
ĀĮŹĒŅ»ÖÖÖŲŅŖµÄ½šŹō£¬ŌŚÉś²ś”¢Éś»īÖŠ¾ßÓŠŠķ¶ąÖŲŅŖµÄÓĆĶ¾£¬ČēĻĀæņĶ¼ŹĒ“ÓĀĮĶĮæóÖŠÖʱøĀĮµÄ¹¤ŅÕĮ÷³Ģ£ŗ 
ŅŃÖŖ£ŗ¢ŁĀĮĶĮæóµÄÖ÷ŅŖ³É·ÖŹĒAl2O3£¬“ĖĶā»¹ŗ¬ÓŠÉŁĮæSiO2”¢Fe2O3µČŌÓÖŹ£»¢ŚČÜŅŗÖŠµÄ¹čĖįÄĘÓėĘ«ĀĮĖįÄĘ·“Ó¦£¬ÄÜÉś³É¹čĀĮĖįŃĪ³Įµķ£¬»Æѧ·½³ĢŹ½ĪŖ£ŗ 2Na2SiO3+2NaAlO2+4H2O===Na2Al2Si2O8”ż+2H2O+4NaOH
»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ČܽāĀĮĶĮæóŹ±£¬·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ____________”£
£Ø2£©ĀĖŌüAµÄÖ÷ŅŖ³É·Ö________£»¹čĀĮĖįŃĪ³ĮµķŠ“³ÉŃõ»ÆĪļµÄŠĪŹ½ŹĒ________”£
£Ø3£©ŌŚ¹¤ŅÕĮ÷³ĢµŚČż²½ÖŠ£¬Ń”ÓƶžŃõ»ÆĢ¼×÷Ėį»Æ¼Į¶ų²»Ź¹ÓĆŃĪĖįµÄŌŅņŹĒ_____________
£Ø4£©ČōøĆ¹¤³§ÓĆmkgĀĮĶĮæó¹²ÖʵĆnkg Al£Ø¼ŁÉčĆæ²½·“Ó¦½ųŠŠĶźČ«£©£¬ŌņĀĮĶĮæóÖŠAl2O3µÄÖŹĮæ·ÖŹżĪŖ______”£
£Ø5£©Čō½«ĀĮČܽā£¬ĻĀĮŠŹŌ¼ĮÖŠ×īŗĆŃ”ÓĆ_______£ØĢī±ąŗÅ£©”£
A£®ÅØĮņĖį B£® Ļ”ĮņĖį C£®Ļ”HNO3 D£®ÅØHNO3
£Ø6£©µē½āŅ±Į¶ĀĮŹ±ÓĆNa3AlF6×÷ÖśČŪ¼Į£¬Na3AlF6ŹĒÅäŗĻĪļ£¬ĘäÖŠÄŚ½ēŹĒ____£¬ÅäĪ»ŹżĪŖ______”£

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĆܱÕČŻĘ÷ÖŠ½ųŠŠµÄæÉÄę·“Ó¦£ŗaA(g)£«bB(g)cC(g)ŌŚ²»Ķ¬ĪĀ¶Č(T1ŗĶT2)¼°Ń¹Ēæ(p1ŗĶp2)ĻĀ£¬»ģŗĻĘųĢåÖŠBµÄÖŹĮæ·ÖŹżw(B)Óė·“Ó¦Ź±¼ä(t)µÄ¹ŲĻµČēĶ¼ĖłŹ¾”£ĻĀĮŠÅŠ¶ĻÕżČ·µÄŹĒ (””””)”£

A£®T1<T2£¬p1<p2£¬a£«b>c£¬Õż·“Ó¦ĪŖĪüČČ·“Ó¦
B£®T1>T2£¬p1<p2£¬a£«b<c£¬Õż·“Ó¦ĪŖĪüČČ·“Ó¦
C£®T1<T2£¬p1>p2£¬a£«b<c£¬Õż·“Ó¦ĪŖĪüČČ·“Ó¦
D£®T1>T2£¬p1>p2£¬a£«b>c£¬Õż·“Ó¦ĪŖ·ÅČČ·“Ó¦
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
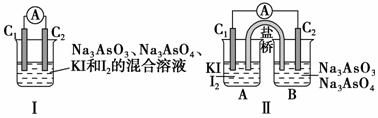
ĻĀĶ¼¢ń”¢¢ņ·Ö±šŹĒ¼×”¢ŅŅĮ½×éĶ¬Ń§½«·“Ó¦”°AsO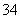 £«2I££«2H£«AsO
£«2I££«2H£«AsO £«I2£«H2O”±Éč¼Ę³ÉµÄŌµē³Ų×°ÖĆ£¬ĘäÖŠC1”¢C2¾łĪŖĢ¼°ō”£¼××éĻņĶ¼¢ńÉÕ±ÖŠÖšµĪ¼ÓČėŹŹĮæÅØŃĪĖį£»ŅŅ×éĻņĶ¼¢ņBÉÕ±ÖŠÖšµĪ¼ÓČėŹŹĮæ40%NaOHČÜŅŗ”£
£«I2£«H2O”±Éč¼Ę³ÉµÄŌµē³Ų×°ÖĆ£¬ĘäÖŠC1”¢C2¾łĪŖĢ¼°ō”£¼××éĻņĶ¼¢ńÉÕ±ÖŠÖšµĪ¼ÓČėŹŹĮæÅØŃĪĖį£»ŅŅ×éĻņĶ¼¢ņBÉÕ±ÖŠÖšµĪ¼ÓČėŹŹĮæ40%NaOHČÜŅŗ”£
ĻĀĮŠŠšŹöÖŠÕżČ·µÄŹĒ (””””)”£
A£®¼××é²Ł×÷Ź±£¬µēĮ÷¼Ę(A)ÖøÕė·¢ÉśĘ«×Ŗ
B£®¼××é²Ł×÷Ź±£¬ČÜŅŗŃÕÉ«±äĒ³
C£®ŅŅ×é²Ł×÷Ź±£¬C2×÷Õż¼«
D£®ŅŅ×é²Ł×÷Ź±£¬C1ÉĻ·¢ÉśµÄµē¼«·“Ó¦ĪŖI2£«2e£===2I£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ijæĪĶā»ī¶ÆŠ”×éÉč¼ĘĮĖČēĶ¼ĖłŹ¾×°ÖĆ£¬µ÷½Ś»¬¶Æ±ä×čĘ÷£¬ŌŚµēĮ÷Ēæ¶ČŹŹÖŠµÄĒéæöĻĀÓĆĘä½ųŠŠ»ŗĀżµē½āNaClČÜŅŗ¼°Ļą¹ŲŹµŃé(“ĖŹ±£¬“ņæŖÖ¹Ė®¼Ša£¬¹Ų±ÕÖ¹Ė®¼Šb)”£ÓÉÓŚ“ÖŠÄ£¬ŹµŃé²¢Ī““ļµ½Ō¤ĘŚÄæµÄ£¬µ«Ņ²æ“µ½ĮĖĮīČĖøߊĖµÄĻÖĻó”£Ēė°ļÖśĖūĆĒ·ÖĪö²¢»Ų“šĻĀĮŠĪŹĢā(Ķ¼ÖŠŃōĄė×Ó½»»»Ä¤Ö»ŌŹŠķŃōĄė×ÓŗĶĖ®·Ö×ÓĶعż)£ŗ
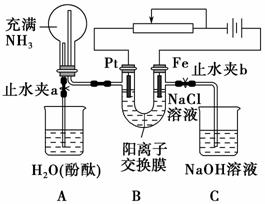
(1)Š“³öB×°ÖĆÖŠµÄµē¼«·“Ó¦Ź½£ŗ
Pt¼«_________________________________________________________£»
Fe¼«____________________________________________________________”£
(2)Š“³ö¹Ū²ģµ½µÄA×°ÖĆÖŠµÄĻÖĻó£ŗ
¢Ł_________________________________________________________£»
¢Ś__________________________________________________________£»
¢Ū___________________________________________________________”£
(3)µ±¹Ū²ģµ½A×°ÖĆÖŠµÄĻÖĻóŗó£¬ĖūĆĒ¹Ų±ÕÖ¹Ė®¼Ša£¬“ņæŖÖ¹Ė®¼Šb£¬ŌŁ¹Ū²ģC×°ÖĆ”£ČōĪŽĻÖĻó£¬ĖµĆ÷ĄķÓÉ£»ČōÓŠĻÖĻó£¬ĒėŠ“³öÓŠ¹Ų·“Ó¦·½³ĢŹ½________________________________________________________________
_______________________________________________________________ӣ
(4)ČōĻė“ļµ½µē½āNaClČÜŅŗµÄÄæµÄ£¬Ó¦ČēŗĪøĽų×°ÖĆ£¬ĒėĢį³öÄćµÄŅā¼ū________________________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŅŅĻ©“ß»ÆŃõ»Æ³ÉŅŅČ©æÉÉč¼Ę³ÉČēÓŅĶ¼ĖłŹ¾µÄČ¼ĮĻµē³Ų£¬
ÄÜŌŚÖʱøŅŅČ©µÄĶ¬Ź±»ńµĆµēÄÜ£¬Ęä×Ü·“Ó¦ĪŖ£ŗ
2CH2£½CH2 +O2”ś2CH3CHO”£ĻĀĮŠÓŠ¹ŲĖµ·ØÕżČ·µÄŹĒ
A£®ĆæÓŠ0.1mol O2·“Ó¦£¬ŌņĒØŅĘH+ 0.4mol
B£®Õż¼«·“Ó¦Ź½ĪŖ£ŗCH2£½CH2£2e- + 2OH- = CH3CHO + H2O
C£®µē×ÓŅĘ¶Æ·½Ļņ£ŗµē¼«a”śĮ×ĖįČÜŅŗ”śµē¼«b D£®øƵē³ŲĪŖæɳäµēµē³Ų
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ÉčNAĪŖ°¢·ü¼ÓµĀĀŽ³£ŹżµÄÖµ”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(””””)
A£®25 ”ę£¬pH£½13µÄNaOHČÜŅŗÖŠ£¬ŗ¬ÓŠOH£µÄŹżÄæĪŖ0.1NA
B£®1.2 g C60ŗĶŹÆÄ«µÄ»ģŗĻĪļÖŠ£¬ŗ¬ÓŠµÄĢ¼Ō×ÓŹżÄæĪŖ0.1NA
C£®2.24 L N2ŗĶNH3µÄ»ģŗĻĘųĢåÖŠ£¬ŗ¬ÓŠµÄ¹²ÓƵē×Ó¶ŌŹżÄæĪŖ0.3NA
D£®±ź×¼×“æöĻĀ£¬6.72 L NO2ÓėĖ®³ä·Ö·“Ó¦×ŖŅʵĵē×ÓŹżÄæĪŖ0.1NA
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ÉčNAĪŖ°¢·ü¼ÓµĀĀŽ³£ŹżµÄÖµ”£ĻĀĮŠŠšŹöÕżČ·µÄŹĒ(””””)
A£®±ź×¼×“æöĻĀ£¬2.24 LĖ®ÖŠĖłŗ¬µē×Ó×ÜŹżĪŖNA
B£®1 L 0.2 mol”¤L£1ĮņĖįĢśČÜŅŗÖŠŗ¬ÓŠµÄSO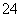 ŹżÄæĪŖ0.6NA
ŹżÄæĪŖ0.6NA
C£®1 mol±½ŗĶ±½¼×ĖįµÄ»ģŗĻĪļĶźČ«Č¼ÉÕŹ±ĻūŗÄO2µÄ·Ö×ÓŹżĪŖ7.5NA
D£®±ź×¼×“æöĻĀ£¬7.1 gĀČĘųÓė×ćĮæŹÆ»ŅČé³ä·Ö·“Ó¦×ŖŅʵē×ÓŹżĪŖ0.2NA
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠĪļÖŹÖŠŹōÓŚŗĻ³Éøß·Ö×Ó²ÄĮĻµÄŹĒ(””””)
A£®ĆŽ»Ø B£®²ĻĖæ
C£®µķ·Ū D£®ÄįĮś
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Ļņŗ¬ÓŠa mol AlCl3µÄČÜŅŗÖŠ¼ÓČėŗ¬b mol KOHµÄČÜŅŗ£¬Éś³É³ĮµķµÄĪļÖŹµÄĮææÉÄÜŹĒ(””””)
¢Ła mol””¢Śb mol””¢Ū mol””¢Ü
mol””¢Ü mol””¢Ż0 mol””¢Ž(4a£b)mol
mol””¢Ż0 mol””¢Ž(4a£b)mol
A£®¢Ł¢Ś¢Ü¢Ż¢Ž B£®¢Ł¢Ü¢Ż¢Ž C£®¢Ł¢Ś¢Ū¢Ż¢Ž D£®¢Ł¢Ū¢Ż
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com