
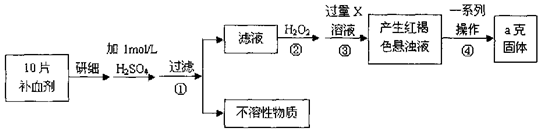
���� ������ͼ��֪����ʵ��ԭ��Ϊ����ҩƷ�е�Fe2+�γ���Һ����Fe2+����ΪFe3+��ʹFe3+ת��Ϊ����������������ת��Ϊ��������ͨ���ⶨ�����������������㲹Ѫ������Ԫ�صĺ�����
��1��˫��ˮ���������ԣ���ԭ������ˮ��
��2����������������ͼӦ������������������
��3���������һϵ�д�������������������Һ����ת��Ϊ����������Ҫ���ˡ�ϴ�ӵ�����������Ȼ������������������
��4��������Ԫ���غ��������չ���ΪFe2O3��
��5���ٸ�������һ�����ʵ���Ũ�ȵ���Һ����Ҫ���������ش�
�����ᡢ���������ǿ�����ԡ�������л�ԭ�ԣ�
�۸�����ؾ���ǿ�����ԣ��ܸ�ʴ��ʽ�ζ��ܵ���Ƥ�ܣ�Ӧ����ʽ�ζ���ʢװ���ζ�ʱΪ���ڹ۲���ɫ�仯���ζ��յ���ɫ��dz�������ڹ۲죻
��6��������Ԫ���غ���м��㣮
��� �⣺��1��˫��ˮ���������ԣ��ܽ�Fe2+ȫ������ΪFe3+��
�ʴ�Ϊ����Fe2+ȫ������ΪFe3+��
��2������ۼ������Һ����������Ӧ���������������ɫ���������Լ����XΪ���Ӧ�����ӷ���ʽΪ��Fe3++3OH-=Fe��OH��3������Fe3++3NH3•H2O=Fe��OH��3��+3NH4+����
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3������Fe3++3NH3•H2O=Fe��OH��3��+3NH4+����
��3���������һϵ�д�������������������Һ����ת��Ϊ����������Ҫ���ˡ�ϴ�ӵ�����������Ȼ��������������������ȴ�������������������
�ʴ�Ϊ��ϴ�ӣ���ȴ��
��4�����չ���ΪFe2O3����Ԫ�ص������ٷ���Ϊ��$\frac{56��2}{56��2+16��3}$��100%=70%������ag��������Ԫ��0.7ag������10Ƭ��Ѫ������Ԫ�ص�����0.7ag����ÿƬ��Ѫ������Ԫ�ص�����0.07ag��
�ʴ�Ϊ��0.07a��
��5���ٸ�������һ�����ʵ���Ũ�ȵ���Һ����Ҫ�������У�250mL����ƿ����ƽ���������ձ�����Ͳ��ҩ�ס���ͷ�ιܣ�
�ʴ�Ϊ��250mL����ƿ��
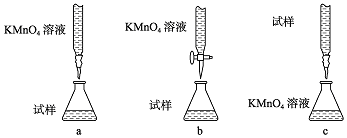
�ڸ��������ǿ�����ԣ��ܽ�����������������������ԣ�������������������ֻ����ϡ�����ữ��
�ʴ�Ϊ��b��
�۸�����ؾ���ǿ�����ԣ��ܸ�ʴ��ʽ�ζ��ܵ���Ƥ�ܣ�Ӧ����ʽ�ζ���ʢװ���ζ�ʱΪ���ڹ۲���ɫ�仯���ζ��յ���ɫ��dz�������ڹ۲죬Ӧ��������صε�����Һ�У�
�ʴ�Ϊ��b��
��6��������Ԫ���غ㣬14mg������ΪFeSO4•7H2OƬ��������������������ҪFeSO4•7H2OƬ������Ϊ��$\frac{14mg}{\frac{56}{278}}$=69.5mg��
�ʴ�Ϊ��69.5��
���� ���⿼����̽��������ɡ��������ʺ����ķ�������Ŀ�Ѷ��еȣ���ȷʵ��Ŀ�ġ�ʵ��ԭ��Ϊ���ؼ�������֪ʶ��϶ࡢ�ۺ��Խ�ǿ����ֿ�����ѧ���ķ�����������������ѧʵ��������


 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ǰ�ߴ� | B�� | ���ߴ� | C�� | ����һ�� | D�� | ��ȷ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
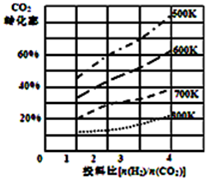 ��ȼú�����е�CO2ת��Ϊ�����ѵķ�Ӧԭ��Ϊ��2CO2��g��+6H2��g��$\stackrel{����}{?}$CH3OCH3��g��+3H2O��l��
��ȼú�����е�CO2ת��Ϊ�����ѵķ�Ӧԭ��Ϊ��2CO2��g��+6H2��g��$\stackrel{����}{?}$CH3OCH3��g��+3H2O��l��| P1 | P2 | P3 | |
| I��2.0molCO2 6.0molH2 | 0.10mol | 0.04mol | 0.02mol |
| ��1.0mol CO2 3.0molH2 | X1 | Y1 | Z1 |
| ��1.0molCH3OCH3 3.0molH2O | X2 | Y3 | Z2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | H2SO4��Һ��NaOH��Һ��� | B�� | HCl����ͨ��Ca��OH��2��Һ�� | ||
| C�� | HNO3��Һ��KOH��Һ��� | D�� | NH4HSO4��Һ��NaOH��Һ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
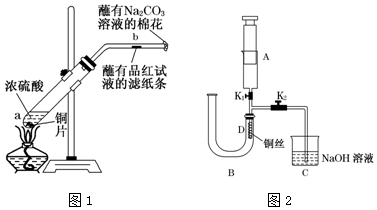
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
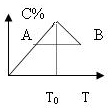 ���ڷ�Ӧ2A+B?2C�ķ�Ӧ������C�İٷֺ������¶ȵı仯��ͼ��ʾ����
���ڷ�Ӧ2A+B?2C�ķ�Ӧ������C�İٷֺ������¶ȵı仯��ͼ��ʾ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Mg2+��Ca2+��HCO3-��Cl- | B�� | Fe2+��Ca2+��Cl-��NO3- | ||
| C�� | K+��Fe2+��SO42-��Br- | D�� | Na+��K+��SO42-��AlO2- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
| A�� | ��ǿ����Һ�У�Na+��K+��CO32-��NO3- | |
| B�� | ��pOH=2����Һ�У�NH4+��Na+��SO42-��AlO2- | |
| C�� | ��pH=1����Һ�У�K+��Ag+��Mg2+��Fe2+ | |
| D�� | ˮ���������c��H+��=1��10-13 mol•L-1����Һ��K+��HCO3-��Br-��Ba2+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��������Һ | B�� | Fe��OH��3���� | ||
| C�� | ����ֲ���ͺ�ˮ��� | D�� | ��ˮ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com