

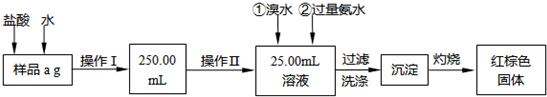
Ēėøł¾ŻÉĻĆęĮ÷³Ģ£¬»Ų“šŅŌĻĀĪŹĢā£ŗ
(1)²Ł×÷¢ńĖłÓƵ½µÄ²£Į§ŅĒĘ÷³żÉÕ±”¢²£Į§°ōĶā£¬»¹±ŲŠėÓŠ___________”¢___________(ĢīŅĒĘ÷Ćū³Ę)£¬²Ł×÷¢ņ±ŲŠėÓƵ½µÄŅĒĘ÷ŹĒ___________(Ģī±ąŗÅ)”£
A.50 mLÉÕ± B.50 mLĮæĶ² C.100 mLĮæĶ² D.25 mLµĪ¶Ø¹Ü
(2)ĒėŠ“³ö¼ÓČėäåĖ®·¢ÉśµÄĄė×Ó·“Ó¦·½³ĢŹ½________________________£¬¼ÓČė°±Ė®ŅŖ¹żĮæµÄŌŅņŹĒ________________________”£
(3)¼ģŃé³ĮµķŹĒ·ńŅŃ¾Ļ“µÓøɾ»µÄ²Ł×÷ŹĒ_________________________________________”£
(4)½«³ĮµķĪļ¼ÓČČ£¬ĄäČ“ÖĮŹŅĪĀ£¬ÓĆĢģĘ½³ĘĮæĘäÖŹĮæĪŖb
(5)ČōÕō·¢ĆóÖŹĮæŹĒW
(1)250 mLČŻĮæĘæ ½ŗĶ·µĪ¹Ü D
(2)2Fe2++Br2![]() 2Fe3++2Br- Ź¹Fe3+³ä·Ö³Įµķ
2Fe3++2Br- Ź¹Fe3+³ä·Ö³Įµķ
(3)ȔɣĮæ×īŗóŅ»“ĪĻ“³öŅŗ£¬µĪ¼ÓAgNO3ČÜŅŗ£¬ČōĪŽ³ĮµķÉś³É£¬ŌņÖ¤Ć÷Ļ“µÓøɾ»(ŗĻĄķµÄ“š°ø¾łæÉ)
(4)ŌŁ“Ī¼ÓČČĄäČ“²¢³ĘĮ棬ֱÖĮĮ½“ĪÖŹĮæ²īŠ”ÓŚ0.1 g
(5)ĢśŌŖĖŲµÄÖŹĮæ·ÖŹżŹĒ![]() ”Į100% ¹ĢĢå¼ÓČČŹ±Ī“³ä·Ö·“Ó¦±äĪŖFe2O3
”Į100% ¹ĢĢå¼ÓČČŹ±Ī“³ä·Ö·“Ó¦±äĪŖFe2O3
½āĪö£ŗŌĶĮĢāÄæ¼°·ÖĪöĮ÷³ĢĶ¼æÉÖŖ£¬±¾ŹµŃéÄæµÄŹĒ²ā¶ØĢśµÄÖŹĮæ·ÖŹż£¬²ÉČ”µÄ·½·ØŹĒŹ¹ŃłĘ·Čܽā”¢·“Ó¦”¢×īÖÕÉś³ÉŃõ»ÆĢś£¬Č»ŗóĶعżŃõ»ÆĢśÖŹĮæĄ“ĒóĢśµÄÖŹĮæ·ÖŹż”£¹ŹĪŹĢā(1)²Ł×÷¢ń»¹ŠčŅŖ250 mLČŻĮæĘ攢½ŗĶ·µĪ¹Ü£¬²Ł×÷¢ņŠčµĪ¶Ø¹Ü”£ĪŹĢā(2)ÖŠ¼ÓäåĖ®¾ĶŹĒČĆ+2¼ŪĢś±äĪŖ+3¼Ū£¬¼Ó°±Ė®¾ĶŹĒŹ¹+3¼ŪĢś³ä·Ö³Įµķ”£³ĮµķĻ“µÓ¾ĶŹĒ°ŃBr-»ņ![]() Ļ“µō£¬¹Źæɽā“šĪŹĢā”£(3)Į½“Ī³ĘĮæµÄĪó²īĻą²ī²»Äܳ¬¹ż
Ļ“µō£¬¹Źæɽā“šĪŹĢā”£(3)Į½“Ī³ĘĮæµÄĪó²īĻą²ī²»Äܳ¬¹ż



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| 7(W2-W1) |
| a |
| 7(W2-W1) |
| a |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| 112(W2-W1) |
| 160a |
| 112(W2-W1) |
| 160a |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗŗž±±Ź”»ĘøŌŹŠ2009”Ŗ2010ѧğ¶Čøßȿğ¼¶ĘŚÄ©æ¼ŹŌĄķæĘ×ŪŗĻÄÜĮ¦²āŹŌ»ÆѧŹŌĢā ĢāŠĶ£ŗŹµŃéĢā
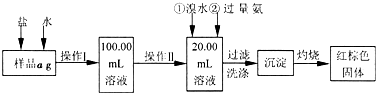
ijĀČ»ÆĢśŃłĘ·ŗ¬ÓŠÉŁĮæFeCl2ŌÓÖŹ”£ĻÖŅŖ²ā¶ØĘäÖŠĢśŌŖĖŲµÄÖŹĮæ·ÖŹż£¬ŹµŃé°“ŅŌĻĀ²½Öč½ųŠŠ£ŗ
Ēėøł¾ŻÉĻĆęĮ÷³Ģ£¬»Ų“šŅŌĻĀĪŹĢā£ŗ
£Ø1£©²Ł×÷IĖłÓƵ½µÄ²£Į§ŅĒĘ÷³żÉÕ±£®²£Į§°ō£®ĮæĶ²Ķā£¬»¹±ŲŠėÓŠ____________ £ØĢī×ŌŃ”ŅĒĘ÷Ćū³Ę£©£¬²Ł×÷II±ŲŠėÓƵ½µÄŅĒĘ÷ŹĒ____________£ØĢī±ąŗÅ£©”£
””””
| A£®50mLÉÕ± | B£®50mLĮæĶ² ”””” | C£®25mL¼īŹ½µĪ¶Ø¹Ü | D£®25mLĖįŹ½µĪ¶Ø¹Ü |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012Ń§ÄźÉ½¶«Ź”¼ĆÄžŹŠøßČżµŚ¶ž“ĪÖŹĮæ¼ģ²ā»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø15·Ö£©Ä³ĀČ»ÆĢśŃłĘ·ŗ¬ÓŠÉŁĮæFeCl2ŌÓÖŹ”£ĻÖŅŖ²ā¶ØĘäÖŠĢśŌŖĖŲµÄÖŹĮæ·ÖŹż£¬ŹµŃé°“ŅŌĻĀ²½Öč½ųŠŠ£ŗ

Ēėøł¾ŻÉĻĆęĮ÷³Ģ£¬»Ų“šŅŌĻĀĪŹĢā£ŗ
£Ø1£©²Ł×÷IĖłÓƵ½µÄ²£Į§ŅĒĘ÷³żÉÕ±£®²£Į§°ō£®ĮæĶ²Ķā£¬»¹±ŲŠėÓŠ____________ £ØĢīŅĒĘ÷Ćū³Ę£©£¬²Ł×÷II±ŲŠėÓƵ½µÄŅĒĘ÷ŹĒ____________£ØĢī±ąŗÅ£©”£
A£®50mLÉÕ±”” B£®50mLĮæĶ²
C£®25mL¼īŹ½µĪ¶Ø¹Ü”” D£®25mLĖįŹ½µĪ¶Ø¹Ü
£Ø2£©Ļ“µÓŹĒĻ“Č„ø½×ÅŌŚ³ĮµķÉĻµÄ____________”££ØŠ“Ąė×Ó·ūŗÅ£©
£Ø3£©¼ģŃé³ĮµķŹĒ·ńŅŃ¾Ļ“µÓøɾ»µÄ²Ł×÷ŹĒ________________________”£
£Ø4£©½«³ĮµķĪļ¼ÓČČ£¬²¢ÖĆÓŚøÉŌļĘ÷ÖŠĄäČ“ÖĮŹŅĪĀ£¬ÓĆĢģĘ½³ĘĮæĘäÖŹĮæĪŖb1g£¬ŌŁ“Ī¼ÓČČ²¢ĄäČ“ÖĮŹŅĪĀ³ĘĮæĘäÖŹĮæĪŖb2g£¬Čōb1£b2=0.3g£¬Ōņ½ÓĻĀĄ“»¹Ó¦½ųŠŠµÄ²Ł×÷ŹĒ_________”£
£Ø5£©ČōÕō·¢ĆóÖŹĮæŹĒW1g£¬Õō·¢ĆóÓė¼ÓČČŗó¹ĢĢå×ÜÖŹĮæŹĒW2g£¬Ōņѳʷ֊ĢśŌŖĖŲµÄÖŹĮæ·ÖŹżŹĒ____________”£
£Ø6£©ÓŠĶ¬Ń§ČĻĪŖ£ŗÉĻŹöĮ÷³ĢÖŠČō²»¼ÓČėĀČĖ®£¬ĘäĖü²½Öč²»±ä£¬ČŌæÉ“ļµ½ÄæµÄ”£ĖūµÄĄķÓÉŹĒ________________________”££ØÓĆ»Æѧ·½³ĢŹ½±ķŹ¾£©
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com