧµēŌ“ŠĀŠĶµē³Ų


 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø1£© 8gŅŗĢ¬µÄCH3OHŌŚŃõĘųÖŠĶźČ«Č¼ÉÕ£¬Éś³É¶žŃõ»ÆĢ¼ĘųĢåŗĶŅŗĢ¬Ė®Ź±ŹĶ·Å³öQ kJµÄČČĮ攣ŹŌŠ“³öŅŗĢ¬CH3OHČ¼ÉÕČȵÄČČ»Æѧ·½³ĢŹ½ ”£
£Ø2£©ŌŚ»Æѧ·“Ó¦¹ż³ĢÖŠ£¬ĘĘ»µ¾É»Æѧ¼üŠčŅŖĪüŹÕÄÜĮ棬ŠĪ³ÉŠĀ»Æѧ¼üÓÖ»įŹĶ·ÅÄÜĮ攣
| »Æѧ¼ü | H”ŖH | N”ŖH | N”ŌN |
| ¼üÄÜ/kJ·mol£1 | 436 | 391 | 945 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(1)CH4(g )+2O2(g )=CO2(g )+2H2O(g ) ¦¤H=-802.3kJ/mol
øĆČČ»Æѧ·“Ó¦·½³ĢŹ½µÄŅāŅåŹĒ_____________________________________”£
(2)ŅŃÖŖ2gŅŅ“¼ĶźČ«Č¼ÉÕÉś³ÉŅŗĢ¬Ė®·Å³öQ kJµÄČČĮ棬Š“³ö±ķŹ¾ŅŅ“¼Č¼ÉÕČȵÄČČ»Æѧ·½
³ĢŹ½£ŗ____________________________________________________________.
(3)ŅŃÖŖ²šæŖ1mol H-H¼ü£¬1mol N-H¼ü£¬1mol 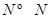 ¼ü·Ö±šŠčŅŖµÄÄÜĮæŹĒ436kJ”¢391KJ”¢946kJ£¬ŌņN2ÓėH2·“Ӧɜ³É1mol NH3(g)µÄČČ»Æѧ·½³ĢŹ½ŹĒ___________________.
¼ü·Ö±šŠčŅŖµÄÄÜĮæŹĒ436kJ”¢391KJ”¢946kJ£¬ŌņN2ÓėH2·“Ӧɜ³É1mol NH3(g)µÄČČ»Æѧ·½³ĢŹ½ŹĒ___________________.
(4)ŅĄ¾ŻøĒĖ¹¶ØĀÉæÉŅŌ¶ŌijŠ©ÄŃŅŌĶعżŹµŃéÖ±½Ó²ā¶ØµÄ»Æѧ·“Ó¦µÄģŹ±ä½ųŠŠĶĘĖć”£
ŅŃÖŖ£ŗC(ŹÆÄ«£¬s)+O2(g)=CO2(g) ¦¤H=-393.5kJ/mol ¢Ł
2H2(g)+O2(g)=2H2O(l) ¦¤H=-571.6kJ/mol ¢Ś
2C2H2(g)+5O2(g)=4CO2(g)+2H2O(l) ¦¤H=-2599kJ/mol ¢Ū
øł¾ŻøĒĖ¹¶ØĀÉ£¬¼ĘĖć298KŹ±ÓÉC£ØŹÆÄ«£¬s£©ŗĶH2(g)Éś³É1mol C2H2(g)·“Ó¦µÄģŹ±ä£ŗ
____________________________.
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(14·Ö)2014Äź10ŌĀ³õ£¬Īķö²ĢģĘų¶ą“ĪĖĮÅ°ŗÓ±±”¢Ģģ½ņ”¢±±¾©µČµŲĒų”£ĘäÖŠ£¬Č¼ĆŗŗĶĘū³µĪ²ĘųŹĒŌģ³ÉæÕĘųĪŪČ¾µÄŌŅņÖ®Ņ»”£
£Ø1£©Ęū³µĪ²Ęų¾»»ÆµÄÖ÷ŅŖŌĄķĪŖ£ŗ2NO(g) + 2CO(g) 2CO2(g)+ N2(g)”£”÷H£¼0
2CO2(g)+ N2(g)”£”÷H£¼0
¢ŁøĆ·“Ó¦µÄĘ½ŗā³£Źż±ķ“ļŹ½ ”£
¢ŚČōøĆ·“Ó¦ŌŚ¾ųČČ”¢ŗćČŻµÄĆܱÕĢåĻµÖŠ½ųŠŠ£¬ĻĀĮŠŹ¾ŅāĶ¼ÕżČ·ĒŅÄÜĖµĆ÷·“Ó¦ŌŚ½ųŠŠµ½t1Ź±æĢ“ļµ½Ę½ŗāדĢ¬µÄŹĒ £ØĢī“śŗÅ£©”£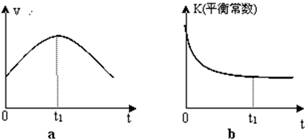
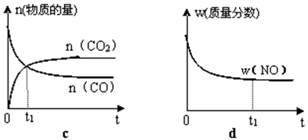
£Ø2£©Ö±½ÓÅÅ·ÅĆŗČ¼ÉÕ²śÉśµÄŃĢĘų»įŅżĘšŃĻÖŲµÄ»·¾³ĪŹĢā”£ĆŗČ¼ÉÕ²śÉśµÄŃĢĘųŗ¬µŖµÄŃõ»ÆĪļ£¬ÓĆCH4“߻ƻ¹ŌNOxæÉŅŌĻū³żµŖŃõ»ÆĪļµÄĪŪČ¾”£
ŅŃÖŖ£ŗCH4(g)+2NO2(g)£½N2(g)£«CO2(g)+2H2O(g) ”÷H£½£867 kJ/mol
2NO2(g)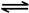 N2O4(g) ”÷H£½£56.9 kJ/mol
N2O4(g) ”÷H£½£56.9 kJ/mol
H2O(g) £½ H2O(l) ¦¤H £½ £44.0 kJ£Æmol
Š“³öCH4“߻ƻ¹ŌN2O4(g)Éś³ÉN2ŗĶH2O(l)µÄČČ»Æѧ·½³ĢŹ½£ŗ ”£
£Ø3£©ŌŚŅ»¶ØĢõ¼žĻĀ£¬Ņ²æÉŅŌÓĆNH3“¦ĄķNOx”£ŅŃÖŖNOÓėNH3·¢Éś·“Ӧɜ³ÉN2ŗĶH2O£¬ĻÖÓŠNOŗĶNH3µÄ»ģŗĻĪļ1mol£¬³ä·Ö·“Ó¦ŗóµĆµ½µÄ»¹Ō²śĪļ±ČŃõ»Æ²śĪļ¶ą1.4 g£¬ŌņŌ·“Ó¦»ģŗĻĪļÖŠNOµÄĪļÖŹµÄĮææÉÄÜŹĒ_____________”£
£Ø4£©ŅŌ¼×ĶéĪŖŌĮĻÖĘČ”ĒāĘųŹĒ¹¤ŅµÉĻ³£ÓƵÄÖĘĒā·½·Ø”£Ōņ2 molCH4Óė×ćĮæH2O£Øg£©·“Ó¦×ī¶ąæÉÉś³É_______mol H2£¬Š“³öøĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½_________________________________________________”£
(5)ÉĻŹö·½·ØÖʵƵÄH2æÉŅŌŗĶCOŌŚŅ»¶ØĢõ¼žĻĀŗĻ³É¼×“¼ŗĶ¶ž¼×ĆŃ£ØCH3OCH3£©¼°Šķ¶ąĢžĄąĪļÖŹ”£µ±Į½ÕßŅŌĪļÖŹµÄĮæ1:1“߻Ʒ“Ó¦£¬ĘäŌ×ÓĄūÓĆĀŹ“ļ100%£¬ŗĻ³ÉµÄĪļÖŹæÉÄÜŹĒ ”£
a.ĘūÓĶ b.¼×“¼ c.¼×Č© d.ŅŅĖį
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
æĘѧ¼ŅĄūÓĆĢ«ŃōÄÜ·Ö½āĖ®Éś³ÉµÄĒāĘųŌŚ“߻ƼĮ×÷ÓĆĻĀ Óė¶žŃõ»ÆĢ¼·“Ӧɜ³É¼×“¼£¬²¢æŖ·¢³öÖ±½ÓŅŌ¼×“¼ĪŖČ¼ĮĻµÄČ¼ĮĻµē³Ų”£ŅŃÖŖ£ŗH2(g)”¢CO(g)ŗĶCH3OH£Ø1£©µÄČ¼ÉÕČČ”÷H·Ö±šĪŖ-285.8 kJ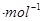 £®
£® ”¢Ņ»283.0 kJ
”¢Ņ»283.0 kJ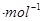 ŗĶŅ»726.5£®kJ
ŗĶŅ»726.5£®kJ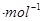 ”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓĆĢ«ŃōÄÜ·Ö½ā10mol H2O(1)ĻūŗĵÄÄÜĮæŹĒ________kJ.
£Ø2£©¼×“¼²»ĶźČ«Č¼ÉÕÉś³ÉŅ»Ńõ»ÆĢ¼ŗĶŅŗĢ¬Ė®µÄČČ»Æѧ·½³ĢŹ½ĪŖ£ŗ
__________________________________________________________________________.
£Ø3£©ŌŚČŻ»żĪŖ2LµÄĆܱÕČŻĘ÷ÖŠ£¬ÓÉCO2ŗĶH2ŗĻ³É¼×“¼£¬ŌŚĘäĖūĢõ¼ž²»±äµÄĒéæöĻĀ£¬
æ¼²éĪĀ¶Č¶Ō·“Ó¦µÄÓ°Ļģ£¬ŹµŃé½į¹ūČēĻĀĶ¼ĖłŹ¾£Ø×¢£ŗ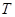 ”¢
”¢ ¾ł“óÓŚ300”ę£©£ŗ
¾ł“óÓŚ300”ę£©£ŗ
ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ_______________£ØĢīŠņŗÅ£©
¢ŁĪĀ¶ČĪŖ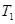 Ź±£¬“Ó·“Ó¦æŖŹ¼µ½·“Ó¦“ļµ½Ę½ŗā£¬Éś³É¼×“¼µÄĘ½¾łĖŁĀŹĪŖ£ŗ
Ź±£¬“Ó·“Ó¦æŖŹ¼µ½·“Ó¦“ļµ½Ę½ŗā£¬Éś³É¼×“¼µÄĘ½¾łĖŁĀŹĪŖ£ŗ
¢ŚøĆ·“Ó¦ŌŚ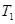 Ź±µÄĘ½ŗā³£Źż±Č
Ź±µÄĘ½ŗā³£Źż±Č Ź±µÄŠ”
Ź±µÄŠ”
¢ŪøĆ·“Ó¦ĪŖ·ÅČČ·“Ó¦
¢Ü“¦ÓŚAµćµÄ·“Ó¦ĢåĻµµÄĪĀ¶Č“Ó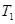 ±äµ½
±äµ½ £¬“ļµ½Ę½ŗāŹ±
£¬“ļµ½Ę½ŗāŹ±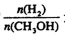 Ōö“ó
Ōö“ó
£Ø4£©ŌŚ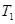 ĪĀ¶ČŹ±£¬½«1mol CO2ŗĶ3mol H2³äČėŅ»ĆܱÕŗćČŻČŻĘ÷ÖŠ£¬³ä·Ö·“Ó¦“ļµ½Ę½ŗāŗó£¬ČōCO2µÄ×Ŗ»ÆĀŹĪŖa£¬Ōņ“ĖŹ±ČŻĘ÷ÄŚµÄŃ¹ĒæÓėĘšŹ¼Ń¹ĒæÖ®±ČĪŖ___________”£
ĪĀ¶ČŹ±£¬½«1mol CO2ŗĶ3mol H2³äČėŅ»ĆܱÕŗćČŻČŻĘ÷ÖŠ£¬³ä·Ö·“Ó¦“ļµ½Ę½ŗāŗó£¬ČōCO2µÄ×Ŗ»ÆĀŹĪŖa£¬Ōņ“ĖŹ±ČŻĘ÷ÄŚµÄŃ¹ĒæÓėĘšŹ¼Ń¹ĒæÖ®±ČĪŖ___________”£
£Ø5£©ŌŚÖ±½ÓŅŌ¼×“¼ĪŖČ¼ĮĻµÄČ¼ĮĻµē³ŲÖŠ£¬µē½āÖŹČÜŅŗĪŖĖįŠŌ£¬øŗ¼«µÄ·“Ó¦Ź½ĪŖ___________________;Õż¼«µÄ·“Ó¦Ź½ĪŖ_____________________________________.ĄķĻėדĢ¬ĻĀ£¬øĆČ¼ĮĻµē³ŲĻūŗÄlmol¼×“¼ĖłÄܲśÉśµÄ×ī“óµēÄÜĪŖ701.8kJ£¬ŌņøĆČ¼ĮĻµē³ŲµÄĄķĀŪŠ§ĀŹĪŖ_______________£ØČ¼ĮĻµē³ŲµÄĄķĀŪŠ§ĀŹŹĒÖøµē³ŲĖł²śÉśµÄ×ī“óµēÄÜÓėČ¼ĮĻµē³Ų·“Ó¦ĖłÄÜŹĶ·ÅµÄČ«²æÄÜĮæÖ®±Č£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąµŲĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶Ö®Ņ»”£
(1)ŌŚ250C 101kPaŹ±£¬H2ÓėO2»ÆŗĻÉś³É1mol H2O(g)·Å³ö241.8kJµÄČČĮ棬ĘäČČ»Æѧ·½³ĢŹ½ĪŖ
___________
ÓÖÖŖ: ¢ŁC(s)£«O2(g)ØTCO2(g) ”÷H£½£393.5kJ/mol
¢ŚCO(g)£«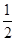 O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
½¹ĢæÓėĖ®ÕōĘų·“Ó¦ŹĒ½«¹ĢĢåĆŗ±äĪŖĘųĢåČ¼ĮĻµÄ·½·Ø£¬C(s)£«H2O(g)ØTCO(g)£«H2(g) ”÷H=____kJ/mol
(2) COæÉŅŌÓėH2O(g)½ųŅ»²½·¢Éś·“Ó¦: CO(g)£«H2O(g) CO2(g)£«H2(g) ”÷H£¼0ŌŚŗćČŻĆܱÕČŻĘ÷ÖŠ£¬ĘšŹ¼Ź±n(H2O)=0.20mol£¬n(CO)£½0.10 mol,ŌŚ8000CŹ±“ļµ½Ę½ŗāדĢ¬£¬K£½1.0£¬ŌņĘ½ŗāŹ±£¬ČŻĘ÷ÖŠCOµÄ×Ŗ»ÆĀŹŹĒ_____________(¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż)”£
CO2(g)£«H2(g) ”÷H£¼0ŌŚŗćČŻĆܱÕČŻĘ÷ÖŠ£¬ĘšŹ¼Ź±n(H2O)=0.20mol£¬n(CO)£½0.10 mol,ŌŚ8000CŹ±“ļµ½Ę½ŗāדĢ¬£¬K£½1.0£¬ŌņĘ½ŗāŹ±£¬ČŻĘ÷ÖŠCOµÄ×Ŗ»ÆĀŹŹĒ_____________(¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż)”£
(3) ¹¤ŅµÉĻ“ÓĆŗĘų»ÆŗóµÄ»ģŗĻĪļÖŠ·ÖĄė³öH2£¬½ųŠŠ°±µÄŗĻ³É£¬ŅŃÖŖ·“Ó¦·“Ó¦N2(g)£«3H2(g 2NH3(g)£Ø”÷H£¼0£©ŌŚµČČŻĢõ¼žĻĀ½ųŠŠ£¬øıäĘäĖū·“Ó¦Ģõ¼ž£¬ŌŚI”¢II”¢III½×¶ĪĢåĻµÖŠø÷ĪļÖŹÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĻĀĶ¼ĖłŹ¾£ŗ
2NH3(g)£Ø”÷H£¼0£©ŌŚµČČŻĢõ¼žĻĀ½ųŠŠ£¬øıäĘäĖū·“Ó¦Ģõ¼ž£¬ŌŚI”¢II”¢III½×¶ĪĢåĻµÖŠø÷ĪļÖŹÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĻĀĶ¼ĖłŹ¾£ŗ
¢ŁN2µÄĘ½¾ł·“Ó¦ĖŁĀŹv1(N2)”¢vII(N2)”¢vIII(N2)“ӓ󵽊”ÅÅĮŠ“ĪŠņĪŖ________£»
¢ŚÓɵŚŅ»“ĪĘ½ŗāµ½µŚ¶ž“ĪĘ½ŗā£¬Ę½ŗāŅĘ¶ÆµÄ·½Ļņ ŹĒ________£¬²ÉČ”µÄ“ėŹ©ŹĒ________”£
¢Ū±Č½ĻµŚII½×¶Ī·“Ó¦ĪĀ¶Č(T2)ŗĶµŚIII½×¶Ī·“Ó¦ĖŁ¶Č£ØT3)µÄøßµĶ£ŗT2________T3Ģī”°”µ”¢=”¢<”±ÅŠ¶ĻµÄĄķÓÉŹĒ________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
°±ĘųŹĒÉś²ś»Æ·Ź”¢ĻõĖįµČµÄÖŲŅŖŌĮĻ£¬Ī§ČĘŗĻ³É°±ČĖĆĒ½ųŠŠĮĖŅ»ĻµĮŠµÄŃŠ¾æ
£Ø1£©ĒāĘų¼ČÄÜÓėµŖĘųÓÖÄÜÓėŃõĘų·¢Éś·“Ó¦£¬µ«ŹĒ·“Ó¦µÄĢõ¼žČ“²»ĻąĶ¬”£
ŅŃÖŖ£ŗ2H2 (g) + O2 (g) = 2H2O (g) ¦¤H =" -483.6" kJ/mol
3H2 (g) + N2 (g)  2NH3 (g) ¦¤H =" -92.4" kJ/mol
2NH3 (g) ¦¤H =" -92.4" kJ/mol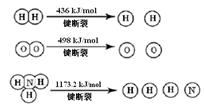
¼ĘĖć¶ĻĮŃ1 mol N”ŌN¼üŠčŅŖÄÜĮæ kJ £¬ µŖĘų·Ö×ÓÖŠ»Æѧ¼ü±ČŃõĘų·Ö×ÓÖŠµÄ»Æѧ¼ü¼ü £ØĢī”°Ēæ”±»ņ”°Čõ”±£©£¬Ņņ“ĖĒāĘųÓė¶žÕß·“Ó¦µÄĢõ¼ž²»Ķ¬”£
£Ø2£©¹ĢµŖŹĒæĘѧ¼ŅÖĀĮ¦ŃŠ¾æµÄÖŲŅŖæĪĢā”£×ŌČ»½ēÖŠ“ęŌŚĢģČ»µÄ“óĘų¹ĢµŖ¹ż³Ģ£ŗN2 (g) + O2 (g) =" 2NO" (g) ¦¤H =" +180.8" kJ/mol £¬¹¤ŅµŗĻ³É°±ŌņŹĒČĖ¹¤¹ĢµŖ”£
·ÖĪöĮ½ÖÖ¹ĢµŖ·“Ó¦µÄĘ½ŗā³£Źż£¬ĻĀĮŠ½įĀŪÕżČ·µÄŹĒ ”£
| ·“Ó¦ | “óĘų¹ĢµŖ | ¹¤Ņµ¹ĢµŖ | ||||
| ĪĀ¶Č/”ę | 27 | 2000 | 25 | 350 | 400 | 450 |
| K | 3.84”Į10-31 | 0.1 | 5”Į108 | 1.847 | 0.507 | 0.152 |
 2NH3 (g)²āµĆ¼×ČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹĪŖ40%”£
2NH3 (g)²āµĆ¼×ČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹĪŖ40%”£| | N2 | H2 | NH3 |
| ¼× | 1 | 3 | 0 |
| ŅŅ | 0.5 | 1.5 | 1 |
| ±ū | 0 | 0 | 4 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
æŖ·¢”¢Ź¹ÓĆĒå½ąÄÜŌ“·¢Õ¹”°µĶĢ¼¾¼Ć”±£¬Õż³ÉĪŖæĘѧ¼ŅŃŠ¾æµÄÖ÷ŅŖæĪĢā”£ĒāĘų”¢¼×“¼ŹĒÓÅÖŹµÄĒå½ąČ¼ĮĻ£¬æÉÖĘ×÷Č¼ĮĻµē³Ų”£
£Ø1£©ŅŃÖŖ£ŗ¢Ł 2CH3OH(1) + 3O2(g) = 2CO2(g) + 4H2O(g) ¦¤H1 =" ØC" 1275.6 kJ/mol
¢Ś 2CO(g) + O2(g) = 2CO2(g) ¦¤H2 =" ØC" 566.0 kJ/mol
¢Ū H2O(g) = H2O(1) ¦¤H3 =" ØC" 44.0 kJ/mol
Š“³ö¼×“¼²»ĶźČ«Č¼ÉÕÉś³ÉŅ»Ńõ»ÆĢ¼ŗĶŅŗĢ¬Ė®µÄČČ»Æѧ·½³ĢŹ½£ŗ___________”£
£Ø2£©Éś²ś¼×“¼µÄŌĮĻCOŗĶH2Ą“Ō“ÓŚ£ŗCH4(g) + H2O(g)  CO(g) + 3H2(g) ¦¤H>0
CO(g) + 3H2(g) ¦¤H>0
¢ŁŅ»¶ØĢõ¼žĻĀCH4µÄĘ½ŗā×Ŗ»ÆĀŹÓėĪĀ¶Č”¢Ń¹ĒæµÄ¹ŲĻµČēĶ¼a”£ŌņTl ________T2(Ģī”°<”±”¢”°>”±”¢”°=”±£¬ĻĀĶ¬)£»A”¢B”¢CČżµć“¦¶ŌÓ¦Ę½ŗā³£Źż£ØKA”¢KB”¢KC£©µÄ“󊔹ŲĻµĪŖ___________”£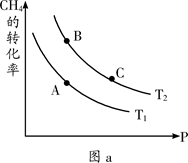
¢Ś100”ꏱ£¬½«1 mol CH4ŗĶ2 mol H2OĶØČėČŻ»żĪŖ1 LµÄ¶ØČŻĆÜ·āČŻĘ÷ÖŠ£¬·¢Éś·“Ó¦£¬ÄÜĖµĆ÷øĆ·“Ó¦ŅŃ¾“ļµ½Ę½ŗāדĢ¬µÄŹĒ__________
a£®ČŻĘ÷ÄŚĘųĢåĆܶČŗć¶Ø
b£®µ„Ī»Ź±¼äÄŚĻūŗÄ0.1 mol CH4Ķ¬Ź±Éś³É0.3 mol H2
c£®ČŻĘ÷µÄŃ¹Ēæŗć¶Ø
d£®3vÕż(CH4) = vÄę(H2)
Čē¹ū“ļµ½Ę½ŗāŹ±CH4µÄ×Ŗ»ÆĀŹĪŖ0.5£¬Ōņ100”ꏱøĆ·“Ó¦µÄĘ½ŗā³£ŹżK =___________
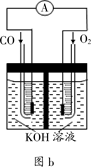
£Ø3£©Ä³ŹµŃ銔×éĄūÓĆCO(g) ”¢ O2(g) ”¢KOH£Øaq£©Éč¼Ę³ÉČēĶ¼bĖłŹ¾µÄµē³Ų×°ÖĆ£¬ŌņøƵē³Ųøŗ¼«µÄµē¼«·“Ó¦Ź½ĪŖ___________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĪŹ“šĢā
£Ø14·Ö£©ŃŠ¾æŅų¼°Ęä»ÆŗĻĪļ¾ßÓŠÖŲŅŖŅāŅ唣
£Øl£©ŅŃÖŖ£ŗ
Ag2O£Øs£©+2HC1£Øg£© 2AgC1£Øs£©+H2O£Ø1£© ”÷H1=£324£®4 kJ”¤mo1£1
2Ag£Øs£©+1/2O2£Øg£© Ag2O£Øs£© ”÷H2=£30£®6 kJ”¤mo1£1
H2£Øg£©+C12£Øg£© 2HC1£Øg£© ”÷H3=£184£®4 kJ£®”¤mo1£1
2H2£Ø9£©+O2£Øg£© 2H2O£Ø1£© ”÷H4=£571£®2 l”¤mo1£1
Š“³öĀČĘųÓėŅųÉś³É¹ĢĢåĀČ»ÆŅųµÄČČ»Æѧ·½³ĢŹ½________”£
£Ø2£©ĆĄĄöµÄŅųŹĪ³£ÓĆFe(NO3)3ČÜŅŗŹ“æĢ£¬Š“³öFe3+ÓėAg·“Ó¦µÄĄė×Ó·½³ĢŹ½___ _£»ŅŖÅŠ¶ØFe(NO3)3ČÜŅŗÖŠNO3”ŖŹĒ·ńŌŚŅųŹĪŹ“æĢÖŠ·¢Éś·“Ó¦£¬æÉČ” µÄĻõĖįČÜŅŗ£¬Č»ŗóøł¾ŻĘäŹĒ·ńÓėAg·¢Éś·“Ó¦Ą“ÅŠ¶Ø”£
£Ø3£©ŅųŠæ¼īŠŌµē³ŲµÄµē½āÖŹČÜŅŗĪŖKOHČÜŅŗ£¬·ÅµēŹ±£¬Õż¼«Ag2O2×Ŗ»ÆĪŖAg£¬øŗ¼«Zn×Ŗ»ÆĪŖZn(OH)2£¬ŌņÕż¼«·“Ó¦Ź½ĪŖ £¬øŗ¼«ø½½üČÜŅŗµÄpH ___ £ØĢī”°Ōö“ó”±”¢”°²»±ä”±»ņ”°¼õŠ””±£©”£
£Ø4£©µē½ā·Ø¾«Į¶ŅųŹ±£¬“ÖŅųÓ¦ÓėÖ±Į÷µēŌ“µÄ ¼«ĻąĮ¬£¬µ±ÓĆAgNO3ŗĶHNO3»ģŗĻČÜŅŗ×öµē½āÖŹČÜŅŗŹ±£¬·¢ĻÖŅõ¼«ÓŠÉŁĮæŗģ×ŲÉ«ĘųĢ壬Ōņ²śÉśøĆĻÖĻóµÄµē¼«·“Ó¦Ź½ĪŖ____”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com