
�Ҵ���ͨ�����۵�������ԭ�Ϸ����Ƶã����ڿ�������Դ��ͨ���Ҵ���ȡ�����������õ�Ӧ��ǰ������֪ͨ���Ҵ���ȡ��������������·�ߣ�
a.ˮ������������CH3CH2OH(g) ��H2O(g)®4H2(g) ��2CO(g) DH =��255.58 kJ��mol-1
b.���ִ�������CH3CH2OH(g)��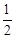 O2(g)®3H2(g)��2CO(g)
DH =��13.76kJ��mol-1
O2(g)®3H2(g)��2CO(g)
DH =��13.76kJ��mol-1
������˵���������( )
A.��ԭ�����ĵĽǶ�������a·��������м�ֵ
B.���������ĵĽǶ�����,b·�������������
C.a·����������Ҫ���ĺܶ�������������ʵ�����������岻��
D.��b·�߽���,��ϵ�������ή��
 ��һ������Ԫͬ�����ؾ�ϵ�д�
��һ������Ԫͬ�����ؾ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| A�������¶ȣ������b·�����Ҵ���ת���� |
| B�����������ĵĽǶ�������b·������������� |
| C���Ҵ���ͨ�����۵�����ԭ�Ϸ����Ƶã����ڿ�������Դ |
| D����a��b֪��2H2��g��+O2��g��=2H2O��g����H=-483.6kJ?mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����ʡ�ʹ�һ�и�������μ�⻯ѧ�Ծ����������� ���ͣ���ѡ��
��֪ͨ���Ҵ���ȡ��������������·�ߣ�
a��CH3CH2OH(g)+H2O(g)==4H2(g)+2CO(g) ��H= +255.6kJ��mol��1
b��CH3CH2OH(g)+1/2O2(g)==3H2(g)+2CO(g) ��H= +13.8kJ��mol��1
������˵������ȷ����
| A�������¶ȣ������b·�����Ҵ���ת���� |
| B�����������ĵĽǶ�������b·������������� |
| C���Ҵ���ͨ�����۵�����ԭ�Ϸ����Ƶã����ڿ�������Դ |
| D����a��b֪��2H2(g)+O2(g)��2H2O(g)��H=��483.6kJ��mol��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ������ʡ����12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��ѧ���������ճ�������������Ҫ��Ӧ�á�����˵������ȷ����
A���̬���ʲ������ľ�һ��ʩ��
B���Ҵ���ͨ�����۵�����ԭ�Ϸ����Ƶã����ڿ�������Դ
C��������ά���ϳ���ά��������ά���ά�����л��߷��ӻ�����
D��MgO���۵�ܸߣ��������������²��ϣ�����ҵ�ϲ���MgO��þ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ��������μ�⻯ѧ�Ծ��������棩 ���ͣ�ѡ����
��֪ͨ���Ҵ���ȡ��������������·�ߣ�
a��CH3CH2OH(g)+H2O(g)==4H2(g)+2CO(g) ��H= +255.6kJ��mol��1
b��CH3CH2OH(g)+1/2O2(g)==3H2(g)+2CO(g) ��H= +13.8kJ��mol��1
������˵������ȷ����
A�������¶ȣ������b·�����Ҵ���ת����
B�����������ĵĽǶ�������b·�������������
C���Ҵ���ͨ�����۵�����ԭ�Ϸ����Ƶã����ڿ�������Դ
D����a��b֪��2H2(g)+O2(g)��2H2O(g)��H=��483.6kJ��mol��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��������ĩ�� ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com