
���ִ���ҵ�����Ȼ���Ϊԭ���Ʊ�������ֹ����������£�
��֪NaHCO3�ڵ������ܽ�Ƚ�С��
��Ӧ��NaCl+CO2+NH3+H2O NaHCO3��+NH4Cl������ĸҺ�����ַ�����
NaHCO3��+NH4Cl������ĸҺ�����ַ�����
��1����ĸҺ�м���ʯ���飬�ɽ�����________ѭ�����á�
��2����ĸҺ��ͨ��NH3������ϸС��ʳ�ο��������£��ɵõ�NH4Cl���塣��д��ͨ��NH3���ܽ�Ƚ�С����ʽ̼����ת��Ϊ�ܽ�Ƚϴ��̼���ε����ӷ���ʽ ___________��
��ij��ѧС��ģ�⡰�����Ƽ������NaCl��NH3��CO2��ˮ��Ϊԭ���Լ���ͼ��ʾװ����ȡNaHCO3��Ȼ���ٽ�NaHCO3�Ƴ�Na2CO3��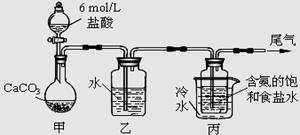
��1��װ�ñ�����ˮ�������� ��
��2����װ�ñ��в�����NaHCO3��ȡNa2CO3ʱ����Ҫ���е�ʵ�������_______��ϴ�ӡ����ա�NaHCO3ת��ΪNa2CO3�Ļ�ѧ����ʽΪ ��
��3�����ڣ�2�������յ�ʱ��϶̣�NaHCO3���ֽⲻ��ȫ����С���һ�ݼ�����t1 min��NaHCO3 ��Ʒ����ɽ���������̽����
ȡ������t1 min��NaHCO3��Ʒ29��6 g��ȫ����ˮ�Ƴ���Һ��Ȼ�������Һ�л����صμ�ϡ���ᣬ�����Ͻ��衣��������ļ��룬��Һ���й����ӵ����ʵ����ı仯��ͼ��ʾ��
������a��Ӧ����Һ�е�������___________�������ӷ�����ͬ��������c��Ӧ����Һ�е�������___________������Ʒ��NaHCO3��Na2CO3�����ʵ���֮���� �� 21
��4����ȡ21��0 g NaHCO3���壬������t2 rnin��ʣ����������Ϊl4��8 g������Ѵ�ʣ�����ȫ�����뵽200 mL 2 mol?L��1�������У����ַ�Ӧ����Һ��H+ �����ʵ���Ũ��Ϊ____________������Һ����仯���Բ��ƣ�
��1��NH3
��2��HCO3�C+NH3=NH4++CO32�C
��1����ȴ��ʹ̼�����ƾ�������
��2������ 2NaHCO3 Na2CO3+H2O+CO2��
Na2CO3+H2O+CO2��
��3��Na+ HCO3- 1:2
��4��0��75 mol/L
���������������1��������NaHCO3������ĸҺNH4Cl�м������ʯ���飬�����ķ�Ӧ��Ca��OH��2+2NH4Cl=2NH3��+2H2O+CaCl2�����ղ���Ϊ�Ȼ��ơ����������а����������ã��ʴ�Ϊ��NH3��
��2��ͨ��NH3���ܽ�Ƚ�С����ʽ̼����ת��Ϊ�ܽ�Ƚϴ��̼���ε����ӷ���ʽHCO3�C+NH3=NH4++CO32�C��
��1��װ�ñ�����ˮ�������ǽ��£�ʹ̼�����ƾ���������
��2����װ�ñ��в�����NaHCO3�����ķ�ӦΪ��NH3+CO2+H2O+NaCl=NaHCO3��+NH4Cl����ȡNa2CO3ʱ��Ҫ���˵õ����壬ϴ�Ӻ�������յõ�̼���ƣ�2NaHCO3 Na2CO3+H2O+CO2��
Na2CO3+H2O+CO2��
��3����Һ���й����ӵ����ʵ����ı仯Ϊ��������ʼ�ղ��䣬̼������Ӽ�С��̼���������Ũ������̼�������ȫ��ת��Ϊ̼��������ӣ��ٵ��������̼��������ӷ�Ӧ���ɶ�����̼��̼��������Ӽ�С������c���߱�ʾ����̼���������Ũ�ȱ仯��̼�������Ũ��0��2mol/L��̼���������Ũ��Ϊ0��1mol/L����Ʒ��NaHCO3��Na2CO3�����ʵ���֮����1��2�� �ʴ�Ϊ��Na+ ��HCO3-�� 1��2��
��4����ȡ21g NaHCO3�������ʵ���=21g/84g/mol=0��25mol��
������t1min��ʣ����������Ϊ14��8g�����ݻ�ѧ����ʽ���ڵ������仯���㣺
2NaHCO3=Na2CO3+CO2��+H2O ��m
2 1 62
0��2mol 0��1mol 21g-14��8g
��Ӧ��NaHCO3���ʵ���=0��25mol-0��2mol=0��05mol��NaHCO3+HCl=NaCl+H2O+CO2���������Ȼ������ʵ���0��05mol��
Na2CO3���ʵ���=0��1mol��Na2CO3+2HCl=2NaCl+H2O+CO2���������Ȼ������ʵ���0��2mol��
ʣ���Ȼ������ʵ���=0��200L��2mol/L-0��05mol-0��2mol=0��15mol��ʣ����Һ��c��H+��=
0��15mol/0��2L=0��75mol/L�ʴ�Ϊ��0��75mol/L
���㣺��ҵ�ƴ����ԭ�����������������е����ʱ仯�������ɷֵķ����жϺ�ͼ������������Ӧ�á�


 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij����С���һЩ�������ʺͻ���������ʽ���̽����
(1)�±�Ϊ�������Ȼ�ͭ��Һ��Ӧ��ʵ�鱨���һ���֣�
| ʵ�鲽�� | ʵ������ |
| ����ĥ������Ƭ(����)����һ��Ũ�ȵ�CuCl2��Һ�� | �������ݣ��������ɵĺ�ɫ���壬��Һ��Ϊ��ɫ |
| ��Ӧ������������Һ���� | |
| ��ɫ����������ˮϴ�Ӻ����ڳ�ʪ������ | һ��ʱ�������ɺ�ɫ��Ϊ��ɫ[������Ҫ�ɷ�ΪCu2(OH)2CO3] |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ɫ������A������ɫ���ߵ�ѹ���������ص㣬�����������˵绯ѧ��ĸ߶����ӡ��ڳ��º���������£�������A�����ȶ��Ĵ��ڣ�������ˮ��Һ�в��ȶ���һ��ʱ���ת��Ϊ���ɫ������ͬʱ����һ�����嵥�ʡ�ij��ȤС���ͬѧ�Ի�����A������ɷ�����ȷ��A�н�����O��K��Fe����Ԫ�ء�ȡ3.96g������A�ķ�ĩ����ˮ���μ�������ϡ���ᣬ��Ӧ�����Һ�м��뺬��0.08mol KOH����Һ��ǡ����ȫ��Ӧ�����ˣ���ϴ�Ӻ�ij���������գ��õ�����ɫ�����ĩ1.60g����������Һ��һ�������������ɵõ�һ�ִ����IJ����ᾧˮ����10.44g��
��1��������A�Ļ�ѧʽΪ ��������A��H2O��Ӧ�����ӷ���ʽΪ ��
��2��������A������Ϊһ�֡���ɫ��Ч��ܡ�ˮ��������ԭ���� ��
��3��������A���Ʊ�����ͨ������������д����KOH�����������ô�������������������Ʊ�A�Ļ�ѧ����ʽ ��
��4��Ŀǰ��������Ի�����A���ȶ��Խ����˴�����̽������ȡ����һ���Ľ�չ�������������п����������Aˮ��Һ�ȶ��Ե���
| A���������� | B��KOH | C������ | D��Fe(NO3)3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������Ʊ��治�����ױ��ʣ�����Na2CO3��
��1��ij����������Ʒ�Ѿ����ֱ��ʣ�����ѡ��һ����Һ ��֤�����������Ѿ����ʡ�
��2��ij����С��Ϊ�˴��Բⶨ�������Ƶ��������������dz�ȡag��Ʒ�����������ͼװ�����ⶨ�������Ƶ�����������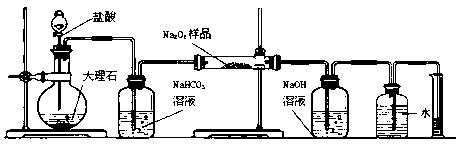
| A�� | B�� | C�� | D����E����F�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��.ʵ����������500mL 0.2mol/L NaCl��Һ
��1����ʵ���õ�����������ҩ�ס��ձ�����Ͳ��������ƽ(���롢����)������������ͷ�ιܣ���ȱ�ٵIJ��������� ��
��2������ʱ�����ȡNaCl�������� g
��3�������ƹ����У�������������ȷ������£����в����ᵼ�������Ƶ���ҺŨ��ƫ�ߵ��� ����ѡ����ţ�
��û��ϴ���ձ��Ͳ�����
�ڼ�����ˮʱ�����������˿̶ȣ�ȡ������ˮʹҺ��ǡ�õ��̶���
������ƿ�����������������ˮ
�ܶ���ʱ���ӱ���
�ݶ���ʱ���ӱ���
��.��10�֣�ij����Ϊ���������һ��ѧ��ȤС����ʵ������Ըô�������Ԫ�صļ�̬����̽���������й����ϵ�֪Fe2+�ܱ�����KMnO4��Һ������ʹ����KMnO4��Һ��ɫ��HNO3����ǿ�����ԡ�����ɶ���Ԫ�ؼ�̬��̽����
��1������������衣
����1����������Ԫ��Ϊ+3�ۣ�
����2�� ��
����3����������Ԫ�ؼ���+3������+2��
��2�����ʵ�鷽����
��3�����ݣ�2����ʵ�鷽������ʵ�飺
����1��ȡһ������ϡ�������Թ��У�������������ӣ�Ŀ���� ��
����2��ȡ������Ʒ���Թ��У����봦���������Ტ���ȣ��õ�A��Һ��
����3��ȡA��Һ���Թܣ��μ�KSCN��Һ������������Ѫ��ɫ������� ������������Ѫ��ɫ�������1��3������
����4��Ϊ�˽�һ��ȷ������3���Ǽ���1���Ǽ���3��������ȡA��Һ���Թܣ�����KMnO4��Һ���������� ���������1��������֮�������3������
��4����˼
ijͬѧ������2�е�ϡ����ij�ϡ����õ���A��Һ����A��Һ�еμ�KSCN��Һ�����Ѫ��ɫ���ɴ˵ó��ô�������Ԫ��Ϊ+3�۵Ľ��ۡ����жϸý����Ƿ����
���������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ͭ���仯�������������������й㷺��Ӧ�á�
��ͭ�ɲ������·����Ʊ���
����һ������ͭ��Cu2S + O2 2Cu + SO2
2Cu + SO2
��������ʪ����ͭ�� CuSO4 + Fe FeSO4 + Cu
�������ַ����У�����Ϊ���ַ������� ��ԭ���� ��д��1��ԭ�ɣ����ֱ��û���ͭ��ʪ����ͭ����640gCu����ת�Ƶĵ�����֮��Ϊ ��
��ӡˢ��·����ʹ�õ�ͭ��Ҫ�������á�
��FeCl3��Һ����ӡˢ��·���Ʊ�CuCl2��2H2O���壬ʵ����ģ����չ������£�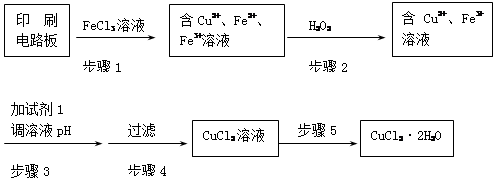
�ٲ���2�з�Ӧ�����ӷ���ʽ�� ��
�ڲ���5������Ũ��CuCl2��Һʱ��Ҫ�μ�Ũ���ᣬĿ����________________ (�û�ѧ����ʽ����ϼ�Ҫ������˵��)���پ���ȴ���ᾧ�����ˣ��õ�CuCl2��2H2O���塣
�۲���4��ϴ�ӳ����IJ���Ϊ
�ܲ���3�п�ѡ�õġ��Լ�1���� (д��һ�ּ���)��Ŀ����ʹ��Һ��pH���ߵ�4����ʱFe3+Ũ��Ϊ [Ksp(Fe(OH)3)=4��10-38]��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
þ������������Ҫ�Ľ������ڹ�ҵ��������;�㷺��
��1��þ��ϡ���ᷴӦ�����ӷ���ʽΪ ��
��2�������������������ȷ�Ӧ�Ļ�ѧ����ʽΪ ��
��3���ڳ�ʪ�Ŀ��������������һ��ˮĤ�����������绯ѧ��ʴ�����������ĵ缫��ӦʽΪ ��
��4�����ڿ����о��кܺõĿ���ʴ���ܣ�����Ʒ���ճ������б��㷺Ӧ�á��ں����ϣ�ͨ�������Ͻ�Al��Zn��Cd��������������ʩ����ԭ����ͼ18��ʾ��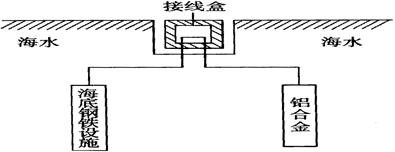
���и��������ĵ缫��ӦΪ ��
��ʵ��Ӧ���У������Ͻ����ѡ�ô������������ܺܺõ��������ã���ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijʵ��С����̽��Na2CO3��NaHCO3�����ʣ�����ʵ������ʢ�����ֹ�����Լ�ƿ��ʧ�˱�ǩ�����ǣ������ȶԹ���A��B���м�����ͨ��ʵ���������̽����
��1���ֱ���ȹ���A��B�����ֹ���A���Ȳ�����������ʹ����ʯ��ˮ����ǡ�A���ȷֽ�Ļ�ѧ����ʽΪ ��
��2����ȡ���ֹ����2 g���ֱ��������С�ձ��У��ٸ���10 mL ����ˮ���������¶ȱ仯�����������ܽ⣬�ָ������£���������Һ�и�����2�η�̪��Һ��
�ٷ���Na2CO3������ȫ�ܽ⣬��NaHCO3������ʣ�࣬�ɴ˵ó����� ��
��ͬѧ�������ձ��л��۲쵽�����������У�ʢ��Na2CO3���ձ��г��ֵ�������
������ĸ��ţ���
a����Һ�¶��½� b����Һ�¶�����
c�������̪�����ɫ d�������̪��ʺ�ɫ
��3����ͼ��ʾ�������������õ�װ��I��II�зֱ����ҩƷ���������ڵĹ���ͬʱ�����Թ��С�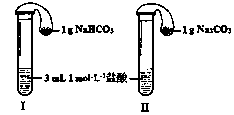
�����Թ��о��������壬 ���I����II�����ķ�Ӧ�̶ȸ�Ϊ���ҡ�
�ڷ�Ӧ����������������ͣ��ָ������£�����˵����ȷ���� ��
a��װ��I����������ϴ� b��װ��II����������ϴ�
c������������������������� d�����������������ݹ������
��4��ͬѧ�ǽ����ֹ���ֱ����Ƴ�0��5 mol��L-1����Һ��������·������Է�Ӧ��������Ԥ�⣺
| ʵ�鷽�� | Ԥ������ | Ԥ������ |
| ����1����2 mL Na2CO3��Һ�еμ�1 mL 0��5 mol��L-1CaCl2��Һ | �а�ɫ���� | Na2CO3��Һ�е�CO32-Ũ�Ƚϴ�����CaCl2������ӦCa2+ + CO32- = CaCO3���� |
| ����2����2 mL NaHCO3��Һ�еμ�1 mL 0��5 mol��L-1CaCl2��Һ | �ް�ɫ���� | NaHCO3��Һ�е�CO32-Ũ�Ⱥ�С��������CaCl2��Ӧ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ؾ�ʯ����Ҫ�ɷ�Ϊ���ᱵ����Ϊ����Fe2O3��MnO��̿�ʵ����ʶ��������ɫ����ҵ�Ͻ��ؾ�ʯ�����������ᡢ�����ڷ�Ӧ���л�ϼ��ȣ�����Ư�ס������پ�ˮϴ��һϵ�й����Ƶð�ɫ���ؾ�ʯ���ϣ��㷺����ֽ�š�����ȵ���������֪MnO�Ǽ��������Al�ۿ��Խ���ɫ��Fe3+ת��Ϊdzɫ��Fe2+��
�ؾ�ʯ���ϵ�������������Ϊ��
��1���ڸ������У�Ϊ�ӿ조Ư�ס��ٶȣ���ȡ�Ĵ�ʩ�� ��
��
��2���������ˡ�Ư�ס������á���д������ֱ��������ʱ�Ļ�ѧ����ʽ��
�� ��
��3������Ư��ˮϴ���˺�����������Fe2+���ӵķ�����
�� ��
��4������Ư��ǰ�����յ���ҪĿ���� �������պ�Ĺ���ĥ��ϸ�ۣ�ʹ�õ��Ǽ��и��������ĥ����˵��������кܸߵ� ��
��5����ҵ������Ϊ�˳��������Դ������Һ���������õ�����ԭ��Fe2O3�����������ǣ�
����Һ��ͨ�����Cl2����Ŀ���� ��
���ټ�������NaOH��������Һ��pH ����Ŀ���� ��������Һ��pH��ΧΪ ��
�й����ӿ�ʼ��������ȫ����ʱ��pH���£�
| ���� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
| Fe2+ | 7.6 | 9.7 |
| Fe3+ | 2.7 | 3.7 |
| Al3+ | 3.8 | 4.7 |
| Mn2+ | 8.3 | 9.8 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com