
| 0.8m(V2-V1) |
| W |
| 0.8m(V2-V1) |
| W |
| 250 |
| 25 |
| 80m(V2-V1)��10-2g |
| Wg |
| 0.8m(V2-V1) |
| W |
| 0.8m(V2-V1) |
| W |


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
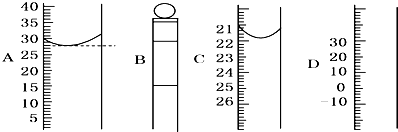
| 1 | 10 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A.��250 mL������ƿ�ж�������250 mL�ռ���Һ
B.����Һ����ȡ25 mL�ռ���Һ����ƿ�У������뼸�μ�����ָʾ��
C.��������ƽ��ȷ��ȡ�ռ���ƷW g�����ձ���������ˮ�ܽ�
D.�����ʵ���Ũ��Ϊc mol��L-1�ı�������Һװ������ϴ������ʽ�ζ����У�����Һ�沢������ʼ����ΪV1 mL
E.����ƿ�µ�һ�Ű�ֽ���ζ�����ɫΪֹ�����¶���ΪV2 mL
�ʹ�����������⣺
(1)��ȷ����������(�ñ����ĸ���)_________��_________��_________��D��_________��
(2)�۲�ζ�����Һ��ĸ߶�ʱӦע�⣺____________________________________��
(3)E����IJ���������ƿ�µ�һ�Ű�ֽ��������___________________________��
(4)D����IJ�����Һ��Ӧ������_________�����첿��Ӧ��__________________��
(5)ijѧ��ʵ��ʱ����ƿ�ô����ռ���Һ��ϴ����ⶨ���ռ���Һ��Ũ��ƫ����ƫ��_________��ԭ����____________________________________��
(6)���ռ���Ʒ���ȵļ��㹫ʽ��__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���Ĵ�ʡ�Ű���ѧ�߶���ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ������
����16�֣���1������ͭ������ܽ������ܽ���̽�����ʵ���ҳ�����ˮ�����Լӿ��ܽ����ʣ���������������ǣ����Ҫ˵��ԭ��______________________����β�������ˮ���Ƴ�����Ľ�Ũ��CuSO4��Һ____________________��
��2��ϡNa2S��Һ��һ�ֳ�������ζ������AlCl3��Һ��������ζ�Ӿ磬�����ӷ���ʽ��ʾ��ζ�Ӿ�����������Ļ�ѧ��Ӧ______________________________________________
��ij�ռ���Ʒ�к����������������õĿ��������ʣ�Ϊ�˲ⶨ�䴿�ȣ��������µζ�������
| A������Һת����250 mL����ƿ�У���ˮ���̶��ߣ� |
| B������Һ��(���ʽ�ζ���)��ȡ25.00 mL�ռ���Һ����ƿ�в��Ӽ��μ�����ָʾ���� |
| C������ƽ��ȷ��ȡ�ռ���Ʒw g�����ձ��м�����ˮ�ܽ⣻ |
| D�������ʵ���Ũ��Ϊm mol?L��1�ı�H2SO4��Һװ����ʽ�ζ��ܣ�����Һ�棬���¿�ʼ�̶���ΪV1 mL�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com