
 ��ҵ��ˮ�Ĵ�����
��ҵ��ˮ�Ĵ�����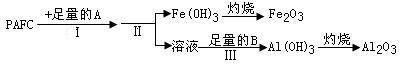
 �����⣺
�����⣺
 ��Һ�м����Լ�X��Ŀ����_________________________________��
��Һ�м����Լ�X��Ŀ����_________________________________��
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ��200mL0��5 mol��L-1��NaOH��Һ����ش��������⣺
��200mL0��5 mol��L-1��NaOH��Һ����ش��������⣺
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
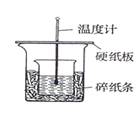
 ___
___  ����50mL 0.50mol/L NaOH��Һ��������ʵ�飬��õ��к�����ֵ�� ____________���� ��ƫ����ƫС������Ӱ�족��
����50mL 0.50mol/L NaOH��Һ��������ʵ�飬��õ��к�����ֵ�� ____________���� ��ƫ����ƫС������Ӱ�족���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| ���� | �����Լ� | ������Լ���Ŀ�� |
| B | ����NaHCO3��Һ | |
| C | | |
| E | ����NaOH��Һ | |
 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A�������Һ�м���BaCl 2��Һ�а�ɫ�����������ټ����ᣬ��������ʧ����ԭ��Һ��һ������SO42�� 2��Һ�а�ɫ�����������ټ����ᣬ��������ʧ����ԭ��Һ��һ������SO42�� |
| B�������Һ�м���NaOH��Һ�����ȣ�����ʹʪ��ĺ�ɫʯ����ֽ�������������ɣ���ԭ��Һ��һ������NH4+ |
| C�������Һ�м�����������ʹ����ʯ��ˮ����ǵ��������ɣ���ԭ��Һ��һ�����д�����CO32�� |
| D�������Һ�м�����뼸����ˮ���ټ���KSCN��Һ����Һ������죬��ԭ��Һ��һ������Fe3+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��������A13+��NH4+��Cl- | B���϶���A13+��Ba2+��HCO3- |
| C������û��K+��HCO3-��NH4+ | D���϶�û��A13+��SO32-��SO42- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com