
�����Ǹ������Դ���⣬���ú�ˮ����ȡ�������Ʒ��
��1���ȼҵ���Ե��ʳ��ˮΪ������ѧ��ҵ��д�����ʳ��ˮ�Ļ�ѧ����ʽ_________
__________________________________________________________.
��2���Ӻ�ˮ����ȡ�峣�ô���������������(Cl2)����ˮ���廯�ƣ�NaBr���е����û����������ÿ���������(Br2)������ʾ��ͼ���£�

��д��������NaBr���û����嵥�ʵĻ�ѧ����ʽ��____________________;�÷�Ӧ��pH=3�����������½��У�����________�ⶨ��ӦҺ�����ȡ�
�ڴ�������ʹ������������—���������������������ͼ������ͨ��SO2��ˮ��Ŀ���ǡ�_____________________________________________________.���û�ѧ����ʽ��ʾ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����£����и���������ָ����Һ�п��ܴ����������(�� ��)
A���ڼ�����Һ�У�K+��Cl-��Na+��CO32-
B����c(H+)/ c(OH-)=1��10-12����Һ�У�Ca2+��Cl-��K+��NH4+
C����������Һ�У�Cu2+��Fe3+��I-��SO42- ks5u
D�������۷�Ӧ�ų���������ɫ��Һ�У�K+��NO3-��Cl-��HCO3-
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����������ѵ���Ҫ������Ѱ�(�����Ķ�������)��һ�������ʸߡ���ɫ�����ڸ���ǿ����ѧ�����ȶ��İ�ɫ���ϡ�����������Ҫ�ɷ�FeTiO3����Fe2O3��SiO2�����ʣ���ȡ�������ѣ��������ᷨ�����������£�


��1����������Ҫ�ɷ���Ũ���ᷴӦ����Ҫ������TiOSO4��FeSO4���÷�Ӧ�Ļ�ѧ����ʽΪ
��
��2��ȡ������ȡҺ���μ�KSCN��Һ���Ѫ��ɫ��˵����Һ�д��� ���ӣ���д���ӷ��ţ���������Һ�л�����Fe2+�ķ����� ��д�����衢�Լ�������
��3����ȡҺ����м��Ӧ�����ӷ���ʽΪ ��X�����壩�Ļ�ѧʽΪ ��
��4���ڢ۲���Ӧ��ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ijCuSO4��Fe2(SO4)3��H2SO4�Ļ����Һ100 mL����֪��Һ�и������ӵ�Ũ����ͬ���� ������Һ�����ӵı仯������SO42�������ʵ���Ũ��Ϊ6mol �������Һ������ܽ����۵�����Ϊ
�������Һ������ܽ����۵�����Ϊ
A��5.6g B��11.2 g C��22.4g D��33.6g
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ� ��
A�����³�ѹ�£�100g98%������Һ�к��е���ԭ����Ϊ4NA
B����״���£������ý���ת��2NA������ʱ���ɴ��������û�����44��8LH2
C��15gCH3+�к��еĵ�����Ϊ0��8NA
D��1molH2O2�����зǼ��Թ��ۼ���ΪNA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
̼����李�����茶����ҹ���Ҫ�ĵ���Ʒ�֣�̼������������������������ӷ���ʧ��Ϊ�˼�����������ȷ�����ʩ����������ⶨ�京������
I��ijѧ�����ⶨ̼����炙����еĺ�������ѡ������װ��ͨ���ⶨ���ɶ�����̼������Ӳ�����ʵĺ�������

(1)��ѡ���Ҫ��װ�ã�ÿ��װ�����ʹ��һ�Σ�����������������˳��Ϊ_______________��װ��װ�ã���������װ��������
(2) ���������Ժ���Ʒ����Բ����ƿ�У��μ�Һ�塣��Һ�����ʺϵ���_________________.
A.ϡ���� B.ϡ���� C.Ũ���� D.��������
II����������гɷ�������泥�������ü�ȩ���ⶨ����������ӦΪ:
2(NH4)2SO4+6HCHO=(CH2)6N4+2H2SO4�ٽ����ɵ������������Ʊ���Һ�ζ����Ӷ�������ĺ������������£�
��3���ò�������ȡ����(NH4)2SO4��Ʒ0.6g���ձ��У�������ˮ�ܽ����100mL��Һ��ȷ��ȡ20.00mL�Ĵ���Һ��Һ����ƿ�У���������l8%�����Լ�ȩ������5min����1-2��_______ָʾ������֪�ζ��յ��pHԼΪ8.8������Ũ��Ϊ0.0800mol/L�������Ʊ���Һ�ζ����յ㡣���ظ��ζ�2�Σ����εζ��Ķ������±���
| �ζ����� | �ζ�ǰ������mL�� | �ζ��������mL�� |
| 1 | 1.20 | 16.21 |
| 2 | 3.00 | 18.90 |
| 3 | 4.50 | 19.49 |
���������Ʒ�еĵ�����������Ϊ_______________��
��4���ڵζ�ʵ��������ֵζ��õļ�ʽ�ζ��ܲ��������ڳ��������ݣ��ζ���ʼʱ�����ݣ����ʵ��ⶨ�ĺ�������ʵ��ֵ________���ƫ��ƫС������Ӱ�족����
��5������ⶨ̼����炙����еĺ�����ʱ��Ҳʹ�ü�ȩ���Ƿ����____________(��ǡ�����������_______________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���ȸʯ��һ�ֺ�ͭ�Ŀ�ʯ����ͭ��̬Ϊ
 ��ͬʱ����
��ͬʱ���� �����ʡ��Թ��ȸʯΪԭ����ȡ����ͭ�Ĺ�����������ͼ��
�����ʡ��Թ��ȸʯΪԭ����ȡ����ͭ�Ĺ�����������ͼ��

��ش��������⣺
����ɲ������ϡ������ ������Ӧ�Ļ�ѧ����ʽ
������Ӧ�Ļ�ѧ����ʽ
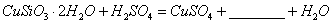 ��
��
�����ӷ���ʽ��ʾ˫��ˮ������_____________________________��
�Ʋ���ڵ�����ҺpHѡ�õ�����Լ���__________________
A. B.CuO C.A12O3 D.
B.CuO C.A12O3 D.
���й��������↑ʼ��������ȫ������pH���±���

���ϱ���֪������ҺpH=4ʱ��������ȫ��ȥ��������______��������ȫ��ȥ��������________��
����ҺBͨ������Ũ���������Ũ��Ϊԭ����һ�룩����ȴ�ᾧ���Եõ� ���塣ijͬѧ��Ϊ������������������������������������������ݶԸ�ͬѧ�Ĺ۵��������ۣ���֪�����£�
���塣ijͬѧ��Ϊ������������������������������������������ݶԸ�ͬѧ�Ĺ۵��������ۣ���֪�����£� ������Һ��
������Һ��
 mol
mol ______________��
______________��
����Ҫ�ⶨ����ͭ�� ���нᾧˮ�ĺ�������Ҫ�������Ǿƾ��ơ�������ƽ�����Ǽܡ������ǡ���������������������ǯ���в���ҩ�ס�_________________��ʵ�����������ͭ�������ʧˮ���ڿ�����ȴ���������ⶨ���______________(�ƫ�ߡ��� ��ƫ�͡����䡱)��
���нᾧˮ�ĺ�������Ҫ�������Ǿƾ��ơ�������ƽ�����Ǽܡ������ǡ���������������������ǯ���в���ҩ�ס�_________________��ʵ�����������ͭ�������ʧˮ���ڿ�����ȴ���������ⶨ���______________(�ƫ�ߡ��� ��ƫ�͡����䡱)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ڱ�״���£���������ƿ�ڷֱ�װ�и��������NH3����һ��������Ȼ������塢NO2��O2�����Ϊ4��1�Ļ�����塣Ȼ��ֱ�����Ȫʵ�飬������ƿ��������Һ�����ʵ���Ũ��֮��Ϊ(�� )
A��2��1��2 B��5��5��4
C��1��1��1 D����ȷ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com