
A��B��C��D��E����5��Ԫ�ء�����գ�
(1)AԪ�ػ�̬ԭ�ӵ��������3��δ�ɶԵ��ӣ��������2�����ӣ���Ԫ�ط���Ϊ________������̬�⻯�ﻯѧʽΪ________��
(2)BԪ�صĸ�һ�����Ӻ�CԪ�ص���һ�����ӵĵ��Ӳ�ṹ�������ͬ��B��Ԫ�ط���Ϊ________��������������ˮ����Ļ�ѧʽΪ________��C��Ԫ�ط���Ϊ________��C�����ڱ��е�λ��Ϊ________��
(3)DԪ�ص����������ӵ�3d�Dz�Ϊ�������D��Ԫ�ط���Ϊ________�����̬ԭ�ӵĵ����Ų�ʽΪ ��
(4)EԪ�ػ�̬ԭ�ӵ�M��ȫ������N��û�гɶԵ��ӣ�ֻ��һ��δ�ɶԵ��ӣ�E��Ԫ�ط���Ϊ________�����̬ԭ�ӵĵ����Ų�ʽΪ ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
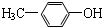
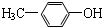
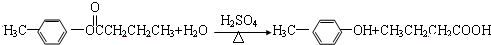
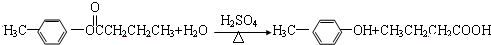


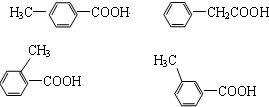

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
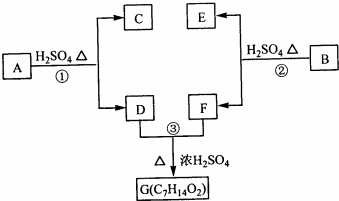
 ������A��B��C��D��E��F��G��H�����л����������ͼ��ʾ��ת����ϵ��
������A��B��C��D��E��F��G��H�����л����������ͼ��ʾ��ת����ϵ��
| ��� | �� | �� | �� | �� | �� |
| ����Ӧ�� | NaOH��Һ | ���� | ����Cu��OH��2 | ϡ���� | H2O |
| ��Ӧ���� | ���� | - | ���� | ���� | ���� HgSO4 |
| ���� |
| ���� |




�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
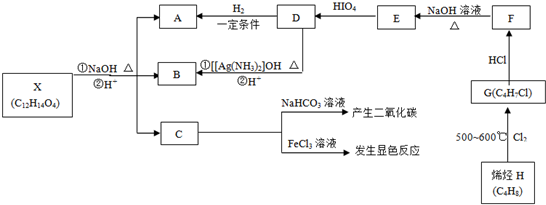
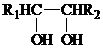 +HIO4��R1CHO+R2CHO+HIO3+H2O
+HIO4��R1CHO+R2CHO+HIO3+H2O

| 500��600�� |
| 500��600�� |
| �� |
| �� |
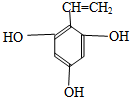
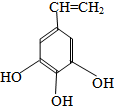 ��
��
 ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


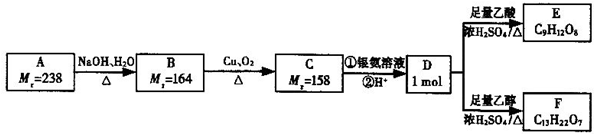
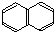 ������ú�����У�����ԭ�Ӿ�����ͬһƽ���ڣ���1mol E ����ܺ�
������ú�����У�����ԭ�Ӿ�����ͬһƽ���ڣ���1mol E ����ܺ�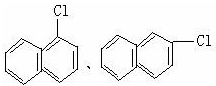

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com