
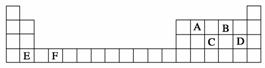
A”¢B”¢C”¢D”¢E”¢FŹĒŌŖĖŲÖÜĘŚ±ķĒ°ĖÄÖÜĘŚµÄŌŖĖŲ£¬ĖüĆĒŌŚŌŖĖŲÖÜĘŚ±ķÖŠµÄĪ»ÖĆČēĻĀĶ¼ĖłŹ¾£ŗ
 ””””
””””
(1)Š“³öFµÄ»łĢ¬Ō×ÓŗĖĶāµē×ÓÅŲ¼Ź½£ŗ”””””””””””””””””””””””””””£EµÄ¾§°ū½į¹¹ČēÉĻĶ¼ĖłŹ¾£¬øĆ¾§°ūÖŠŗ¬ÓŠ½šŹōŌ×ӵďżÄæĪŖ________”£
(2)CŌŖĖŲµÄµŚŅ»µēĄėÄܱČĶ¬ÖÜĘŚĻąĮŚµÄĮ½ÖÖŌŖĖŲµÄµŚŅ»µēĄėÄܶ¼øßµÄŌŅņŹĒ____________________________”£
(3)ÓÉA”¢B”¢DČżÖÖŌŖĖŲŠĪ³ÉµÄŅ»ÖÖĪļÖŹĖ×³Ę¹āĘų£¬·Ö×ÓÖŠAŌ×Ó²ÉČ”sp2ŌӻƳɼü”£¹āĘų·Ö×ӵĽį¹¹Ź½ŹĒ________£¬ĘäÖŠĢ¼ŃõŌ×ÓÖ®¼äµÄ¹²¼Ū¼üŹĒ________(ĢīŠņŗÅ)£ŗ
a£®2øö¦Ņ¼ü
b£®2øö¦Š¼ü
c£®1øö¦Ņ¼ü”¢1øö¦Š¼ü
(4)
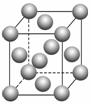
EB¾§°ū
EB¾§°ūČēÓŅĶ¼ĖłŹ¾£¬EB¾§ĢåÖŠE2£«µÄÅäĪ»ŹżĪŖ____________£¬EB¾§ĢåŗĶNaCl¾§ĢåÖŠĄė×ÓÅÅĮŠ·½Ź½ĻąĶ¬£¬Ę侧øńÄÜ·Ö±šĪŖ£ŗEB£3 401 kJ”¤mol£1”¢NaCl£786 kJ”¤mol£1”£µ¼ÖĀĮ½Õß¾§øńÄܲīŅģµÄÖ÷ŅŖŌŅņŹĒ____________________________________________”£
(5)ŌŖĖŲAŠĪ³ÉµÄµ„ÖŹµÄ¾§ĢåĄąŠĶæÉŅŌŹĒŌ×Ó¾§Ģ壬Čē________(ĢīŠ“ĪļÖŹĆū³Ę)£¬Ņ²æÉŅŌŹĒ________£¬ČēC60”£
 ŠĀĢāŠĶČ«³Ģ¼ģ²āĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø
ŠĀĢāŠĶČ«³Ģ¼ģ²āĘŚÄ©³å“Ģ100·ÖĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
NH4Al(SO4)2·12H2OµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ453”£ÓūÅäÖĘ100 mL pHĪŖ2”¢ÅضČŌ¼ĪŖ0.1 mol·L£1µÄNH4Al(SO4)2ČÜŅŗ£¬ÅäÖĘ¹ż³ĢĪŖ
¢ŁÓĆĶŠÅĢĢģĘ½³ĘĮæNH4Al(SO4)2·12H2O¹ĢĢå________________________________________________________________________g£»
¢Ś½«ÉĻŹö¹ĢĢåÖĆÓŚÉÕ±ÖŠ£¬________________________________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠĖµ·ØÖŠ£¬ÕżČ·µÄŹĒ(””””)
A£®Ēā¼üŹĒŅ»ÖÖ»Æѧ¼ü
B£®Ēā¼üŹ¹ĪļÖŹ¾ßÓŠ½ĻøßµÄČŪ”¢·Šµć
C£®ÄÜÓėĖ®·Ö×ÓŠĪ³ÉĒā¼üµÄĪļÖŹŅ×ČÜÓŚĖ®
D£®Ė®½į³É±łĢå»żÅņÕĶÓėĒā¼üĪŽ¹Ų
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ČēĶ¼ŹĒijĪŽ»ś»ÆŗĻĪļµÄ¶ž¾Ū·Ö×Ó£¬øĆ·Ö×ÓÖŠA”¢BĮ½ÖÖŌŖĖŲ¶¼ŹĒµŚ3ÖÜĘŚµÄŌŖĖŲ£¬·Ö×ÓÖŠĖłÓŠŌ×ÓµÄ×īĶā²ćµē×ÓŹż¶¼“ļµ½8øöµē×ÓµÄĪČ¶Ø½į¹¹”£ĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ(””””)

A£®øĆ»ÆŗĻĪļµÄ»ÆѧŹ½ŹĒAl2Cl6 B£®øĆ»ÆŗĻĪļŹĒĄė×Ó»ÆŗĻĪļ£¬ŌŚČŪȌדĢ¬ĻĀÄܵ¼µē
C£®øĆ»ÆŗĻĪļŌŚ¹ĢĢ¬Ź±ĖłŠĪ³ÉµÄ¾§ĢåŹĒ·Ö×Ó¾§Ģå D£®øĆ»ÆŗĻĪļÖŠ²»“ęŌŚĄė×Ó¼ü£¬Ņ²²»ŗ¬ÓŠ·Ē¼«ŠŌ¹²¼Ū¼ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
±µŌŚŃõĘųÖŠČ¼ÉÕŹ±µĆµ½Ņ»ÖÖ±µµÄŃõ»ÆĪļ¾§Ģ壬¾§Ģå½į¹¹ČēĶ¼ĖłŹ¾£¬ÓŠ¹ŲĖµ·ØÕżČ·µÄŹĒ(””””)
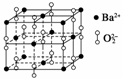
A£®øĆ¾§ĢåŹōÓŚ·Ö×Ó¾§Ģå B£®¾§ĢåµÄ»ÆѧŹ½ĪŖBa2O2

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(””””)
A£®ŅņĪŖSO2¾ßÓŠĘư׊Ō£¬ĖłŅŌĖüÄÜŹ¹Ę·ŗģČÜŅŗ”¢äåĖ®”¢ĖįŠŌKMnO4ČÜŅŗ”¢ŹÆČļČÜŅŗĶŹÉ«
B£®ÄÜŹ¹Ę·ŗģČÜŅŗĶŹÉ«µÄĪļÖŹ²»Ņ»¶ØŹĒSO2
C£®SO2”¢ĘÆ°×·Ū”¢»īŠŌĢ攢Na2O2¶¼ÄÜŹ¹ŗģÄ«Ė®ĶŹÉ«£¬µ«ŌĄķ²»¶¼ĻąĶ¬
D£®µČĪļÖŹµÄĮæµÄSO2ŗĶCl2»ģŗĻŗóĶØČė×°ÓŠŹŖČóµÄÓŠÉ«²¼ĢõµÄ¼ÆĘųĘæÖŠ£¬Ęư׊§¹ūøüŗĆ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ČēĶ¼ĖłŹ¾ŹĒŅ»ÖÖ×ŪŗĻ“¦ĄķSO2·ĻĘųµÄ¹¤ŅÕĮ÷³Ģ£¬ČōĆæ²½¶¼ĶźČ«·“Ó¦”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(””””)
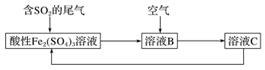
A£®ČÜŅŗBÖŠ·¢ÉśµÄ·“Ó¦ĪŖ2SO2£«O2===2SO3
B£®æÉÓĆĖįŠŌøßĆĢĖį¼ŲČÜŅŗ¼ģŃéČÜŅŗCÖŠŹĒ·ńŗ¬ÓŠFe2£«
C£®ÓÉŅŌÉĻĮ÷³ĢæÉĶĘÖŖŃõ»ÆŠŌ£ŗFe3£«>O2>SO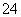
D£®“Ė¹¤ŅÕµÄÓŵćÖ®Ņ»ŹĒĪļÖŹÄÜŃ»·ĄūÓĆ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Įņ“śĮņĖįÄĘŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤²śĘ·”£Ä³ŠĖȤŠ”×éÄāÖʱøĮņ“śĮņĖįÄĘ¾§Ģå(Na2S2O3·5H2O)”£
¢ń.[²éŌÄ׏ĮĻ]
(1)Na2S2O3·5H2OŹĒĪŽÉ«ĶøĆ÷¾§Ģ壬Ņ×ČÜÓŚĖ®£¬ĘäĻ”ČÜŅŗÓėBaCl2ČÜŅŗ»ģŗĻĪŽ³ĮµķÉś³É”£
(2)ĻņNa2CO3ŗĶNa2S»ģŗĻČÜŅŗÖŠĶØČėSO2æÉÖʵĆNa2S2O3£¬ĖłµĆ²śĘ·³£ŗ¬ÓŠÉŁĮæNa2SO3ŗĶNa2SO4”£
(3)Na2SO3Ņ×±»Ńõ»Æ£»BaSO3ÄŃČÜÓŚĖ®£¬æÉČÜÓŚĻ”HCl”£
¢ņ.[Öʱø²śĘ·]
ŹµŃé×°ÖĆČēĶ¼ĖłŹ¾(Ź”ĀŌ¼Š³Ö×°ÖĆ)£ŗ
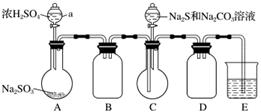
ŹµŃé²½Öč£ŗ
(1)¼ģ²é×°ÖĆĘųĆÜŠŌ£¬°“Ķ¼Ź¾¼ÓČėŹŌ¼Į”£
ŅĒĘ÷aµÄĆū³ĘŹĒ________£»EÖŠµÄŹŌ¼ĮŹĒ________(Ń”ĢīĻĀĮŠ×ÖÄø±ąŗÅ)”£
A£®Ļ”H2SO4
B£®NaOHČÜŅŗ
C£®±„ŗĶNaHSO3ČÜŅŗ
(2)ĻČĻņCÖŠÉÕĘæ¼ÓČėNa2SŗĶNa2CO3»ģŗĻČÜŅŗ£¬ŌŁĻņAÖŠÉÕĘæµĪ¼ÓÅØH2SO4”£
(3)“żNa2SŗĶNa2CO3ĶźČ«ĻūŗÄŗ󣬽įŹų·“Ó¦”£¹żĀĖCÖŠ»ģŗĻĪļ£¬ĀĖŅŗ¾__________(ĢīŠ“²Ł×÷Ćū³Ę)”¢½į¾§”¢¹żĀĖ”¢Ļ“µÓ”¢øÉŌļ£¬µĆµ½²śĘ·”£
¢ó.[Ģ½¾æÓė·“Ė¼]
(1)ĪŖŃéÖ¤²śĘ·ÖŠŗ¬ÓŠNa2SO3ŗĶNa2SO4£¬øĆŠ”×éÉč¼ĘĮĖŅŌĻĀŹµŃé·½°ø£¬Ēė½«·½°ø²¹³äĶźÕū”£
(ĖłŠčŹŌ¼Į“ÓĻ”HNO3”¢Ļ”H2SO4”¢Ļ”HCl”¢ÕōĮóĖ®ÖŠŃ”Ōń)
Č”ŹŹĮæ²śĘ·Åä³ÉĻ”ČÜŅŗ£¬µĪ¼Ó×ćĮæBaCl2ČÜŅŗ£¬ÓŠ°×É«³ĮµķÉś³É£¬________________£¬Čō³ĮµķĪ“ĶźČ«Čܽā£¬²¢ÓŠ“Ģ¼¤ŠŌĘųĪ¶µÄĘųĢå²śÉś£¬ŌņæÉČ·¶Ø²śĘ·ÖŠŗ¬ÓŠNa2SO3ŗĶNa2SO4”£
(2)ĪŖ¼õɣװÖĆCÖŠÉś³ÉNa2SO4µÄĮ棬ŌŚ²»øıäŌӊװÖƵĻł“”ÉĻ¶ŌŹµŃé²½Öč(2)½ųŠŠĮĖøĽų£¬øĽųŗóµÄ²Ł×÷ŹĒ
________________________________________________________________________ӣ
(3)Na2S2O3·5H2OµÄČܽā¶ČĖęĪĀ¶ČÉżøßĻŌÖųŌö“ó£¬ĖłµĆ²śĘ·Ķعż________________·½·ØĢį“攣
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
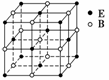
Ō×ÓŹżĻąĶ¬”¢µē×Ó×ÜŹżĻąĶ¬µÄĮ£×Ó£¬»„³ĘĪŖµČµē×ÓĢ唣ŚŌŖĖŲæÉŠĪ³ÉČżøöµČµē×ÓĢåŅõĄė×Ó£ŗBO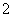 ”¢BC
”¢BC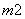 ŗĶBN
ŗĶBN £¬Ōņm”¢nÖµĪŖ(””””)
£¬Ōņm”¢nÖµĪŖ(””””)
A£®5”¢3 B£®2”¢4 C£®3”¢1 D£®1”¢2
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com